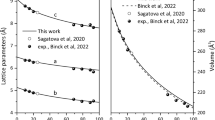
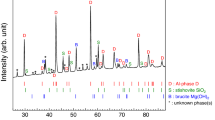
Abstract
Although aluminium is the fifth most abundant element in the Earth's mantle, its effect on the physical properties of perovskite, the main mineral phase in the lower mantle, has largely been ignored. It is becoming clear, however, that many properties of MgSiO3 perovskites are remarkably sensitive to small amounts of aluminium1,2,3,4. In particular, perovskite with only 5 wt% Al2O3 has a bulk modulus 10% lower than that of the pure magnesian end-member12. The increased compressibility may be due to the high concentrations of oxygen vacancies required to balance the charge of the aluminium5; if so, this would have important consequences for the mantle, as aluminous perovskites could be weaker, have lower seismic velocities and be hosts for water. To test whether oxygen vacancies exist in aluminous perovskites, I have calculated the compressibility of end-member defect-bearing perovskites using ab initio methods. The results show that perovskites with oxygen vacancies do have significantly greater compressibilities than those without such vacancies. But the results also suggest that oxygen vacancies become unfavourable at high pressures, in which case only the physical properties of the shallow lower mantle would be affected by aluminium—with the deeper mantle retaining properties similar to those of aluminium-free perovskite.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Xu, Y. S., McCammon, C. & Poe, B. T. The effect of alumina on the electrical conductivity of silicate perovskite. Science 282, 922 –924 (1998).
Zhang, J. & Weidner, D. J. Thermal equation of state of aluminium-enriched silicate perovskite. Science 284 , 782–784 (1999).
McCammon, C. A. Perovskite as a possible sink for ferric iron in the lower mantle. Nature 387, 694–696 ( 1997).
Wood, B. J. & Rubie, D. C. The effect of alumina on phase transformations at the 660-kilometer discontinuity from Fe–Mg partitioning experiments. Science 273, 1522– 1524 (1996).
Navrotsky, A. A lesson from ceramics. Science 284, 1788 –1789 (1999).
Richmond, N. C. & Brodholt, J. P. Calculated role of aluminum in the incorporation of ferric iron into magnesium silicate perovskite. Am. Miner. 83, 947– 951 (1998).
Andrault, D., Neuville, D. R., Flamk, A. M. & Wang, Y. Cation sites in Al-rich MgSiO3 perovskites. Am. Miner. 83, 1045–1053 ( 1998).
Ito, E., Kubo, A., Katsura, M., Akaogi, M. & Fujita, T. High-pressure transformation of pyrope (Mg3Al 2Si3O12) in a sintered diamond cubic anvil assembly. Geophys. Res. Lett. 25, 821– 824 (1998).
McCammon, C., Langenhorst, F. & Seifert, F. How well do we know the properties of lower mantle perovskites? Eos 80 F742 (1999 ).
Smyth, D. M., Chang, E. K. & Liu, D. H. Travels through perovskite space. Phase Transit. 58, 57–73 ( 1996).
Kesson, S. E., Gerald, J. D. F., Shelley, J. M. G. & Withers, R. L. Phase-relations, structure and crystal-chemistry of some aluminous silicate perovskites. Earth. Planet. Sci. Lett. 134, 187–201 (1995).
McCammon, C. A. The crystal chemistry of ferric iron in Fe0.05Mg0.95SiO 3 perovskite as determined by Mössbauer spectroscopy in the temperature range 80–293 K. Phys. Chem. Miner. 25, 292–300 (1998).
Fei, Y. Effects of temperature and composition on the bulk-modulus of (Mg,Fe)O. Am. Miner. 84, 272–276 ( 1999).
CASTEP version 3.9 (Molecular Simulations Inc., San Diego, 1999).
Acknowledgements
I thank C. McCammon for comments on this manuscript, N. Ross for her help, and D. Dobson, A. Oganov, L. Vocadlo, I. Wood and D. Price for discussions. I also thank the Royal Society for a University Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brodholt, J. Pressure-induced changes in the compression mechanism of aluminous perovskite in the Earth's mantle. Nature 407, 620–622 (2000). https://doi.org/10.1038/35036565
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/35036565
This article is cited by
-
Variation in bridgmanite grain size accounts for the mid-mantle viscosity jump
Nature (2023)
-
Pressure-induced tuning of lattice distortion in a high-entropy oxide
Communications Chemistry (2019)
-
Sound velocity of CaSiO3 perovskite suggests the presence of basaltic crust in the Earth’s lower mantle
Nature (2019)
-
The elastic solid solution model for minerals at high pressures and temperatures
Contributions to Mineralogy and Petrology (2018)
-
First-principles calculations of elasticity of minerals at high temperature and pressure
Science China Earth Sciences (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.