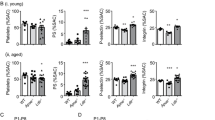
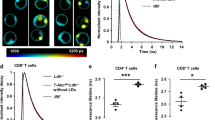
Abstract
Platelets have a key role in atherogenesis and its complications. Both hypercholesterolemia and increased platelet production promote atherothrombosis; however, a potential link between altered cholesterol homeostasis and platelet production has not been explored. Here we show that transplantation of bone marrow deficient in ABCG4, a transporter of unknown function, into Ldlr−/− mice resulted in thrombocytosis, accelerated thrombosis and atherosclerosis. Although not detected in atherosclerotic lesions, Abcg4 was highly expressed in bone marrow megakaryocyte progenitors (MkPs). Abcg4−/− MkPs had defective cholesterol efflux to high-density lipoprotein (HDL), increased cell surface expression of the thrombopoietin (TPO) receptor (c-MPL) and enhanced proliferation. These consequences of ABCG4 deficiency seemed to reflect disruption of negative feedback regulation of c-MPL signaling by the E3 ligase c-CBL and the cholesterol-sensing LYN kinase. HDL infusion reduced platelet counts in Ldlr−/− mice and in a mouse model of myeloproliferative neoplasm in an ABCG4-dependent fashion. HDL infusions may offer a new approach to reducing atherothrombotic events associated with increased platelet production.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Labarthe, D.R. & Dunbar, S.B. Global cardiovascular health promotion and disease prevention: 2011 and beyond. Circulation 125, 2667–2676 (2012).
Libby, P., Ridker, P.M. & Hansson, G.K. Progress and challenges in translating the biology of atherosclerosis. Nature 473, 317–325 (2011).
Koenen, R.R. et al. Disrupting functional interactions between platelet chemokines inhibits atherosclerosis in hyperlipidemic mice. Nat. Med. 15, 97–103 (2009).
Huo, Y. et al. Circulating activated platelets exacerbate atherosclerosis in mice deficient in apolipoprotein E. Nat. Med. 9, 61–67 (2003).
Coller, B.S. Historical perspective and future directions in platelet research. J. Thromb. Haemost. 9 (suppl. 1), 374–395 (2011).
Martin, J.F., Kristensen, S.D., Mathur, A., Grove, E.L. & Choudry, F.A. The causal role of megakaryocyte-platelet hyperactivity in acute coronary syndromes. Nat. Rev. Cardiol. 9, 658–670 (2012).
Trip, M.D., Cats, V.M., van Capelle, F.J. & Vreeken, J. Platelet hyperreactivity and prognosis in survivors of myocardial infarction. N. Engl. J. Med. 322, 1549–1554 (1990).
Hasselbalch, H.C. Perspectives on chronic inflammation in essential thrombocythemia, polycythemia vera, and myelofibrosis. Blood 3219–3225 (2012).
Tefferi, A. & Vainchenker, W. Myeloproliferative neoplasms: molecular pathophysiology, essential clinical understanding, and treatment strategies. J. Clin. Oncol. 29, 573–582 (2011).
Steinberg, D. The statins in preventive cardiology. N. Engl. J. Med. 359, 1426–1427 (2008).
Tall, A.R., Yvan-Charvet, L., Terasaka, N., Pagler, T. & Wang, N. HDL, ABC transporters, and cholesterol efflux: implications for the treatment of atherosclerosis. Cell Metab. 7, 365–375 (2008).
Pathansali, R., Smith, N. & Bath, P. Altered megakaryocyte-platelet haemostatic axis in hypercholesterolaemia. Platelets 12, 292–297 (2001).
Wang, N., Lan, D., Chen, W., Matsuura, F. & Tall, A.R. ATP-binding cassette transporters G1 and G4 mediate cellular cholesterol efflux to high-density lipoproteins. Proc. Natl. Acad. Sci. USA 101, 9774–9779 (2004).
Wang, N., Ranalletta, M., Matsuura, F., Peng, F. & Tall, A.R. LXR-induced redistribution of ABCG1 to plasma membrane in macrophages enhances cholesterol mass efflux to HDL. Arterioscler. Thromb. Vasc. Biol. 26, 1310–1316 (2006).
Annilo, T. et al. Human and mouse orthologs of a new ATP-binding cassette gene, ABCG4. Cytogenet. Cell Genet. 94, 196–201 (2001).
Bojanic, D.D. et al. Differential expression and function of ABCG1 and ABCG4 during development and aging. J. Lipid Res. 51, 169–181 (2010).
Ranalletta, M. et al. Decreased atherosclerosis in low-density lipoprotein receptor knockout mice transplanted with Abcg1−/− bone marrow. Arterioscler. Thromb. Vasc. Biol. 26, 2308–2315 (2006).
Meurs, I. et al. The effect of ABCG1 deficiency on atherosclerotic lesion development in LDL receptor knockout mice depends on the stage of atherogenesis. Atherosclerosis 221, 41–47 (2012).
Yvan-Charvet, L. et al. ATP-binding cassette transporters and HDL suppress hematopoietic stem cell proliferation. Science 328, 1689–1693 (2010).
