Abstract
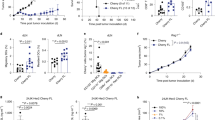
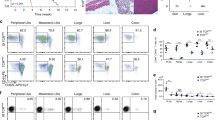
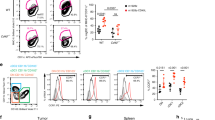
CD8+ T cells can mediate eradication of established tumors, and strategies to amplify tumor-reactive T-cell numbers by immunization or ex vivo expansion followed by adoptive transfer are currently being explored in individuals with cancer1,2,3. Generating effective CD8+ T cell–mediated responses to tumors is often impeded by T-cell tolerance to relevant tumor antigens, as most of these antigens are also expressed in normal tissues. We examined whether such tolerant T cells could be rescued and functionally restored for use in therapy of established tumors. We used a transgenic T-cell receptor (TCR) mouse model in which peripheral CD8+ T cells specific for a candidate tumor antigen also expressed in liver are tolerant, failing to proliferate or secrete interleukin (IL)-2 in response to antigen4. Molecular and cellular analysis showed that these tolerant T cells expressed the IL-15 receptor α chain, and could be induced to proliferate in vitro in response to exogenous IL-15. Such proliferation abrogated tolerance and the rescued cells became effective in treating leukemia. Therefore, high-affinity CD8+ T cells are not necessarily deleted by encounter with self-antigen in the periphery, and can potentially be rescued and expanded for use in tumor immunotherapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Blattman, J.N. & Greenberg, P.D. Cancer immunotherapy: a treatment for the masses. Science 305, 200–205 (2004).
Rosenberg, S.A., Yang, J.C. & Restifo, N.P. Cancer immunotherapy: moving beyond current vaccines. Nat. Med. 10, 909–915 (2004).
Kershaw, M.H., Teng, M.W., Smyth, M.J. & Darcy, P.K. Supernatural T cells: genetic modification of T cells for cancer therapy. Nat. Rev. Immunol. 5, 928–940 (2005).
Ohlen, C. et al. CD8+ T cell tolerance to a tumor-associated antigen is maintained at the level of expansion rather than effector function. J. Exp. Med. 195, 1407–1418 (2002).
Redmond, W.L. & Sherman, L.A. Peripheral tolerance of CD8 T lymphocytes. Immunity 22, 275–284 (2005).
Greenberg, P.D. & Cheever, M.A. Treatment of disseminated leukemia with cyclophosphamide and immune cells: tumor immunity reflects long-term persistence of tumor-specific donor T cells. J. Immunol. 133, 3401–3407 (1984).
Grayson, J.M., Zajac, A.J., Altman, J.D. & Ahmed, R. Cutting edge: increased expression of Bcl-2 in antigen-specific memory CD8+ T cells. J. Immunol. 164, 3950–3954 (2000).
Dumortier, H. et al. Antigen presentation by an immature myeloid dendritic cell line does not cause CTL deletion in vivo, but generates CD8+ central memory-like T cells that can be rescued for full effector function. J. Immunol. 175, 855–863 (2005).
Carson, W.E. et al. Endogenous production of interleukin 15 by activated human monocytes is critical for optimal production of interferon-gamma by natural killer cells in vitro. J. Clin. Invest. 96, 2578–2582 (1995).
Ferretti, S., Bonneau, O., Dubois, G.R., Jones, C.E. & Trifilieff, A. IL-17, produced by lymphocytes and neutrophils, is necessary for lipopolysaccharide-induced airway neutrophilia: IL-15 as a possible trigger. J. Immunol. 170, 2106–2112 (2003).
Betts, M.R. et al. Sensitive and viable identification of antigen-specific CD8+ T cells by a flow cytometric assay for degranulation. J. Immunol. Methods 281, 65–78 (2003).
Ohlen, C., Kalos, M., Hong, D.J., Shur, A.C. & Greenberg, P.D. Expression of a tolerizing tumor antigen in peripheral tissue does not preclude recovery of high-affinity CD8+ T cells or CTL immunotherapy of tumors expressing the antigen. J. Immunol. 166, 2863–2870 (2001).
Wirth, S., Guidotti, L.G., Ando, K., Schlicht, H.J. & Chisari, F.V. Breaking tolerance leads to autoantibody production but not autoimmune liver disease in hepatitis B virus envelope transgenic mice. J. Immunol. 154, 2504–2515 (1995).
Guidotti, L.G. et al. Intracellular inactivation of the hepatitis B virus by cytotoxic T lymphocytes. Immunity 4, 25–36 (1996).
Limmer, A. et al. Failure to induce organ-specific autoimmunity by breaking of tolerance: importance of the microenvironment. Eur. J. Immunol. 28, 2395–2406 (1998).
Ahmed, R. & Gray, D. Immunological memory and protective immunity: understanding their relation. Science 272, 54–60 (1996).
