Abstract
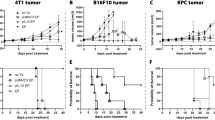
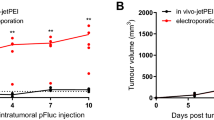
Enhanced tumor delivery of plasmid DNA with electric pulses in vivo has been confirmed in many preclinical models. Intratumor electrotransfer of plasmids encoding therapeutic molecules has reached Phase II clinical trials. In multiple preclinical studies, a reduction in tumor growth, increased survival or complete tumor regression have been observed in control groups in which vector or backbone plasmid DNA electrotransfer was performed. This study explores factors that could produce this antitumor effect. The specific electrotransfer pulse protocol employed significantly potentiated the regression. Tumor regression was observed after delivery of single-stranded or double-stranded DNA with or without CpG motifs in both immunocompetent and immunodeficient mice, indicating the involvement of the innate immune system in response to DNA. In conclusion, this study demonstrated that the observed antitumor effects are not due to a single factor, but to a combination of factors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Heller LC, Heller R . In vivo electroporation for gene therapy. Hum Gene Ther 2006; 17: 890–897.
Cemazar M, Sersa G . Electrotransfer of therapeutic molecules into tissues. Curr Opin Mol Ther 2007; 9: 554–562.
Heller LC, Heller R . Translation of electroporation mediated DNA delivery to the clinic. In: Markov M, Miklavcic D, Pakhomov A (eds) Advanced Electroporation Techniques in Biology and Medicine. CRC Press, 2010 pp 19–1-19-13.
Heller LC, Heller R . Electroporation gene therapy preclinical and clinical trials for melanoma. Curr Gene Ther 2010; 10: 312–317.
Pavlin D, Cemazar M, Cör A, Sersa G, Pogacnik A, Tozon N . Electrogene therapy with interleukin-12 in canine mast cell tumors. Radiol Oncol 2011; 45: 30–39.
Daud AI, DeConti RC, Andrews S, Urbas P, Riker AI, Sondak VK et al. Phase I trial of interleukin-12 plasmid electroporation in patients with metastatic melanoma. J Clin Oncol 2008; 26: 5896–5903.
Niu G, Heller R, Catlett-Falcone R, Coppola D, Jaroszeski M, Dalton W et al. Gene therapy with dominant-negative Stat3 suppresses growth of the murine melanoma B16 tumor in vivo. Cancer Res 1999; 59: 5059–5063.
Heller L, Coppola D . Electrically mediated delivery of vector plasmid DNA elicits an antitumor effect. Gene Ther 2002; 9: 1321–1325.
McCray AN, Ugen KE, Muthumani K, Kim JJ, Weiner DB, Heller R . Complete regression of established subcutaneous B16 murine melanoma tumors after delivery of an HIV-1 Vpr-expressing plasmid by in vivo electroporation. Mol Ther 2006; 14: 647–655.
Ugen KE, Kutzler MA, Marrero B, Westover J, Coppola D, Weiner DB et al. Regression of subcutaneous B16 melanoma tumors after intratumoral delivery of an IL-15-expressing plasmid followed by in vivo electroporation. Cancer Gene Ther 2006; 13: 969–974.
Marrero B, Shirley S, Heller R . Delivery of interleukin-15 to B16 melanoma by electroporation leads to tumor regression and long-term survival. Technol Cancer Res Treat 2013 (e-pub ahead of print; doi:10.7785/tcrtexpress.2013.600252.
Slack A, Bovenzi V, Bigey P, Ivanov MA, Ramchandani S, Bhattacharya S et al. Antisense MBD2 gene therapy inhibits tumorigenesis. J Gene Med 2002; 4: 381–389.
Prud'homme GJ, Glinka Y, Khan AS, Draghia-Akli R . Electroporation-enhanced nonviral gene transfer for the prevention or treatment of immunological endocrine and neoplastic diseases. Curr Gene Ther 2006; 6: 243–273.
Grosel A, Sersa G, Kranjc S, Cemazar M . Electrogene therapy with p53 of murine sarcomas alone or combined with electrochemotherapy using cisplatin. DNA Cell Biol 2006; 25: 674–683.
Deharvengt S, Rejiba S, Wack S, Aprahamian M, Hajri A . Efficient electrogene therapy for pancreatic adenocarcinoma treatment using the bacterial purine nucleoside phosphorylase suicide gene with fludarabine. Int.J.Oncol 2007; 30: 1397–1406.
Radkevich-Brown O, Piechocki MP, Back JB, Weise AM, Pilon-Thomas S, Wei WZ et al. electroporation induces anti-tumor immunity and tumor regression. Cancer Immunol.Immunother 2010; 59: 409–417.