Mazzone, A. et al. Increased expression of neutrophil and monocyte adhesion molecules in unstable coronary artery disease. Circulation 88, 358–363 (1993).
Mause, S.F., von Hundelshausen, P., Zernecke, A., Koenen, R.R. & Weber, C. Platelet microparticles: a transcellular delivery system for RANTES promoting monocyte recruitment on endothelium. Arterioscler. Thromb. Vasc. Biol. 25, 1512–1518 (2005).
Guthikonda, S. et al. Role of reticulated platelets and platelet size heterogeneity on platelet activity after dual antiplatelet therapy with aspirin and clopidogrel in patients with stable coronary artery disease. J. Am. Coll. Cardiol. 52, 743–749 (2008).
Lakkis, N. et al. Reticulated platelets in acute coronary syndrome: a marker of platelet activity. J. Am. Coll. Cardiol. 44, 2091–2093 (2004).
Stohlawetz, P. et al. Measurement of the levels of reticulated platelets after plateletpheresis to monitor activity of thrombopoiesis. Transfusion 38, 454–458 (1998).
Nofer, J.R. & van Eck, M. HDL scavenger receptor class B type I and platelet function. Curr. Opin. Lipidol. 22, 277–282 (2011).
Villmow, T., Kemkes-Matthes, B. & Matzdorff, A.C. Markers of platelet activation and platelet-leukocyte interaction in patients with myeloproliferative syndromes. Thromb. Res. 108, 139–145 (2002).
Pikman, Y. et al. MPLW515L is a novel somatic activating mutation in myelofibrosis with myeloid metaplasia. PLoS Med. 3, e270 (2006).
Frontelo, P. et al. Novel role for EKLF in megakaryocyte lineage commitment. Blood 110, 3871–3880 (2007).
Hitchcock, I.S., Chen, M.M., King, J.R. & Kaushansky, K. YRRL motifs in the cytoplasmic domain of the thrombopoietin receptor regulate receptor internalization and degradation. Blood 112, 2222–2231 (2008).
Tiedt, R. et al. Pronounced thrombocytosis in transgenic mice expressing reduced levels of Mpl in platelets and terminally differentiated megakaryocytes. Blood 113, 1768–1777 (2009).
Kelemen, E., Lehoczky, D., Jakab, K., Batai, A. & Vargha, P. Responses to single-dose thrombopoietin decrease with higher platelet counts in mice. Acta Haematol. 101, 41–45 (1999).
Brown, M.S. & Goldstein, J.L. Cholesterol feedback: from Schoenheimer's bottle to Scap's MELADL. J. Lipid Res. 50 (suppl.), S15–S27 (2009).
Lingwood, D. & Simons, K. Lipid rafts as a membrane-organizing principle. Science 327, 46–50 (2010).
Saur, S.J., Sangkhae, V., Geddis, A.E., Kaushansky, K. & Hitchcock, I.S. Ubiquitination and degradation of the thrombopoietin receptor c-Mpl. Blood 115, 1254–1263 (2010).
Nadeau, S. et al. Oncogenic signaling by leukemia-associated mutant Cbl proteins. Biochem. Anal. Biochem. S6 (2012).
Hunter, S., Burton, E.A., Wu, S.C. & Anderson, S.M. Fyn associates with Cbl and phosphorylates tyrosine 731 in Cbl, a binding site for phosphatidylinositol 3-kinase. J. Biol. Chem. 274, 2097–2106 (1999).
Blake, R.A. et al. SU6656, a selective src family kinase inhibitor, used to probe growth factor signaling. Mol. Cell Biol. 20, 9018–9027 (2000).
Lannutti, B.J., Shim, M.H., Blake, N., Reems, J.A. & Drachman, J.G. Identification and activation of Src family kinases in primary megakaryocytes. Exp. Hematol. 31, 1268–1274 (2003).
Oneyama, C. et al. Transforming potential of Src family kinases is limited by the cholesterol-enriched membrane microdomain. Mol. Cell Biol. 29, 6462–6472 (2009).
Lannutti, B.J., Minear, J., Blake, N. & Drachman, J.G. Increased megakaryocytopoiesis in Lyn-deficient mice. Oncogene 25, 3316–3324 (2006).
Ingley, E. et al. Lyn deficiency reduces GATA-1, EKLF and STAT5, and induces extramedullary stress erythropoiesis. Oncogene 24, 336–343 (2005).
Saporito, M.S., Ochman, A.R., Lipinski, C.A., Handler, J.A. & Reaume, A.G. MLR-1023 is a potent and selective allosteric activator of Lyn kinase in vitro that improves glucose tolerance in vivo. J. Pharmacol. Exp. Ther. 342, 15–22 (2012).
Tardif, J.C. et al. Effects of reconstituted high-density lipoprotein infusions on coronary atherosclerosis: a randomized controlled trial. J. Am. Med. Assoc. 297, 1675–1682 (2007).
Koppikar, P. et al. Efficacy of the JAK2 inhibitor INCB16562 in a murine model of MPLW515L-induced thrombocytosis and myelofibrosis. Blood 115, 2919–2927 (2010).
