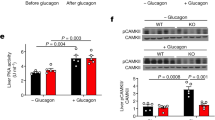
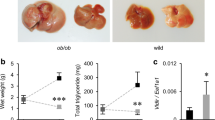
Abstract
Increased production of very low-density lipoprotein (VLDL) is a critical feature of the metabolic syndrome. Here we report that a selective increase in brain glucose lowered circulating triglycerides (TG) through the inhibition of TG-VLDL secretion by the liver. We found that the effect of glucose required its conversion to lactate, leading to activation of ATP-sensitive potassium channels and to decreased hepatic activity of stearoyl-CoA desaturase-1 (SCD1). SCD1 catalyzed the synthesis of oleyl-CoA from stearoyl-CoA. Curtailing the liver activity of SCD1 was sufficient to lower the hepatic levels of oleyl-CoA and to recapitulate the effects of central glucose administration on VLDL secretion. Notably, portal infusion of oleic acid restored hepatic oleyl-CoA to control levels and negated the effects of both central glucose and SCD1 deficiency on TG-VLDL secretion. These central effects of glucose (but not those of lactate) were rapidly lost in diet-induced obesity. These findings indicate that a defect in brain glucose sensing could play a critical role in the etiology of the metabolic syndrome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
18 April 2007
In the version of this article initially published on-line, it was not indicated that the first two authors (T.K.T. L. and R.G.-J.) contributed equally to this work. The error has been corrected in the HTML and PDF versions of the article.
Notes
*NOTE: Nat. Med. 13, 171-180 (2007); published online 4 February 2007; corrected after print 18 April 2007. In the version of this article initially published on-line, it was not indicated that the first two authors (T.K.T. L. and R.G.-J.) contributed equally to this work. The error has been corrected in the HTML and PDF versions of the article.
References
Reaven, G.M., Chen, Y.D., Jeppesen, J., Maheux, P. & Krauss, R.M. Insulin resistance and hyperinsulinemia in individuals with small, dense low density lipoprotein particles. J. Clin. Invest. 92, 141–146 (1993).
Ginsberg, H.N. New perspectives on atherogenesis: role of abnormal triglyceride-rich lipoprotein metabolism. Circulation 106, 2137–2142 (2002).
Horton, J.D., Goldstein, J.L. & Brown, M.S. SREBPs: activators of the complete program of cholesterol and fatty acid synthesis in the liver. J. Clin. Invest. 109, 1125–1131 (2002).
Reilly, M.P. & Rader, D.J. The metabolic syndrome: more than the sum of its parts? Circulation 108, 1546–1551 (2003).
Vikramadithyan, R.K. et al. Human aldose reductase expression accelerates diabetic atherosclerosis in transgenic mice. J. Clin. Invest. 115, 2434–2443 (2005).
Lin, J. et al. Hyperlipidemic effects of dietary saturated fats mediated through PGC-1beta coactivation of SREBP. Cell 120, 261–273 (2005).
Wolfrum, C., Asilmaz, E., Luca, E., Friedman, J.M. & Stoffel, M. Foxa2 regulates lipid metabolism and ketogenesis in the liver during fasting and in diabetes. Nature 432, 1027–1032 (2004).
Flier, J.S. Obesity wars: molecular progress confronts an expanding epidemic. Cell 116, 337–350 (2004).
Lewis, G.F., Uffelman, K.D., Szeto, L.W., Weller, B. & Steiner, G. Interaction between free fatty acids and insulin in the acute control of very low density lipoprotein production in humans. J. Clin. Invest. 95, 158–166 (1995).
Pan, M., Liang Js, J.S., Fisher, E.A. & Ginsberg, H.N. The late addition of core lipids to nascent apolipoprotein B100, resulting in the assembly and secretion of triglyceride-rich lipoproteins, is independent of both microsomal triglyceride transfer protein activity and new triglyceride synthesis. J. Biol. Chem. 277, 4413–4421 (2002).
Fisher, E.A. & Ginsberg, H.N. Complexity in the secretory pathway: the assembly and secretion of apolipoprotein B-containing lipoproteins. J. Biol. Chem. 277, 17377–17380 (2002).
Wu, X., Zhou, M., Huang, L.S., Wetterau, J. & Ginsberg, H.N. Demonstration of a physical interaction between microsomal triglyceride transfer protein and apolipoprotein B during the assembly of ApoB-containing lipoproteins. J. Biol. Chem. 271, 10277–10281 (1996).
Hollenbeck, C.B. Dietary fructose effects on lipoprotein metabolism and risk for coronary artery disease. Am. J. Clin. Nutr. 58, 800S–809S (1993).
Verschoor, L., Chen, Y.D., Reaven, E.P. & Reaven, G.M. Glucose and fructose feeding lead to alterations in structure and function of very low density lipoproteins. Horm. Metab. Res. 17, 285–288 (1985).