Wang YS, Tsang YW, Chi CH, Chang CC, Chu RM, Chi KH . Synergistic anti-tumor effect of combination radio- and immunotherapy by electro-gene therapy plus intra-tumor injection of dendritic cells. Cancer Lett 2008; 266: 275–285.
Vidic S, Markelc B, Sersa G, Coer A, Kamensek U, Tevz G et al. MicroRNAs targeting mutant K-ras by electrotransfer inhibit human colorectal adenocarcinoma cell growth in vitro and in vivo. Cancer Gene Therapy 2010; 17: 409–419.
Elez R, Piiper A, Kronenberger B, Kock M, Brendel M, Hermann E et al. Tumor regression by combination antisense therapy against Plk1 and Bcl-2. Oncogene 2003; 22: 69–80.
Spugnini EP, Biroccio A, De Mori R, Scarsella M, D'Angelo C, Baldi A et al. Electroporation increases antitumoral efficacy of the bcl-2 antisense G3139 and chemotherapy in a human melanoma xenograft. J Transl Med 2011; 9: 125.
Rols MP, Delteil C, Golzio M, Dumond P, Cros S, Teissie J . In vivo electrically mediated protein and gene transfer in murine melanoma. Nat Biotechnol 1998; 16: 168–171.
Lohr F, Lo DY, Zaharoff DA, Hu K, Zhang X, Li Y et al. Effective tumor therapy with plasmid-encoded cytokines combined with in vivo electroporation. Cancer Res 2001; 61: 3281–3284.
Cichon T, Jamrozy L, Glogowska J, Missol-Kolka E, Szala S . Electrotransfer of gene encoding endostatin into normal and neoplastic mouse tissues: Inhibition of primary tumor growth and metastatic spread. Cancer Gene Ther 2002; 9: 771–777.
Krieg AM, Yi AK, Matson S, Waldschmidt TJ, Bishop GA, Teasdale R et al. CpG motifs in bacterial DNA trigger direct B-cell activation. Nature 1995; 374: 546–549.
Lipford GB, Sparwasser T, Bauer M, Zimmermann S, Koch ES, Heeg K et al. Immunostimulatory DNA: sequence-dependent production of potentially harmful or useful cytokines. Eur J Immunol 1997; 27: 3420–3426.
Todorovic V, Sersa G, Mlakar V, Glavac D, Cemazar M . Assessment of the tumourigenic and metastatic properties of SK-MEL28 melanoma cells surviving electrochemotherapy with bleomycin. Radiol Oncol 2012; 46: 32–45.
Bauer S, Kirschning CJ, Hacker H, Redecke V, Hausmann S, Akira S et al. Human TLR9 confers responsiveness to bacterial DNA via species-specific CpG motif recognition. Proc Natl Acad Sci USA 2001; 98: 9237–9242.
Rols MP, Teissie J . Electropermeabilization of mammalian cells to macromolecules: control by pulse duration. Biophys J 1998; 75: 1415–1423.
Fritz V, Fajas L . Metabolism and proliferation share common regulatory pathways in cancer cells. Oncogene 2010; 29: 4369–4377.
Heller LC, Cruz YL, Ferraro B, Yang H, Heller R . Plasmid injection and application of electric pulses alter endogenous mRNA and protein expression in B16.F10 mouse melanomas. Cancer Gene Ther 2010; 17: 864–871.
Henshaw J, Mossop B, Yuan F . Relaxin treatment of solid tumors: effects on electric field-mediated gene delivery. Mol.Cancer Ther 2008; 7: 2566–2573.
Henshaw JW, Yuan F . Field distribution and DNA transport in solid tumors during electric field-mediated gene delivery. J Pharm.Sci 2008; 97: 691–711.
Hemmi H, Takeuchi O, Kawai T, Kaisho T, Sato S, Sanjo H et al. A Toll-like receptor recognizes bacterial DNA. Nature 2000; 408: 740–745.
Krieg AM . Development of TLR9 agonists for cancer therapy. J Clin Invest 2007; 117: 1184–1194.
Vollmer J, Krieg AM . Immunotherapeutic applications of CpG oligodeoxynucleotide TLR9 agonists. Adv Drug Deliv Rev 2009; 61: 195–204.
Sharma S, Karakousis CP, Takita H, Shin K, Brooks SP . Intra-tumoral injection of CpG results in the inhibition of tumor growth in murine Colon-26 and B-16 tumors. Biotechnol Lett 2003; 25: 149–153.
Kunikata N, Sano K, Honda M, Ishii K, Matsunaga J, Okuyama R et al. Peritumoral CpG oligodeoxynucleotide treatment inhibits tumor growth and metastasis of B16F10 melanoma cells. J Invest Dermatol 2004; 123: 395–402.
Ballas ZK, Krieg AM, Warren T, Rasmussen W, Davis HL, Waldschmidt M et al. Divergent therapeutic and immunologic effects of oligodeoxynucleotides with distinct CpG motifs. J Immunol 2001; 167: 4878–4886.
