Abstract
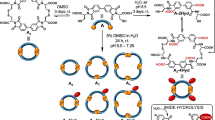
This protocol describes the synthesis of cyclic anhydride precursors of the environment-sensitive fluorophores 4-dimethylaminophthalimide (4-DMAP) and 6-dimethylaminonaphthalimide (6-DMN). The condensation of these anhydrides with a primary amino group confers on molecules of interest solvatochromic properties. In particular, two strategies for the insertion of the chromophores into peptides are presented in two companion protocols. The anhydride syntheses can be completed on the gram scale in 2 d for the 4-DMAP precursor and 10–15 d for the 6-DMN precursor.
This is a preview of subscription content, access via your institution
Access options


Similar content being viewed by others
References
Valeur, B. Molecular Fluorescence: Principles and Applications (Wiley-VCH, Weinheim, Germany, 2002).
Lakowicz, J.R. Principles of Fluorescence Spectroscopy (Springer, New York, 2006).
Vazquez, M.E., Blanco, J.B. & Imperiali, B. Photophysics and biological applications of the environment-sensitive fluorophore 6-N,N-dimethylamino-2,3-naphthalimide. J. Am. Chem. Soc. 127, 1300–1306 (2005).
Vazquez, M.E., Rothman, D.M. & Imperiali, B. A new environment-sensitive fluorescent amino acid for Fmoc-based solid phase peptide synthesis. Org. Biomol. Chem. 2, 1965–1966 (2004).
Sainlos, M. & Imperiali, B. Tools for investigating peptide–protein interactions: peptide incorporation of environment-sensitive fluorophores via on-resin derivatization. Nat. Protoc. 2, 3201–3209 (2007).
Sainlos, M. & Imperiali, B. Tools for investigating peptide–protein interactions: peptide incorporation of environment-sensitive fluorophores through SPPS-based 'building block' approach. Nat. Protoc. 2, 3210–3218 (2007).
Acknowledgements
This research was supported by NSF CHE-0414243 (B.I.) and the Cell Migration Consortium (GM064346). The award of a Marie Curie Fellowship to M.S. is also gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that a patent on the environment–sensitive fluorophores is pending: “Fluorescent Probes for Biological Studies” by Imperiali et al. US Patent Application serial No. 11/106,349, filed April 13, 2005, pending.
Rights and permissions
About this article
Cite this article
Sainlos, M., Imperiali, B. Synthesis of anhydride precursors of the environment-sensitive fluorophores 4-DMAP and 6-DMN. Nat Protoc 2, 3219–3225 (2007). https://doi.org/10.1038/nprot.2007.444
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.444
This article is cited by
-
Biomimetic divalent ligands for the acute disruption of synaptic AMPAR stabilization
Nature Chemical Biology (2011)
-
Short and scalable synthesis of an anhydride precursor of the environment-sensitive fluorophore 6-dimethylaminonaphthalimide
Nature Protocols (2011)
-
Tools for investigating peptide–protein interactions: peptide incorporation of environment-sensitive fluorophores via on-resin derivatization
Nature Protocols (2007)
-
Tools for investigating peptide–protein interactions: peptide incorporation of environment-sensitive fluorophores through SPPS-based 'building block' approach
Nature Protocols (2007)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.