Taghibiglou, C. et al. Mechanisms of hepatic very low density lipoprotein overproduction in insulin resistance. Evidence for enhanced lipoprotein assembly, reduced intracellular ApoB degradation, and increased microsomal triglyceride transfer protein in a fructose-fed hamster model. J. Biol. Chem. 275, 8416–8425 (2000).
Friedman, J.M. Obesity in the new millennium. Nature 404, 632–634 (2000).
Schwartz, M.W. & Porte, D., Jr . Diabetes, obesity, and the brain. Science 307, 375–379 (2005).
Lam, T.K., Schwartz, G.J. & Rossetti, L. Hypothalamic sensing of fatty acids. Nat. Neurosci. 8, 579–584 (2005).
Howard, J.K. et al. Enhanced leptin sensitivity and attenuation of diet-induced obesity in mice with haploinsufficiency of Socs3. Nat. Med. 10, 734–738 (2004).
Morgan, K., Obici, S. & Rossetti, L. Hypothalamic responses to long-chain fatty acids are nutritionally regulated. J. Biol. Chem. 279, 31139–31148 (2004).
Davis, J.D., Wirtshafter, D., Asin, K.E. & Brief, D. Sustained intracerebroventricular infusion of brain fuels reduces body weight and food intake in rats. Science 212, 81–83 (1981).
Woods, S.C. & McKay, L.D. Intraventricular alloxan eliminates feeding elicited by 2-deoxyglucose. Science 202, 1209–1211 (1978).
Borg, M.A., Sherwin, R.S., Borg, W.P., Tamborlane, W.V. & Shulman, G.I. Local ventromedial hypothalamus glucose perfusion blocks counterregulation during systemic hypoglycemia in awake rats. J. Clin. Invest. 99, 361–365 (1997).
Levin, B.E., Routh, V.H., Kang, L., Sanders, N.M. & Dunn-Meynell,, A.A. Neuronal glucosensing: what do we know after 50 years? Diabetes 53, 2521–2528 (2004).
Lam, T.K., Gutierrez-Juarez, R., Pocai, A. & Rossetti, L. Regulation of blood glucose by hypothalamic pyruvate metabolism. Science 309, 943–947 (2005).
Pellerin, L. & Magistretti, P.J. Neuroscience. Let there be (NADH) light. Science 305, 50–52 (2004).
Pocai, A. et al. Hypothalamic KATP channels control hepatic glucose production. Nature 434, 1026–1031 (2005).
Obici, S., Zhang, B.B., Karkanias, G. & Rossetti, L. Hypothalamic insulin signaling is required for inhibition of glucose production. Nat. Med. 8, 1376–1382 (2002).
Lam, T.K. et al. Hypothalamic sensing of circulating fatty acids is required for glucose homeostasis. Nat. Med. 11, 320–327 (2005).
Spanswick, D., Smith, M.A., Groppi, V.E., Logan, S.D. & Ashford, M.L. Leptin inhibits hypothalamic neurons by activation of ATP-sensitive potassium channels. Nature 390, 521–525 (1997).
Spanswick, D., Smith, M.A., Mirshamsi, S., Routh, V.H. & Ashford, M.L. Insulin activates ATP-sensitive K+ channels in hypothalamic neurons of lean, but not obese rats. Nat. Neurosci. 3, 757–758 (2000).
Adeli, K., Taghibiglou, C., Van Iderstine, S.C. & Lewis, G.F. Mechanisms of hepatic very low-density lipoprotein overproduction in insulin resistance. Trends Cardiovasc. Med. 11, 170–176 (2001).
Carpentier, A. et al. Ameliorated hepatic insulin resistance is associated with normalization of microsomal triglyceride transfer protein expression and reduction in very low density lipoprotein assembly and secretion in the fructose-fed hamster. J. Biol. Chem. 277, 28795–28802 (2002).
Olofsson, S.O. & Boren, J. Apolipoprotein B: a clinically important apolipoprotein which assembles atherogenic lipoproteins and promotes the development of atherosclerosis. J. Intern. Med. 258, 395–410 (2005).
Qiu, W. et al. Hepatic PTP-1B expression regulates the assembly and secretion of apolipoprotein B-containing lipoproteins: evidence from protein tyrosine phosphatase-1B overexpression, knockout, and RNAi studies. Diabetes 53, 3057–3066 (2004).
Qiu, W. et al. Oleate-mediated stimulation of microsomal triglyceride transfer protein (MTP) gene promoter: implications for hepatic MTP overexpression in insulin resistance. Biochemistry 44, 3041–3049 (2005).
Jiang, G. et al. Prevention of obesity in mice by antisense oligonucleotide inhibitors of stearoyl-CoA desaturase-1. J. Clin. Invest. 115, 1030–1038 (2005).
Gutierrez-Juarez, R. et al. Critical role of stearoyl-CoA desaturase-1 (SCD1) in the onset of diet-induced hepatic insulin resistance. J. Clin. Invest. 116, 1686–1695 (2006).
Gill, J.M. et al. Hepatic production of VLDL1 but not VLDL2 is related to insulin resistance in normoglycaemic middle-aged subjects. Atherosclerosis 176, 49–56 (2004).
