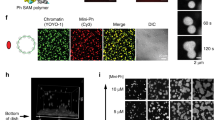
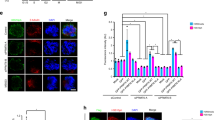
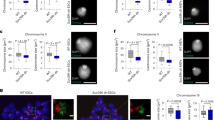
Abstract
Heterochromatin protein 1 (HP1) has an essential role in heterochromatin formation and mitotic progression through its interaction with various proteins. We have identified a unique HP1α-binding protein, POGZ (pogo transposable element-derived protein with zinc finger domain), using an advanced proteomics approach. Proteins generally interact with HP1 through a PxVxL (where x is any amino-acid residue) motif; however, POGZ was found to bind to HP1α through a zinc-finger-like motif. Binding by POGZ, mediated through its zinc-finger-like motif, competed with PxVxL proteins and destabilized the HP1α–chromatin interaction. Depletion experiments confirmed that the POGZ HP1-binding domain is essential for normal mitotic progression and dissociation of HP1α from mitotic chromosome arms. Furthermore, POGZ is required for the correct activation and dissociation of Aurora B kinase from chromosome arms during M phase. These results reveal POGZ as an essential protein that links HP1α dissociation with Aurora B kinase activation during mitosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fischer, T. et al. Diverse roles of HP1 proteins in heterochromatin assembly and functions in fission yeast. Proc. Natl Acad. Sci. USA 106, 8998–9003 (2009).
Hediger, F. & Gasser, S. M. Heterochromatin protein 1: don't judge the book by its cover! Curr. Opin. Genet. Dev. 16, 143–150 (2006).
Hiragami, K. & Festenstein, R. Heterochromatin protein 1: a pervasive controlling influence. Cell Mol. Life Sci. 62, 2711–2726 (2005).
Loyola, A. et al. The HP1α–CAF1–SetDB1-containing complex provides H3K9me1 for Suv39-mediated K9me3 in pericentric heterochromatin. EMBO Rep. 10, 769–775 (2009).
Maison, C. & Almouzni, G. HP1 and the dynamics of heterochromatin maintenance. Nat. Rev. Mol. Cell Biol. 5, 296–304 (2004).
Motamedi, M. R. et al. HP1 proteins form distinct complexes and mediate heterochromatic gene silencing by nonoverlapping mechanisms. Mol. Cell 32, 778–790 (2008).
Lachner, M., O'Carroll, D., Rea, S., Mechtler, K. & Jenuwein, T. Methylation of histone H3 lysine 9 creates a binding site for HP1 proteins. Nature 410, 116–120 (2001).
Nielsen, A. L. et al. Heterochromatin formation in mammalian cells: interaction between histones and HP1 proteins. Mol. Cell 7, 729–739 (2001).
Brasher, S. V. et al. The structure of mouse HP1 suggests a unique mode of single peptide recognition by the shadow chromo domain dimer. EMBO J. 19, 1587–1597 (2000).
Smothers, J. F. & Henikoff, S. The HP1 chromo shadow domain binds a consensus peptide pentamer. Curr. Biol. 10, 27–30 (2000).
Thiru, A. et al. Structural basis of HP1/PxVxL motif peptide interactions and HP1 localisation to heterochromatin. EMBO J. 23, 489–499 (2004).
Cheutin, T. et al. Maintenance of stable heterochromatin domains by dynamic HP1 binding. Science 299, 721–725 (2003).
Fischle, W. et al. Regulation of HP1-chromatin binding by histone H3 methylation and phosphorylation. Nature 438, 1116–1122 (2005).
Hirota, T., Lipp, J. J., Toh, B. H. & Peters, J. M. Histone H3 serine 10 phosphorylation by Aurora B causes HP1 dissociation from heterochromatin. Nature 438, 1176–1180 (2005).
Ruchaud, S., Carmena, M. & Earnshaw, W. C. Chromosomal passengers: conducting cell division. Nat. Rev. Mol. Cell Biol. 8, 798–812 (2007).