Marty, C. et al. Ligand-independent thrombopoietin mutant receptor requires cell surface localization for endogenous activity. J. Biol. Chem. 284, 11781–11791 (2009).
Shaw, J.A. et al. Infusion of reconstituted high-density lipoprotein leads to acute changes in human atherosclerotic plaque. Circ. Res. 103, 1084–1091 (2008).
Brown, M.S. & Goldstein, J.L. Suppression of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity and inhibition of growth of human fibroblasts by 7-ketocholesterol. J. Biol. Chem. 249, 7306–7314 (1974).
Brown, M.S. & Goldstein, J.L. Multivalent feedback regulation of HMG CoA reductase, a control mechanism coordinating isoprenoid synthesis and cell growth. J. Lipid Res. 21, 505–517 (1980).
Murphy, A.J. et al. ApoE regulates hematopoietic stem cell proliferation, monocytosis, and monocyte accumulation in atherosclerotic lesions in mice. J. Clin. Invest. 121, 4138–4149 (2011).
Bensinger, S.J. et al. LXR signaling couples sterol metabolism to proliferation in the acquired immune response. Cell 134, 97–111 (2008).
Armstrong, A.J., Gebre, A.K., Parks, J.S. & Hedrick, C.C. ATP-binding cassette transporter G1 negatively regulates thymocyte and peripheral lymphocyte proliferation. J. Immunol. 184, 173–183 (2010).
Kovárová, M. et al. Structure-function analysis of Lyn kinase association with lipid rafts and initiation of early signaling events after Fcɛ receptor I aggregation. Mol. Cell Biol. 21, 8318–8328 (2001).
Gieger, C. et al. New gene functions in megakaryopoiesis and platelet formation. Nature 480, 201–208 (2011).
Barter, P.J. et al. Effects of torcetrapib in patients at high risk for coronary events. N. Engl. J. Med. 357, 2109–2122 (2007).
Boden, W.E. et al. Niacin in patients with low HDL cholesterol levels receiving intensive statin therapy. N. Engl. J. Med. 365, 2255–2267 (2011).
Rader, D.J. & Tall, A.R. The not-so-simple HDL story: is it time to revise the HDL cholesterol hypothesis? Nat. Med. 18, 1344–1346 (2012).
Verstovsek, S. et al. Safety and efficacy of INCB018424, a JAK1 and JAK2 inhibitor, in myelofibrosis. N. Engl. J. Med. 363, 1117–1127 (2010).
Wolanskyj, A.P., Schwager, S.M., McClure, R.F., Larson, D.R. & Tefferi, A. Essential thrombocythemia beyond the first decade: life expectancy, long-term complication rates, and prognostic factors. Mayo Clin. Proc. 81, 159–166 (2006).
Dutta, P. et al. Myocardial infarction accelerates atherosclerosis. Nature 487, 325–329 (2012).
Tall, A.R., Yvan-Charvet, L., Westerterp, M. & Murphy, A.J. Cholesterol efflux: a novel regulator of myelopoiesis and atherogenesis. Arterioscler. Thromb. Vasc. Biol. 32, 2547–2552 (2012).
Chan, V.W., Meng, F., Soriano, P., DeFranco, A.L. & Lowell, C.A. Characterization of the B lymphocyte populations in Lyn-deficient mice and the role of Lyn in signal initiation and down-regulation. Immunity 7, 69–81 (1997).
Tong, W., Ibarra, Y.M. & Lodish, H.F. Signals emanating from the membrane proximal region of the thrombopoietin receptor (mpl) support hematopoietic stem cell self-renewal. Exp. Hematol. 35, 1447–1455 (2007).
Bersenev, A., Wu, C., Balcerek, J. & Tong, W. Lnk controls mouse hematopoietic stem cell self-renewal and quiescence through direct interactions with JAK2. J. Clin. Invest. 118, 2832–2844 (2008).
Acknowledgements
A.J.M. was supported by a postdoctoral fellowship from the American Heart Association (12POST11890019). This study was supported by US National Institutes of Health grant HL107653 (A.R.T.). We thank A.L. DeFranco and C.A. Lowell of the University of California, San Francisco for providing Lyn−/− bone marrow cells and W. Tong of the University of Pennsylvania for providing the antibody to c-MPL for flow cytometry analysis.
Author information
Authors and Affiliations
Contributions
A.J.M., N. Bijl and N.W. conceived the study, designed, performed and analyzed the experiments and wrote the manuscript. L.Y.-C., C.B.W., N. Bhagwat, A.R., Y.W. and J.A.S. designed, performed and analyzed experiments. R.L.L. and H.N. provided intellectual input and assisted with the preparation of the manuscript. A.R.T. conceived the study and contributed to writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Information (PDF 6732 kb)
Rights and permissions
About this article
Cite this article
Murphy, A., Bijl, N., Yvan-Charvet, L. et al. Cholesterol efflux in megakaryocyte progenitors suppresses platelet production and thrombocytosis. Nat Med 19, 586–594 (2013). https://doi.org/10.1038/nm.3150
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3150