Abstract
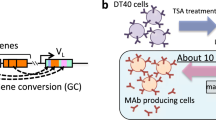
Here, we describe a protocol for using the ADLib (Autonomously Diversifying Library) system to rapidly generate specific monoclonal antibodies using DT40, a chicken B-cell line that undergoes constitutive gene conversion at both light- and heavy-chain immunoglobulin loci. We previously developed the ADLib system on the basis of our finding that gene conversion in DT40 cells was enhanced by treatment of the cells with a histone deacetylase inhibitor, trichostatin A (TSA). TSA treatment evolves a diversified library of DT40 cells (ADLib), in which each cell has different surface IgM specificity. Antigen-specific DT40 cells are selected from ADLib using antigen-conjugated magnetic beads, and their specificity can be examined by various immunological assays, using culture supernatant containing secreted IgM. The whole process from selection to screening can be completed in about 1 week. Thus, the ADLib system will accelerate biological studies, including drug discovery and design.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Köhler, G. & Milstein, C. Continuous cultures of fused cells secreting antibody of predefined specificity. Nature 256, 495–497 (1975).
Griffiths, A.D. & Duncan, A.R. Strategies for selection of antibodies by phage display. Curr. Opin. Biotechnol. 9, 102–108 (1998).
Smith, G.P. Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface. Science 228, 1315–1317 (1985).
Winter, G., Griffiths, A.D., Hawkins, R.E. & Hoogenboom, H.R. Making antibodies by phage display technology. Annu. Rev. Immunol. 12, 433–455 (1994).
Ohta, K., Shibata, T. & Nicolas, A. Changes in chromatin structure at recombination initiation sites during yeast meiosis. EMBO J. 13, 5754–5763 (1994).
Yamada, T. et al. Roles of histone acetylation and chromatin remodeling factor in a meiotic recombination hotspot. EMBO J. 23, 1792–1803 (2004).
Reynaud, C.A., Anquez, V., Dahan, A. & Weill, J.C. A single rearrangement event generates most of the chicken immunoglobulin light chain diversity. Cell 40, 283–291 (1985).
Reynaud, C.A., Anquez, V., Grimal, H. & Weill, J.C. A hyperconversion mechanism generates the chicken light chain preimmune repertoire. Cell 48, 379–388 (1987).
Reynaud, C.A., Dahan, A., Anquez, V. & Weill, J.C. Somatic hyperconversion diversifies the single Vh gene of the chicken with a high incidence in the D region. Cell 59, 171–183 (1989).
McCormack, W.T. & Thompson, C.B. Somatic diversification of the chicken immunoglobulin light-chain gene. Adv. Immunol. 48, 41–67 (1990).
McCormack, W.T. & Thompson, C.B. Chicken IgL variable region gene conversions display pseudogene donor preference and 5′ to 3′ polarity. Genes Dev. 4, 548–558 (1990).
Seo, H. et al. Rapid generation of specific antibodies by enhanced homologous recombination. Nat. Biotechnol. 23, 731–735 (2005).
Muramatsu, M. et al. Class switch recombination and hypermutation require activation-induced cytidine deaminase (AID), a potential RNA editing enzyme. Cell 102, 553–563 (2000).
Arakawa, H., Hauschild, J. & Buerstedde, J.M. Requirement of the activation-induced deaminase (AID) gene for immunoglobulin gene conversion. Science 295, 1301–1306 (2002).
Harris, R.S., Sale, J.E., Petersen-Mahrt, S.K. & Neuberger, M.S. AID is essential for immunoglobulin V gene conversion in a cultured B cell line. Curr. Biol. 12, 435–438 (2002).
Buerstedde, J.M. et al. Light chain gene conversion continues at high rate in an ALV-induced cell line. EMBO J. 9, 921–927 (1990).
Takata, M. et al. Homologous recombination and non-homologous end-joining pathways of DNA double-strand break repair have overlapping roles in the maintenance of chromosomal integrity in vertebrate cells. EMBO J. 17, 5497–5508 (1998).
Sonoda, E. et al. Rad51-deficient vertebrate cells accumulate chromosomal breaks prior to cell death. EMBO J. 17, 598–608 (1998).
Acknowledgements
We thank Drs. S. Takeda, M. Takata, T. Yamada, K. Hirota, T. Fukuda and K. Okuhara for helpful advice and discussion; Mr. H. Sasanuma for helpful advice on antibody selection; Ms. K. Arai, A. Nakamura, M. Masuoka, M. Asakawa and Mr. N. Harigai for their experimental support; and all members of Chiome Bioscience Inc. for their help. This work was supported by grants from the REDS (Rational Evolutionary Design of Advanced Biomolecule, Saitama) project, basic research from the Bio-oriented Technology Research Advancement Institution (to T. Shibata and K. Ohta), the New Energy and Industrial Technology Development Organization (to M. Fujiwara) and grants-in-aid for scientific research on priority areas from the Ministry of Education, Culture, Sports, Science and Technology of Japan (to K. Ohta).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
1. We are applying patents that are based on the research. 2. Some authors are founders, employees, and stock-holders of Chiome Bioscience, Inc., a private venture company in Tokyo, Japan.
Rights and permissions
About this article
Cite this article
Seo, H., Hashimoto, Si., Tsuchiya, K. et al. An ex vivo method for rapid generation of monoclonal antibodies (ADLib system). Nat Protoc 1, 1502–1506 (2006). https://doi.org/10.1038/nprot.2006.248
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.248
This article is cited by
-
Prediction and verification of the AD-FTLD common pathomechanism based on dynamic molecular network analysis
Communications Biology (2021)
-
Streamlined human antibody generation and optimization by exploiting designed immunoglobulin loci in a B cell line
Cellular & Molecular Immunology (2021)
-
HMGB1 signaling phosphorylates Ku70 and impairs DNA damage repair in Alzheimer’s disease pathology
Communications Biology (2021)
-
Methods and cell-based strategies to produce antibody libraries: current state
Applied Microbiology and Biotechnology (2021)
-
Ex vivo evolution of human antibodies by CRISPR-X: from a naive B cell repertoire to affinity matured antibodies
BMC Biotechnology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.