Abstract
Background:
Constipation and fecal incontinence are considerable problems for most individuals with spinal cord injury (SCI). Neurogenic bowel symptoms are caused by several factors including abnormal rectal wall properties. Stimulation of the dorsal genital nerve (DGN) can inhibit bladder contractions and because of common innervation inhibitory effects are anticipated in the rectum too. Therefore, DNG could have a future role in the treatment of neurogenic fecal incontinence.
Aim:
To study the effect of acute DGN stimulation on the rectal cross sectional area (CSA) in SCI patients.
Methods:
Seven patients with complete supraconal SCI (median age 50 years) were included. Stimulation was applied via plaster-electrodes using an amplitude of twice the genito–anal reflex threshold (pulse width: 200 μs; pulse rate: 20 Hz). A pressure controlled phasic (10, 20 and 30 cmH2O) rectal distension protocol was repeated four times with subjects randomized to stimulation during 1st and 3rd distension series or 2nd and 4th distension series. The rectal CSA and pressure were measured using impedance planimetry and manometry.
Results:
All patients completed the investigation. Median stimulation amplitude was 51 mA (range 30–64). CSA was smaller during stimulation and differences reached statistical significance at distension pressures of 20 cmH2O (average decrease 9%; P=0.02) and 30 cmH2O (average decrease 4%; P=0.03) above resting rectal pressure. Accordingly, rectal pressure-CSA relation was significantly reduced during stimulation at 20 (P=0.03) and 30 cmH2O distension (P=0.02).
Conclusion:
DGN Stimulation in patients with supraconal SCI results in an acute decrease of rectal CSA and the rectal pressure-CSA relation.
Similar content being viewed by others
Introduction
Most subjects with spinal cord injury (SCI) have constipation and fecal incontinence, often resulting in restricted social activities and impaired quality of life.1 Symptoms may be caused by abnormal rectal compliance and contractility, reduced anorectal sensibility, lack of external anal sphincter control and abnormal colorectal motility. The severity of neurogenic bowel dysfunction mainly depends on the completeness and level of injury, but time since injury is important too.2, 3 Most authors have found that rectal compliance is reduced in patients with supraconal SCI and data suggest that it is increased in those with conal or cauda equina lesions.4, 5 One group has found increased rectal compliance in patients with conal or cauda equina lesions.6
Neurogenic bowel dysfunction is usually treated conservatively with oral laxatives, suppositories and digital anorectal stimulation. Further treatment includes transanal irrigation, antegrade irrigation through an appendicostomy, colostomy or electrical stimulation, with the Brindley anterior root stimulator for assisted defecation.7, 8
Treatment is often unsatisfactory and new modalities should be explored. Stimulation of the dorsal genital nerve (DGN) can suppress vesical detrusor contractions and increase bladder capacity in patients with supraconal SCI.9 Also, a pilot study has indicated that DGN stimulation can increase rectal compliance in SCI patients.10 If data can be reproduced with other methods, DGN may have a future role in alleviating bowel symptoms in individuals with supraconal SCI.
The aim of the present study was to investigate whether DGN stimulation has an acute effect on the rectal cross sectional area (CSA) in patients with supraconal SCI.
Subjects and methods
Subjects
Seven subjects with supraconal SCI were included (one female, median age 50 years; range: 39–67 years), median time since injury being 19 years (range: 12–33 years). The lesion was motor and sensory complete in all the patients. Median neurogenic bowel dysfunction score was 14 (range: 5–19).11 Further demographics are given in Table 1. The study was performed in accordance with the Helsinki Declaration II and was approved by the local ethical committee (M-20090145). All participants gave their fully informed written consent.