Hayashi-Takanaka, Y., Yamagata, K., Nozaki, N. & Kimura, H. Visualizing histone modifications in living cells: spatiotemporal dynamics of H3 phosphorylation during interphase. J. Cell Biol. 187, 781–790 (2009).
Sessa, F. et al. Mechanism of Aurora B activation by INCENP and inhibition by hesperadin. Mol. Cell 18, 379–391 (2005).
Rosasco-Nitcher, S. E., Lan, W., Khorasanizadeh, S. & Stukenberg, P. T. Centromeric Aurora-B activation requires TD-60, microtubules, and substrate priming phosphorylation. Science 319, 469–472 (2008).
Kelly, A. E. et al. Chromosomal enrichment and activation of the Aurora B pathway are coupled to spatially regulate spindle assembly. Dev. Cell 12, 31–43 (2007).
Ainsztein, A. M., Kandels-Lewis, S. E., Mackay, A. M. & Earnshaw, W. C. INCENP centromere and spindle targeting: identification of essential conserved motifs and involvement of heterochromatin protein HP1. J. Cell Biol. 143, 1763–1774 (1998).
Obuse, C. et al. A conserved Mis12 centromere complex is linked to heterochromatic HP1 and outer kinetochore protein Zwint-1. Nat. Cell Biol. 6, 1135–1141 (2004).
Kiyomitsu, T., Iwasaki, O., Obuse, C. & Yanagida, M. Inner centromere formation requires hMis14, a trident kinetochore protein that specifically recruits HP1 to human chromosomes. J. Cell Biol. 188, 791–807 (2010).
Lechner, M. S., Schultz, D. C., Negorev, D., Maul, G. G. & Rauscher, F. J. 3rd. The mammalian heterochromatin protein 1 binds diverse nuclear proteins through a common motif that targets the chromoshadow domain. Biochem. Biophys. Res. Commun. 331, 929–937 (2005).
Tudor, M., Lobocka, M., Goodell, M., Pettitt, J. & O'Hare, K. The pogo transposable element family of Drosophila melanogaster. Mol. Gen. Genet. 232, 126–134 (1992).
Bartholomeeusen, K. et al. Lens epithelium-derived growth factor/p75 interacts with the transposase-derived DDE domain of PogZ. J. Biol. Chem. 284, 11467–11477 (2009).
Yamagishi, Y., Sakuno, T., Shimura, M. & Watanabe, Y. Heterochromatin links to centromeric protection by recruiting shugoshin. Nature 455, 251–255 (2008).
Kitajima, T. S., Hauf, S., Ohsugi, M., Yamamoto, T. & Watanabe, Y. Human Bub1 defines the persistent cohesion site along the mitotic chromosome by affecting Shugoshin localization. Curr. Biol. 15, 353–359 (2005).
Hauf, S., Waizenegger, I. C. & Peters, J. M. Cohesin cleavage by separase required for anaphase and cytokinesis in human cells. Science 293, 1320–1323 (2001).
Cheeseman, I. M. et al. A conserved protein network controls assembly of the outer kinetochore and its ability to sustain tension. Genes Dev. 18, 2255–2268 (2004).
Dai, J., Sullivan, B. A. & Higgins, J. M. Regulation of mitotic chromosome cohesion by Haspin and Aurora B. Dev. Cell 11, 741–750 (2006).
Pouwels, J. et al. Shugoshin 1 plays a central role in kinetochore assembly and is required for kinetochore targeting of Plk1. Cell Cycle 6, 1579–1585 (2007).
Ditchfield, C. et al. Aurora B couples chromosome alignment with anaphase by targeting BubR1, Mad2, and Cenp-E to kinetochores. J. Cell Biol. 161, 267–280 (2003).
Tatsumi, Y., Ohta, S., Kimura, H., Tsurimoto, T. & Obuse, C. The ORC1 cycle in human cells: I. cell cycle-regulated oscillation of human ORC1. J. Biol. Chem. 278, 41528–41534 (2003).
