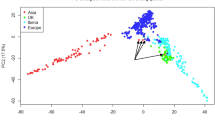
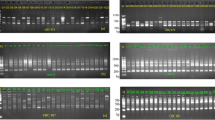
Abstract
Gliricidia sepium and G. maculata are multi-purpose leguminous trees native to Central America and Mexico. Research programmes have been initiated to define the native distribution of Gliricidia and sample the spectrum of genetic variation. To date, there has been little systematic assessment of genetic variability in multi-purpose tree species. Accurate estimates of diversity between- and within-populations are considered a prerequisite for the optimization of sampling and breeding strategies. We have used a PCR-based polymorphic assay procedure (RAPDs) to monitor genetic variability in Gliricidia. Extensive genetic variability was detected between species and the variability was partitioned into between- and within-population components. On average, most (60 per cent) of the variation occurs between G. sepium populations but oligonucleotide primers differed in their capacity to detect variability between and within populations. Population-specific genetic markers were identified. RAPDs provide a cost-effective method for the precise and routine evaluation of variability and may be used to identify areas of maximum diversity. The approaches outlined have general applicability to a range of organisms and are discussed in relation to the exploitation of multi-purpose tree species of the tropics.
Similar content being viewed by others
Article PDF
References
Baird, E, Cooper-Bland, S, Waugh, R, De Maine, M, and Powell, W. 1992. Molecular characterization of inter- and intra-specific somatic hybrids of potato using randomly amplified polymorphic DNA (RAPD) markers. Mol Gen Genet, (in press).
Brown, A D H. 1979. Enzyme polymorphisms in plant populations. Theor Pop Biol, 15, 1–42.
Clegg, M T. 1980. Measuring plant mating systems. Bioscience, 30, 814–818.
Clegg, M T. 1989a. Analyses of molecular diversity within and among plant species. In: Helentjaris, T. and Burr, B. (ed.) Development and Application of Molecular Markers to Problems in Plant Genetics. Current Communications in Molecular Biology, Cold Spring Harbor Laboratory, New York. pp. 51–56.
Clegg, M T. 1989b. Molecular diversity in plant populations. In: Brown, A. H. D. (ed.) Population Genetics, Plant Breeding and Genetic Conservation, Sinauer Associates, Sunderland, MA. pp. 98–115.
Edwards, K, Johnstone, C, and Thompson, C. 1991. A Simple method for the preparation of plant genomic DNA for PCR analysis. Nucl Acids Res, 19, 1349.
Feinberg, A P, and Vogelstein, B. 1984. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity (Addendum). Anal Biochem, 137, 266–267.
Gottlieb, L D. 1977. Electrophoretic evidence and plant systematics. Ann Missouri Bot Gard, 64, 161–180.
Hamrick, J L. 1990. Isozymes and the analysis of genetic structure in plant populations. In: Soltis, E. D. and Soltis, P. S. (eds) Isozymes in Plant Biology. Chapman and Hall, London, pp. 87–105.
Hughes, C E. 1987. Biological considerations in designing a seed collection strategy for Gliricidia sepium (JACO.) WALP. (Leguminosae). Commonwealth For Rev, 66, 31–48.
Innis, M A, Gelfond, D H, Sninsky, J J, and White, T J. 1990. PCR protocols. In: A Guide to Methods and Applications. Academic Press, New York.
King, L M, and Schaal, B A. 1989. Ribosomal DNA variation and distribution in Rudbeckia missouriensis. Evolution, 43, 1117–1119.
Loveless, M D, and Hamrick, J L. 1984. Ecological determinants of genetic structure in plant populations. Ann Rev Ecol Syst, 15, 65–95.
Maniatis, T, Fritsch, E F, and Sambrook, J. 1982. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, New York.
Martin, G B, Williams, J G K, and Tanksley, S D. 1991. Rapid identification of markers linked to a Pseudomonas resistance gene in tomato by using random primers and near isogenic lines. Proc Nat Acad Sci, USA, 88, 2336–2340.
Michelmore, R W, Paran, I, and Kesseli, R V. 1992. Identification of markers linked to disease resistance genes by bulked segregant analysis: A rapid method to detect markers in specific genomic regions using segregating populations. Proc Natl Acad Sci, USA, 88, 9828–9832.
Namkoong, G. 1986. Genetics and the forests of the future. Unasylva, 152, 2–18.
Nei, M. 1972. Genetic distance between populations. Am Nat, 106, 283–292.
Nei, M, and Li, W-H. 1976. The transit distribution of allele frequencies under mutation pressure. Genet Res, 28, 205–214.
Nei, M, and Li, W-H. 1979. Mathematical model for studying genetical variation in terms of restriction endonucleases. Proc Natl Acad Sci, USA, 74, 5267–5273.
Quiros, C F, Hu, J, This, P, Chevre, A M, and Delseny, M. 1991. Development and chromosomal localization of genome-specific markers by polymerase chain reaction in Brassica. Theor Appl Genet, 82, 627–632.
Rafalski, J A, Tingey, S V, and Williams, J G K. 1991. RAPD markers — A new technology for genetic mapping and plant breeding. Agbiotech News Inf, 3, 645–648.
Saiki, R K, Gelfond, D H, Stoffel, S, Scharf, S J, Higuchi, R, Horn, B T, Mullis, K B, and Erlich, H A. 1988. Primer directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science, 239, 487–491.
Simons, A J. 1991. Genetic improvement of non-industrial trees. Agroforest Syst, 18, 197–212.
Soltis, E D, and Soltis, P S. 1990. Isozymes in Plant Biology. Chapman and Hall, London.
Welsh, J, and McClelland, M. 1990. Fingerprinting genomes using PCR with arbitrary primers. Nucl Acids Res, 18, 7213–7218.
Wilde, J, Waugh, R, and Powell, W. 1992. Genetic fingerprinting of Theobroma clones using RAPD markers. Theor Appl Genet, 88, 871–877.
Williams, J G K, Kubelik, A R, Livak, K J, Rafalski, J A, and Tingey, S V. 1990. DNA polymorphisms amplified by arbitrary primers are useful as genetic markers. Nucl Acids Res, 18, 6531–6535.
Yeh, F C H. 1989. Isozyme analysis for revealing population structure for use in breeding strategies. In: Gibson, G. L. Griffin, A. R. and Matheson, A. C. (eds). Breeding Tropical Trees: Population Structure and Genetic Improvement Strategies in Clonal and Seedling Forestry, IVFRO Pattaya, Thailand, pp. 119–131.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chalmers, K., Waugh, R., Sprent, J. et al. Detection of genetic variation between and within populations of Gliricidia sepium and G. maculata using RAPD markers. Heredity 69, 465–472 (1992). https://doi.org/10.1038/hdy.1992.151
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1992.151
Keywords
This article is cited by
-
Genetic diversity in populations of the medicinal plant Leonurus cardiaca L. revealed by inter-primer binding site (iPBS) markers
Genetic Resources and Crop Evolution (2017)
-
Biomarker System for Genetic Analysis of Stored Grain Pests Callosobruchus maculatus and Callosobruchus chinensis
Proceedings of the National Academy of Sciences, India Section B: Biological Sciences (2015)
-
Efficient genomic DNA extraction protocol from medicinal rich Passiflora foetida containing high level of polysaccharide and polyphenol
SpringerPlus (2014)
-
Morphological and AFLP diversity in Thaumatococcus daniellii, the source of the protein sweetener thaumatin
Genetic Resources and Crop Evolution (2012)
-
Genetic Variation in Chinese Pine (Pinus tabulaeformis), a Woody Species Endemic to China
Biochemical Genetics (2009)