Stimulation
Stimulation was performed using a constant current stimulator (Digitimer model DS7A, Digitme Ltd., Welwyn garden city, UK) with the frequency controlled by a waveform generator (Hewlet-Packard model 33120A, Palo Alto, CA, USA). Square pulses with a pulse width of 200 μs and a frequency of 20 Hz were used. The amplitude was set at two times the threshold of the genito-anal reflex. One electrode (dimension: 10 × 20 mm2, Neuroline 700, Ambu, Ballerup, Denmark) was placed at the base of the penis or on the clitoris as cathode, and a second electrode (diameter: 32 mm, PALS Platinium, Axelgaard, Lystrup, Denmark) was placed 2–3 cm lateral to the base of the penis or labia major. Precautions were taken to ensure good contact between skin and the electrodes. The genito–anal reflex threshold was identified visually during slowly stepwise increase of the amplitude before the investigation started.
Impedance planimetry
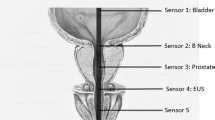
Rectal impedance planimetry allows simultaneous monitoring of rectal CSA and rectal pressure. The method avoids most sources of error associated with volume based methods.12 At a constant current I, the potential difference (ΔV) between two detection electrodes and the CSA are proportionally related (CSA=I d σ−1 ΔV −1). The electrodes were 3 mm (d) apart and contained in a fluid with the conductivity σ. The rectal probe, used in the present study, had two excitation electrodes (60 mm apart), providing a sinusoidal current of 0.1 mA at 10 kHz, and a pair of detection electrodes (Figure 1). The rectal CSA was measured ∼60 mm above the anal verge. The electrodes were within a non-compliant flaccid bag (diameter: 90 mm, length: 90 mm, maximum volume: ≈570 ml), which was filled with 0.9% saline at 37 °C. The pressure within the bag was controlled by elevation of an open water container. Before measurements a multipoint calibration was done using circular plastic tubes with an inner diameter in the range from 283 mm2 to 4322 mm2. Intraluminal rectal pressure and anal pressure were measured with pressure transducers (Baxter, Deerfield, IL, USA) connected to perfused catheters within the bag and a within a balloon placed in the anal canal, respectively. The pressure transducers were calibrated using 0 cmH2O and 100 cmH2O as minimum and maximum. All signals were sampled at 10 Hz and data were visualized and stored using custom made software (Openlab, Gatehouse, Noerresundby, Denmark). Before placement of the probe, resting rectal pressure was measured using a water-perfused catheter placed in the rectum. During the investigation, the patient was in the left lateral position and was not allowed to talk. The equipment for impedance planimetry has been described previously.13
Schematic illustration of the system for impedance planimetry. An alternating current is generated by the impedance planimetry box and lead to the saline within the bag through the excitation electrodes (E). The potential difference and, thereby, the rectal CSA is determined between the detection electrodes (D). Anal pressure (PA) and rectal pressure (PR) are simultaneously registered. The sampling frequency of all data is 10 Hz. A full color version of this figure is available at the Spinal Cord journal online.
Distension protocol
A pressure controlled phasic distension protocol with a total length of 96 min was executed (Figure 2). Before initiation of the phasic distensions, a distension pressure of 10 cmH2O above resting rectal pressure, lasting 12 min, was applied to condition the rectal wall. This was followed by 4 min distension at resting rectal pressure. Hereafter, distensions at three pressure levels (10, 20 and 30 cmH2O) were done. Each of the distensions lasted for 4 min and was separated by 4 min with the pressure at resting rectal pressure. This distension sequence was repeated four times, with DGN stimulation during 1st and 3rd distension series or 2nd and 4th distension series as randomized. Between the distensions, no DGN stimulation was applied.
An example of a recording (patient no. 7). Four distension series with distension 10, 20 and 30 cmH2O above resting rectal pressure are shown. During the first 20 cmH2O distension, CSA increased suddenly in the end so therefore mean CSA was calculated during the steady state period before the last minute. DGN stimulation was done during the first and the third series. A full color version of this figure is available at the Spinal Cord journal online.
