Abstract
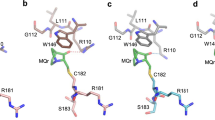
Pirh2 (p53-induced RING-H2 domain protein; also known as Rchy1) is an E3 ubiquitin ligase involved in a negative-feedback loop with p53. Using NMR spectroscopy, we show that Pirh2 is a unique cysteine-rich protein comprising three modular domains. The protein binds nine zinc ions using a variety of zinc coordination schemes, including a RING domain and a left-handed β-spiral in which three zinc ions align three consecutive small β-sheets in an interleaved fashion. We show that Pirh2-p53 interaction is dependent on the C-terminal zinc binding module of Pirh2, which binds to the tetramerization domain of p53. As a result, Pirh2 preferentially ubiquitylates the tetrameric form of p53 in vitro and in vivo, suggesting that Pirh2 regulates protein turnover of the transcriptionally active form of p53.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Vousden, K.H. & Prives, C. P53 and prognosis: new insights and further complexity. Cell 120, 7–10 (2005).
Levine, A.J. p53, the cellular gatekeeper for growth and division. Cell 88, 323–331 (1997).
Michael, D. & Oren, M. The p53-Mdm2 module and the ubiquitin system. Semin. Cancer Biol. 13, 49–58 (2003).
Honda, R., Tanaka, H. & Yasuda, H. Oncoprotein MDM2 is a ubiquitin ligase E3 for tumor suppressor p53. FEBS Lett. 420, 25–27 (1997).
Chene, P. Inhibiting the p53–MDM2 interaction: an important target for cancer therapy. Nat. Rev. Cancer 3, 102–109 (2003).
Leng, R.P. et al. Pirh2, a p53-induced ubiquitin-protein ligase, promotes p53 degradation. Cell 112, 779–791 (2003).
Dornan, D. et al. The ubiquitin ligase COP1 is a critical negative regulator of p53. Nature 429, 86–92 (2004).
Chen, D. et al. ARF-BP1/Mule is a critical mediator of the ARF tumor suppressor. Cell 121, 1071–1083 (2005).
Rajendra, R. et al. Topors functions as an E3 ubiquitin ligase with specific E2 enzymes and ubiquitinates p53. J. Biol. Chem. 279, 36440–36444 (2004).
Duan, W. et al. Expression of Pirh2, a newly identified ubiquitin protein ligase, in lung cancer. J. Natl. Cancer Inst. 96, 1718–1721 (2004).
Holm, L. & Sander, C. Dali: a network tool for protein structure comparison. Trends Biochem. Sci. 20, 478–480 (1995).
Borden, K.L. & Freemont, P.S. The RING finger domain: a recent example of a sequence-structure family. Curr. Opin. Struct. Biol. 6, 395–401 (1996).
Fang, S. & Weissman, A.M. A field guide to ubiquitylation. Cell. Mol. Life Sci. 61, 1546–1561 (2004).
Laine, A. et al. Regulation of p53 localization and activity by Ubc13. Mol. Cell. Biol. 26, 8901–8913 (2006).
Davison, T.S. et al. Structure and functionality of a designed p53 dimer. J. Mol. Biol. 307, 605–617 (2001).
Friedler, A., Veprintsev, D.B., Rutherford, T., von Glos, K.I. & Fersht, A.R. Binding of Rad51 and other peptide sequences to a promiscuous, highly electrostatic binding site in p53. J. Biol. Chem. 280, 8051–8059 (2005).
Joazeiro, C.A. & Weissman, A.M. RING finger proteins: mediators of ubiquitin ligase activity. Cell 102, 549–552 (2000).
Eletr, Z.M. & Kuhlman, B. Sequence determinants of E2–E6AP binding affinity and specificity. J. Mol. Biol. 369, 419–428 (2007).
Dominguez, C. et al. Structural model of the UbcH5B/CNOT4 complex revealed by combining NMR, mutagenesis, and docking approaches. Structure 12, 633–644 (2004).
Zheng, N., Wang, P., Jeffrey, P.D. & Pavletich, N.P. Structure of a c-Cbl-UbcH7 complex: RING domain function in ubiquitin-protein ligases. Cell 102, 533–539 (2000).
Ayed, A. et al. Latent and active p53 are identical in conformation. Nat. Struct. Biol. 8, 756–760 (2001).
Kay, L.E. NMR methods for the study of protein structure and dynamics. Biochem. Cell Biol. 75, 1–15 (1997).