Wolint, P., Betts, M.R., Koup, R.A. & Oxenius, A. Immediate cytotoxicity but not degranulation distinguishes effector and memory subsets of CD8+ T cells. J. Exp. Med. 199, 925–936 (2004).
Zhang, X., Sun, S., Hwang, I., Tough, D.F. & Sprent, J. Potent and selective stimulation of memory-phenotype CD8+ T cells in vivo by IL-15. Immunity 8, 591–599 (1998).
Grabstein, K.H. et al. Cloning of a T cell growth factor that interacts with the beta chain of the interleukin-2 receptor. Science 264, 965–968 (1994).
Waldmann, T.A. The IL-2/IL-15 receptor systems: targets for immunotherapy. J. Clin. Immunol. 22, 51–56 (2002).
Bamford, R.N. et al. The interleukin (IL) 2 receptor beta chain is shared by IL-2 and a cytokine, provisionally designated IL-T, that stimulates T-cell proliferation and the induction of lymphokine-activated killer cells. Proc. Natl. Acad. Sci. USA 91, 4940–4944 (1994).
Smith, K.A. Interleukin-2: inception, impact, and implications. Science 240, 1169–1176 (1988).
Nelson, B.H. & Willerford, D.M. Biology of the interleukin-2 receptor. Adv. Immunol. 70, 1–81 (1998).
Lee, K.H. et al. The immunological synapse balances T cell receptor signaling and degradation. Science 302, 1218–1222 (2003).
Grakoui, A. et al. The immunological synapse: a molecular machine controlling T cell activation. Science 285, 221–227 (1999).
Purbhoo, M.A., Irvine, D.J., Huppa, J.B. & Davis, M.M. T cell killing does not require the formation of a stable mature immunological synapse. Nat. Immunol. 5, 524–530 (2004).
xDeSilva, D.R., Urdahl, K.B. & Jenkins, M.K. Clonal anergy is induced in vitro by T cell receptor occupancy in the absence of proliferation. J. Immunol. 147, 3261–3267 (1991).
Oh, S., Perera, L.P., Burke, D.S., Waldmann, T.A. & Berzofsky, J.A. IL-15/IL-15Ralpha-mediated avidity maturation of memory CD8+ T cells. Proc. Natl. Acad. Sci. USA 101, 15154–15159 (2004).
Vamosi, G. et al. IL-2 and IL-15 receptor alpha-subunits are coexpressed in a supramolecular receptor cluster in lipid rafts of T cells. Proc. Natl. Acad. Sci. USA 101, 11082–11087 (2004).
Dubois, S., Mariner, J., Waldmann, T.A. & Tagaya, Y. IL-15Ralpha recycles and presents IL-15 In trans to neighboring cells. Immunity 17, 537–547 (2002).
Bulanova, E. et al. The IL-15R alpha chain signals through association with Syk in human B cells. J. Immunol. 167, 6292–6302 (2001).
Rosenberg, S.A. & Dudley, M.E. Cancer regression in patients with metastatic melanoma after the transfer of autologous antitumor lymphocytes. Proc. Natl. Acad. Sci. USA 101 Suppl 2, 14639–14645 (2004).
Zippelius, A. et al. Effector function of human tumor-specific CD8 T cells in melanoma lesions: a state of local functional tolerance. Cancer Res. 64, 2865–2873 (2004).
Scherer, A. et al. Optimized protocol for linear RNA amplification and application to gene expression profiling of human renal biopsies. BioTechniques 34, 546–50, 552–4, 556 (2003).
Su, A.I. et al. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc. Natl. Acad. Sci. USA 101, 6062–6067 (2004).
Acknowledgements
This work was supported in part by grants CA33084 and CA18029 from the US National Institutes of Health/National Cancer Institute, and by a grant from the Leukemia and Lymphoma Society. R. Teague was supported by a Ruth L. Kirschstein National Research Service Award.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Teague, R., Sather, B., Sacks, J. et al. Interleukin-15 rescues tolerant CD8+ T cells for use in adoptive immunotherapy of established tumors. Nat Med 12, 335–341 (2006). https://doi.org/10.1038/nm1359
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1359
This article is cited by
-
Recent advances and applications of CRISPR-Cas9 in cancer immunotherapy
Molecular Cancer (2023)
-
Targeting ubiquitin signaling for cancer immunotherapy
Signal Transduction and Targeted Therapy (2021)
-
Decreased levels of circulating cytokines VEGF, TNF-β and IL-15 indicate PD-L1 overexpression in tumours of primary breast cancer patients
Scientific Reports (2021)
-
Cancer therapy in mice using a pure population of CD8+ T cell specific to the AH1 tumor rejection antigen
Cancer Immunology, Immunotherapy (2021)
-
Disturbed mitochondrial dynamics in CD8+ TILs reinforce T cell exhaustion
Nature Immunology (2020)