Data analysis
The CSA and rectal pressure were determined when steady state was present at each pressure level. Steady state CSA, calculated as the mean CSA for a 1-min period, was assumed when the difference in mean CSA, for two 10 s periods one minute apart, was less than 10% (Figure 3). Data that did not meet this requirement were discarded. For each distension pressure, the mean CSA from two distensions during stimulation was compared with the mean CSA from two distensions without stimulation. The rectal pressure-CSA relation (CSA/PR) was calculated for each pressure level. The circumferential wall tension was calculated using Laplaces’ law T=Δp r, where T is the circumferential wall tension, r is the radius of the bag and Δp is the transmural pressure calculated as the difference between resting rectal pressure and rectal pressure during distension.
An example of a stimulated distension. A pressure controlled (PR) rectal distension is performed. When distension is started the stimulation is turned on, the anal sphincter contracts, which is seen as an increase in anal pressure (PA) (arrow). The rectal CSA increases and begins to stabilize after ∼2 min. A full color version of this figure is available at the Spinal Cord journal online.
Statistics
Numerical data are given as medians with ranges. Statistical comparisons were made using Wilcoxon's test for non-parametric comparison of paired measurements.
Results
All patients tolerated the investigation well. Three patients had lesions above Th6 and none of them experienced symptoms of autonomic dysreflexia during electrical stimulation. The median resting rectal pressure was 9.5 cmH2O (range: 8–16 cmH2O). Electrical stimulation above the reflex threshold could be seen as a brief (1–3 s) increase in anal pressure (Figure 3). The median stimulation amplitude was 51 mA (range: 30–64 mA). Filling of the bag resulted in an increase in rectal CSA. Most of the increase occurred within the first 30 s, and thereafter, the rectal CSA became stable, with changes of less than 10% during the fourth minute in all of the patients. Data from the 10 cmH2O distensions were discarded as they were not reproducible and reliable.
The median CSA was smaller with than without stimulation in all seven patients at 20 cmH2O distension pressure (P=0.02), and in six of seven patients at 30 cmH2O distension pressure (P=0.03) (Figure 4, Table 2). The median decrease in rectal CSA was 9% (7 cm2) at 20 cmH2O distension pressures and 4% (1 cm2) at 30 cmH2O at distension pressures. The rectal pressure-CSA relation was also significantly smaller during stimulation at 20 cmH2O (medians 1.0 cm2 per cmH2O vs 1.1 cm2 per cmH2O) (P=0.03)) and 30 cmH2O distension (medians 0.9 cm2 per cmH2O vs 0.9 cm2 per cmH2O) (P=0.02) (Table 3). The rectal wall tension was unchanged during stimulation (Table 3).
Discussion
This study shows that the rectal CSA is reduced during acute DGN stimulation in subjects with complete supraconal SCI.
The stimulation parameters were chosen based on experience with DGN stimulation to achieve inhibition of bladder contractions. A pulse width of 200 μs, a pulse rate of 20 Hz and an amplitude of at least twice the genito–anal reflex threshold have been used.9
It has been demonstrated that the configuration of the distension profile (that is, phasic, ramp or staircase) has only little effect on distensibility.14 Furthermore, a randomized stimulation protocol was used to avoid bias from a potential carry over effect from stimulation and relaxation as a result of repeated distensions.
The relationship between pressure and CSA can be described by a first order system. The continuous decrease in CSA change per time unit inferred that longer distensions would produce more stable CSAs. This had to be balanced against the potential impairment of mucosa blood flow during prolonged distension and efforts to minimize the discomfort of the patients. Distensions lasting 4 min were chosen as a safe compromise. Preferably, calculation of mean CSA was done for the last minute of each distension. However, during some distensions, changes in CSA >10% were seen during the last minute. If a steady state period was present before the last minute, this was used to calculate the mean CSA.