Bax, A. et al. Measurement of homo- and heteronuclear J couplings from quantitative J correlation. Methods Enzymol. 239, 79–105 (1994).
Lemak, A., Steren, C.A., Arrowsmith, C.H. & Llinas, M. Sequence specific resonance assignment via Multicanonical Monte Carlo search using an ABACUS approach. J. Biomol. NMR 41, 29–41 (2008).
Cornilescu, G., Delaglio, F. & Bax, A. Protein backbone angle restraints from searching a database for chemical shift and sequence homology. J. Biomol. NMR 13, 289–302 (1999).
Alberts, I.L., Nadassy, K. & Wodak, S.J. Analysis of zinc binding sites in protein crystal structures. Protein Sci. 7, 1700–1716 (1998).
Guntert, P., Mumenthaler, C. & Wuthrich, K. Torsion angle dynamics for NMR structure calculation with the new program DYANA. J. Mol. Biol. 273, 283–298 (1997).
Grishaev, A. et al. ABACUS, a direct method for protein NMR structure computation via assembly of fragments. Proteins 61, 36–43 (2005).
Brunger, A.T. et al. Crystallography & NMR system: a new software suite for macromolecular structure determination. Acta Crystallogr. D Biol. Crystallogr. 54, 905–921 (1998).
Linge, J.P., Williams, M.A., Spronk, C.A., Bonvin, A.M. & Nilges, M. Refinement of protein structures in explicit solvent. Proteins 50, 496–506 (2003).
Koradi, R., Billeter, M. & Wuthrich, K. MOLMOL: a program for display and analysis of macromolecular structures. J. Mol. Graph. 14, 51–55 (1996).
Laskowski, R.A., Rullmann, J.A., MacArthur, M.W., Kaptein, R. & Thornton, J.M. AQUA and PROCHECK-NMR: programs for checking the quality of proteins structures solved by NMR. J. Biomol. NMR 8, 477–486 (1996).
Bhattacharya, A., Tejero, R. & Montelione, G.T. Evaluating protein structures determined by structural genomics consortia. Proteins 66, 778–795 (2007).
Banks, L., Matlashewski, G. & Crawford, L. Isolation of human-p53 specific monoclonal antibodies and their use in the studies of human p53 expression. Eur. J. Biochem. 159, 529–534 (1986).
Gouet, P., Courcelle, E., Stuart, D.I. & Metoz, F. ESPript: analysis of multiple sequence alignments in PostScript. Bioinformatics 15, 305–308 (1999).
Acknowledgements
We thank A. Ayed for helpful comments on the manuscript and members of the Arrowsmith lab for technical advice and discussion. This work was funded by the Canadian Cancer Society through grants from the National Cancer Institute of Canada, the Protein Structure Initiative of the National Institutes of Health (P50-GM62413-01) through the Northeast Structural Genomics Consortium, and the Canada Research Chairs program (to C.H.A.). Y.S. is supported by a fellowship from the Leukemia and Lymphoma Research Society of Canada.
Author information
Authors and Affiliations
Contributions
Y.S., R.C.L. and E.T. designed and conducted experiments; Y.S., A.L., B.W. and M.S. determined the structures; R.C.L., J.L., S.S. and M.K. collected NMR data; S.D. provided constructs and reagents; and Y.S. and C.H.A. wrote the paper with input from all other authors.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 1567 kb)
Rights and permissions
About this article
Cite this article
Sheng, Y., Laister, R., Lemak, A. et al. Molecular basis of Pirh2-mediated p53 ubiquitylation. Nat Struct Mol Biol 15, 1334–1342 (2008). https://doi.org/10.1038/nsmb.1521
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.1521
This article is cited by
-
The UAS thioredoxin-like domain of UBXN7 regulates E3 ubiquitin ligase activity of RNF111/Arkadia
BMC Biology (2023)
-
Recognition of an Ala-rich C-degron by the E3 ligase Pirh2
Nature Communications (2023)
-
Functional roles of E3 ubiquitin ligases in prostate cancer
Journal of Molecular Medicine (2022)
-
p53 regulation by ubiquitin and ubiquitin-like modifications
Genome Instability & Disease (2022)
-
Genome-wide analysis of RING-type E3 ligase family identifies potential candidates regulating high amylose starch biosynthesis in wheat (Triticum aestivum L.)
Scientific Reports (2021)