Albritton, L. M., Tseng, L., Scadden, D. & Cunningham, J. M. A putative murine ecotropic retrovirus receptor gene encodes a multiple membrane-spanning protein and confers susceptibility to virus infection. Cell 57, 659–666 (1989).
Kitamura, T. et al. Retrovirus-mediated gene transfer and expression cloning: powerful tools in functional genomics. Exp. Hematol. 31, 1007–1014 (2003).
Kiyomitsu, T., Obuse, C. & Yanagida, M. Human Blinkin/AF15q14 is required for chromosome alignment and the mitotic checkpoint through direct interaction with Bub1 and BubR1. Dev. Cell 13, 663–676 (2007).
Taddei, A., Maison, C., Roche, D. & Almouzni, G. Reversible disruption of pericentric heterochromatin and centromere function by inhibiting deacetylases. Nat. Cell Biol. 3, 114–120 (2001).
Ishihama, Y. et al. Exponentially modified protein abundance index (emPAI) for estimation of absolute protein amount in proteomics by the number of sequenced peptides per protein. Mol. Cell Proteomics 4, 1265–1272 (2005).
Ando, S., Yang, H., Nozaki, N., Okazaki, T. & Yoda, K. CENP-A, -B, and -C chromatin complex that contains the I-type α-satellite array constitutes the prekinetochore in HeLa cells. Mol. Cell Biol. 22, 2229–2241 (2002).
Acknowledgements
We thank D. Goto, J. Nakayama, K. Hamada, Y. Murakami, G. Almouzni and M. Yanagida for comments, Y. Shinkai, M. Tachibana, M. Yanagida, T. Kiyomitsu, K. Yoda, S. Hatakeyama, K. Hanada, T. Kitamura, A. Imura and J-M. Peters for materials, T. Iwanaga and N. Sakai for technical instruction, and E. Ono for administration. R-S.N. was supported by a JSPS (Japan Society for the Promotion of Science) special fellowship. This work was supported by grants-in-aid from MEXT (Ministry of Education, Culture, Sports, Science and Technology) of Japan, and by Inamori, Uehara Memorial, Naito, Mochida Memorial and Matrix Science K.K. Foundations (C.O.).
Author information
Authors and Affiliations
Contributions
R-S.N. performed most of the experiments and analysis. K.N. analysed mass-spectrometry data. H-T.M. quantified microscopy and FRAP data with a help of H.K. O.I. generated some HP1-related constructs. T.H. provided CPC-related materials. N.N. and H.K. provided Histone H3-related antibodies. C.O. conceived the study. R-S.N., K.N., H.K. and C.O. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2337 kb)
Supplementary Information
Supplementary Movie 1 (MOV 1168 kb)
Supplementary Information
Supplementary Movie 2 (MOV 593 kb)
Supplementary Information
Supplementary Movie 3 (MOV 982 kb)
Supplementary Information
Supplementary Movie 4 (MOV 971 kb)
Rights and permissions
About this article
Cite this article
Nozawa, RS., Nagao, K., Masuda, HT. et al. Human POGZ modulates dissociation of HP1α from mitotic chromosome arms through Aurora B activation. Nat Cell Biol 12, 719–727 (2010). https://doi.org/10.1038/ncb2075
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2075
This article is cited by
-
Oligogenic inheritance in severe adult obesity
International Journal of Obesity (2024)
-
Chromodomain on Y-like 2 (CDYL2) implicated in mitosis and genome stability regulation via interaction with CHAMP1 and POGZ
Cellular and Molecular Life Sciences (2023)
-
Autism-associated protein POGZ controls ESCs and ESC neural induction by association with esBAF
Molecular Autism (2022)
-
Further delineation of the clinical spectrum of White–Sutton syndrome: 12 new individuals and a review of the literature
European Journal of Human Genetics (2022)
-
Expanding the neurological and behavioral phenotype of White-Sutton syndrome: a case report
Italian Journal of Pediatrics (2021)