Previously, we have used impedance planimetry for description of rectal CSA in SCI patients with supraconal SCI and reported a median rectal CSA of 11 cm2 during distension at 10 cmH2O and 18 cm2 during distension at 30 cmH2O.4 Larger CSAs were measured in the present study. The patients from the two studies are, however, not directly comparable as in the previous study, patients were investigated just after injury and again 1 year later, while the median time since injury was 19 years in the present study. It has been shown, that constipation becomes more severe with time since injury and it is likely that the rectal wall properties change too.3
During rectal distensions at 20 and 30 cmH2O, the median pressure-CSA relation was smaller during stimulation compared with the control distensions. Our findings are in contrast with data presented by Chung et al.10 who found increased rectal compliance during acute DGN stimulation. They also used a stimulation amplitude of twice the reflex threshold, but rectal compliance was measured using a barostat (Distender Series II, G&J Electronics, Toronto, ON, Canada). Furthermore, they tested different stimulation frequencies (0.2, 2 and 20 Hz) and rectal compliance was larger during stimulation at 20 Hz compared with stimulation at a lower frequency. Chung et al.10 described a maximum increase of rectal compliance of 50% (at a rectal volume of 200 ml) during stimulation at 20 Hz. The maximum rectal compliance was ∼7 ml per cmH2O without stimulation, and 12 ml per cmH2O during stimulation. In that study, all the patients had complete supraconal lesions, but the duration of the injury was not mentioned. Rectal compliance both during and without stimulation were within the range of normal rectal compliance reported in the literature, which ranges from 4.5 ml per cmH2O–17 ml per cmH2O.15, 16 This wide range of measured rectal compliance warrants cautiousness when comparing data between different centers.
Traditionally, rectal compliance is studied with pressure-volume based methods using balloons (that is, barostat). The effect of rhizotomy and the response to feeding have been investigated in SCI patients.5, 17 There are, however, some methodological problems with these techniques.12 Impedance planimetry determines rectal CSA, thereby avoiding some of the inherent sources of error with pressure-volume measurements.18 The impedance planimetry probe used for this experiment was validated in vitro and accuracy was fair with a mean error of 7.3% (range: 0–14%). No low pass filter was included in the signal conditioning system (no anti-aliasing). However, it is unlikely that this had any influence on the results. At low distension pressures (10 cmH2O) the quality of rectal CSA measurements was not reliable. This could be caused by folding of the bag in irregular shape or eccentric position of the probe in the rectum. In another study, ultrasound was used to confirm the correct positioning of the probe during distension.19
Various implanted devices applying electrical stimulation have been used for treating neurogenic fecal incontinence. The use of Interstim Therapy has been investigated by Schurch et al.,20 who performed a test stimulation in three SCI patients with complete lesions. Both an early latency reflex corresponding to the genito–anal reflex and a late latency reflex, with high variability in latency were found indicating a polysegmental response. In none of the patients did the test stimulation reduce neurogenic incontinence suggesting that spino-bulbo-spinal pathways are necessary for sacral neuromodulation to be effective.
A previous study on the bladder showed inhibitory effects from DGN stimulation, including suppression of bladder contractions, higher bladder capacity and lower storage pressure.9 Though, we did not investigate phasic rectal contractions similar inhibitory effects could not be shown in this study. A fundamental difference between the bladder and the rectum is that the latter is modulated by the enteric nervous system, which could explain why results from stimulation of the bladder are not directly applicable to the bowel. It was hypothesized that DGN reduces neurogenic fecal incontinence by reducing rectal tone and contractility. This is not supported by the present study. Even though a reduction in CSA during stimulation was seen in all patients, the changes were relatively small. If this will have clinical implications remain to be studied. An alternative mode of action could be that DGN, by increasing rectal motor activity improved rectal emptying at defecation thereby reducing fecal incontinence. Larger studies of the effects of DGN stimulation are needed and it is possible that chronic effects may differ from those found in acute experiments.
In conclusion and in contrary to our hypothesis, it was shown that acute DGN stimulation in subjects with supraconal SCI results in reduced rectal compliance CSA.
Data Archiving
There was no data to deposit.
References
Glickman S, Kamm MA . Bowel dysfunction in spinal-cord-injury patients. Lancet 1996; 347: 1651–1653.
Liu CW, Huang CC, Chen CH, Yang YH, Chen TW, Huang MH . Prediction of severe neurogenic bowel dysfunction in persons with spinal cord injury. Spinal Cord 2010; 48: 554–559.