Krieg AM . Immune effects and mechanisms of action of CpG motifs. Vaccine 2000; 19: 618–622.
Yu YZ, Li N, Ma Y, Wang S, Yu WY, Sun ZW . Three types of human CpG motifs differentially modulate and augment immunogenicity of nonviral and viral replicon DNA vaccines as built-in adjuvants. Eur J Immunol 2013; 43: 228–239.
Belizário JE . Immunodeficient mouse models: an overview. Open Immunol J 2009; 2: 79–85.
Hornung V, Rothenfusser S, Britsch S, Krug A, Jahrsdorfer B, Giese T et al. Quantitative expression of toll-like receptor 1-10 mRNA in cellular subsets of human peripheral blood mononuclear cells and sensitivity to CpG oligodeoxynucleotides. J Immunol 2002; 168: 4531–4537.
Ballas ZK, Rasmussen WL, Krieg AM . Induction of NK activity in murine and human cells by CpG motifs in oligodeoxynucleotides and bacterial DNA. J Immunol 1996; 157: 1840–1845.
Yasuda K, Rutz M, Schlatter B, Metzger J, Luppa PB, Schmitz F et al. CpG motif-independent activation of TLR9 upon endosomal translocation of ‘natural’ phosphodiester DNA. Eur J Immunol 2006; 36: 431–436.
Yasuda K, Yu P, Kirschning CJ, Schlatter B, Schmitz F, Heit A et al. Endosomal translocation of vertebrate DNA activates dendritic cells via TLR9-dependent and -independent pathways. J Immunol 2005; 174: 6129–6136.
Lenert PS . Classification, mechanisms of action, and therapeutic applications of inhibitory oligonucleotides for Toll-like receptors (TLR) 7 and 9. Mediat Inflamm 2010; 2010: 986596.
Desmet CJ, Ishii KJ . Nucleic acid sensing at the interface between innate and adaptive immunity in vaccination. Nat Rev Immunol 2012; 12: 479–491.
Hornung V, Latz E . Intracellular DNA recognition. Nat Rev Immunol 2010; 10: 123–130.
Keating SE, Baran M, Bowie AG . Cytosolic DNA sensors regulating type I interferon induction. Trends Immunol 2011; 32: 574–581.
Wu M, Yuan F . Membrane binding of plasmid DNA and endocytic pathways are involved in electrotransfection of mammalian cells. PLoS One 2011; 6: e20923.
Rosazza C, Phez E, Escoffre JM, Cezanne L, Zumbusch A, Rols MP . Cholesterol implications in plasmid DNA electrotransfer: Evidence for the involvement of endocytotic pathways. Int J Pharm 2012; 423: 134–143.
Golzio M, Teissie J, Rols MP . Direct visualization at the single-cell level of electrically mediated gene delivery. Proc Natl Acad Sci USA 2002; 99: 1292–1297.
Vaughan EE, Dean DA . Intracellular trafficking of plasmids during transfection is mediated by microtubules. Mol Ther 2006; 13: 422–428.
Mann CJ, Anguela XM, Montane J, Obach M, Roca C, Ruzo A et al. Molecular signature of the immune and tissue response to non-coding plasmid DNA in skeletal muscle after electrotransfer. Gene Ther 2012; 19: 1177–1186.
Acknowledgements
The work in this study was supported by NIH 1 R21 CA106860 (LH), by the Slovene Research Agency P3-0003 and J3-4259 (MC and VT), and by a Bilateral scientific cooperation between Republic of Slovenia and United States of America BI-US/11-12-011.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Heller, L., Todorovic, V. & Cemazar, M. Electrotransfer of single-stranded or double-stranded DNA induces complete regression of palpable B16.F10 mouse melanomas. Cancer Gene Ther 20, 695–700 (2013). https://doi.org/10.1038/cgt.2013.71
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2013.71
Keywords
This article is cited by
-
In vitro exploration of a myeloid-derived suppressor cell line as vehicle for cancer gene therapy
Cancer Gene Therapy (2017)
-
Cytosolic DNA Sensor Upregulation Accompanies DNA Electrotransfer in B16.F10 Melanoma Cells
Molecular Therapy - Nucleic Acids (2016)
-
Bystander Effect Induced by Electroporation is Possibly Mediated by Microvesicles and Dependent on Pulse Amplitude, Repetition Frequency and Cell Type
The Journal of Membrane Biology (2016)
-
Endoglin (CD105) Silencing Mediated by shRNA Under the Control of Endothelin-1 Promoter for Targeted Gene Therapy of Melanoma
Molecular Therapy - Nucleic Acids (2015)
-
Mcam Silencing With RNA Interference Using Magnetofection has Antitumor Effect in Murine Melanoma
Molecular Therapy - Nucleic Acids (2014)