Attie, A.D. et al. Relationship between stearoyl-CoA desaturase activity and plasma triglycerides in human and mouse hypertriglyceridemia. J. Lipid Res. 43, 1899–1907 (2002).
Miyazaki, M., Kim, Y.C., Gray-Keller, M.P., Attie, A.D. & Ntambi, J.M. The biosynthesis of hepatic cholesterol esters and triglycerides is impaired in mice with a disruption of the gene for stearoyl-CoA desaturase 1. J. Biol. Chem. 275, 30132–30138 (2000).
Cohen, P. et al. Role for stearoyl-CoA desaturase-1 in leptin-mediated weight loss. Science 297, 240–243 (2002).
Asilmaz, E. et al. Site and mechanism of leptin action in a rodent form of congenital lipodystrophy. J. Clin. Invest. 113, 414–424 (2004).
Kalopissis, A.D., Griglio, S., Malewiak, M.I., Rozen, R. & Liepvre, X.L. Very-low-density-lipoprotein secretion by isolated hepatocytes of fat-fed rats. Biochem. J. 198, 373–377 (1981).
Francone, O.L. et al. Effect of a high-fat-diet on the incorporation of stored triacylglycerol into hepatic VLDL. Am. J. Physiol. 263, E615–E623 (1992).
Kraus, W.E. et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N. Engl. J. Med. 347, 1483–1492 (2002).
Obici, S. et al. Central melanocortin receptors regulate insulin action. J. Clin. Invest. 108, 1079–1085 (2001).
Muse, E.D. et al. Role of resistin in diet-induced hepatic insulin resistance. J. Clin. Invest. 114, 232–239 (2004).
Otvos, J.D., Jeyarajah, E.J., Bennett, D.W. & Krauss, R.M. Development of a proton nuclear magnetic resonance spectroscopic method for determining plasma lipoprotein concentrations and subspecies distributions from a single, rapid measurement. Clin. Chem. 38, 1632–1638 (1992).
Otvos, J.D., Jeyarajah, E.J. & Bennett, D.W. Quantification of plasma lipoproteins by proton nuclear magnetic resonance spectroscopy. Clin. Chem. 37, 377–386 (1991).
Acknowledgements
We thank C. Baveghems and B. Liu for technical assistance. This work was supported by grants from the US National Institutes of Health (DK45024, DK48321 and AG 21654 to L.R.; DK47208 to G.J.S.) and the Albert Einstein College of Medicine Diabetes Research and Training Center (DK 20541). T.K.T.L. was supported by a fellowship from the US National Institutes of Health (F32-DK072876) and is currently appointed as the John Kitson McIvor Endowed Chair in Diabetes Research at the University Health Network and University of Toronto.
Author information
Authors and Affiliations
Contributions
T.K.T.L and R.G.-J. conducted the experiments and data analyses, and wrote the manuscript; A.P. conducted the experiments; S.B. provided the SCD1 antisense oligodeoxynucleotide; P.T. performed the Apo100/48 and TG content determination in HPLC fractions; G.J.S. performed the selective hepatic branch vagotomy; L.R. supervised the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
A dose-response curve of hypothalamic and plasma glucose concentrations. (PDF 207 kb)
Supplementary Fig. 2
The rate of appearance of total (small + medium + large) VLDL particles was not different among groups. (PDF 230 kb)
Supplementary Fig. 3
ApoB100/48 secretion in 10h fasted rats. (PDF 372 kb)
Supplementary Fig. 4
Effect of ICV glucose on TG-VLDL secretion in 5h and 10h fasted rats. (PDF 229 kb)
Supplementary Fig. 5
Effect of HFD in male Wistar and Sprague Dawley rats. (PDF 246 kb)
Supplementary Table 1
Characteristics of the groups during the central glucose administration. (PDF 3 kb)
Supplementary Table 2
Characteristics of the groups during central lactate administration. (PDF 3 kb)
Supplementary Table 3
Diet Composition (PDF 2 kb)
Rights and permissions
About this article
Cite this article
Lam, T., Gutierrez-Juarez, R., Pocai, A. et al. Brain glucose metabolism controls the hepatic secretion of triglyceride-rich lipoproteins. Nat Med 13, 171–180 (2007). https://doi.org/10.1038/nm1540
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1540
This article is cited by
-
Antipsychotics impair regulation of glucose metabolism by central glucose
Molecular Psychiatry (2022)
-
Brain insulin signalling in metabolic homeostasis and disease
Nature Reviews Endocrinology (2021)
-
AMPK activator C24 inhibits hepatic lipogenesis and ameliorates dyslipidemia in HFHC diet-induced animal models
Acta Pharmacologica Sinica (2021)
-
Hypothalamic glucose-sensing mechanisms
Diabetologia (2021)
-
Brain leptin reduces liver lipids by increasing hepatic triglyceride secretion and lowering lipogenesis
Nature Communications (2019)