Faaborg PM, Christensen P, Finnerup N, Laurberg S, Krogh K . The pattern of colorectal dysfunction changes with time since spinal cord injury. Spinal Cord 2008; 46: 234–238.
Krogh K, Mosdal C, Gregersen H, Laurberg S . Rectal wall properties in patients with acute and chronic spinal cord lesions. Dis Colon Rectum 2002; 45: 641–649.
Sun WM, MacDonagh R, Forster D, Thomas DG, Smallwood R, Read NW . Anorectal function in patients with complete spinal transection before and after sacral posterior rhizotomy. Gastroenterology 1995; 108: 990–998.
Storrie JB, Harding S, Raeburn AJ, Trivedi PM, Preziosi G, Emmanuel AV . Medium-term outcome with transanal irrigation for neurogenic bowel dysfunction is related to rectal compliance. Gut 2009; 58: A87.
Christensen P, Bazzocchi G, Coggrave M, Abel R, Hultling C, Krogh K et al. A randomized, controlled trial of transanal irrigation versus conservative bowel management in spinal cord-injured patients. Gastroenterology 2006; 131: 738–747.
Furlan JC, Urbach DR, Fehlings MG . Optimal treatment for severe neurogenic bowel dysfunction after chronic spinal cord injury: a decision analysis. Br J Surg 2007; 94: 1139–1150.
Hansen J, Media S, Nohr M, Biering-Sorensen F, Sinkjaer T, Rijkhoff NJ . Treatment of neurogenic detrusor overactivity in spinal cord injured patients by conditional electrical stimulation. J Urol 2005; 173: 2035–2039.
Chung EAL, Woodhouse JB, Balasubramaniam V, Emmanuel AV, Craggs MD . Does sacral afferent stimulation influence bowel compliance? Neurourol Urodyn 2005; 24: 498–499.
Krogh K, Christensen P, Sabroe S, Laurberg S . Neurogenic bowel dysfunction score. Spinal Cord 2006; 44: 625–631.
Madoff RD, Orrom WJ, Rothenberger DA, Goldberg SM . Rectal compliance: a critical reappraisal. Int J Colorectal Dis 1990; 5: 37–40.
Gregersen H, Djurhuus JC . Impedance planimetry: a new approach to biomechanical intestinal wall properties. Dig Dis 1991; 9: 332–340.
Hammer HF, Phillips SF, Camilleri M, Hanson RB . Rectal tone, distensibility, and perception: reproducibility and response to different distensions. Am J Physiol 1998; 274: G584–G590.
Oettle GJ, Heaton KW . ‘Rectal dissatisfaction’ in the irritable bowel syndrome. A manometric and radiological study. Int J Colorectal Dis 1986; 1: 183–185.
Sorensen M, Rasmussen OO, Tetzschner T, Christiansen J . Physiological variation in rectal compliance. Br J Surg 1992; 79: 1106–1108.
Suttor VP, Ng C, Rutkowski S, Hansen RD, Kellow JE, Malcolm A . Colorectal responses to distension and feeding in patients with spinal cord injury. Am J Physiol Gastrointest Liver Physiol 2009; 296: G1344–G1349.
Krogh K, Ryhammer AM, Lundby L, Gregersen H, Laurberg TS . Comparison of methods used for measurement of rectal compliance. Dis Colon Rectum 2001; 44: 199–206.
Andersen IS, Michelsen HB, Krogh K, Buntzen S, Laurberg S . Impedance Planimetric Description of Normal Rectoanal Motility in Humans. Dis Colon Rectum 2007; 50: 1840–1848.
Schurch B, Reilly I, Reitz A, Curt A . Electrophysiological recordings during the peripheral nerve evaluation (PNE) test in complete spinal cord injury patients. World J Urol 2003; 20: 319–322.
Acknowledgements
This work was supported by the Danish National Advanced Technology Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Worsøe, J., Fynne, L., Laurberg, S. et al. Acute effect of electrical stimulation of the dorsal genital nerve on rectal capacity in patients with spinal cord injury. Spinal Cord 50, 462–466 (2012). https://doi.org/10.1038/sc.2011.159
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2011.159