Abstract
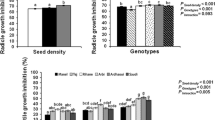
In previous studies, 15 putative allelopathic compounds detected in rice root exudates were quantified by GC/MS/MS. In this study, multiple regression analysis on these compounds determined that five selected phenolics, namely caffeic, p-hydroxybenzoic, vanillic, syringic, and p-coumaric acids, from rice exudates were best correlated with the observed allelopathic effect on arrowhead (Sagittaria montevidensis) root growth. Despite this positive association, determination of the phenolic acid dose-response curve established that the amount quantified in the exudates was much lower than the required threshold concentration for arrowhead inhibition. A similar dose-response curve resulted from a combination of all 15 quantified compounds. Significant differences between the amounts of trans-ferulic acid, abietic acid, and an indole also existed between allelopathic and non-allelopathic rice cultivars. The potential roles of these three compounds in rice allelopathy were examined by chemoassay. Overall, neither the addition of trans-ferulic acid nor 5-hydroxyindole-3-acetic acid to the phenolic mix significantly contributed to phytotoxicity, although at higher concentrations, trans-ferulic acid appeared to act antagonistically to the phytotoxic effects of the phenolic mix. The addition of abietic acid also decreased the inhibitory effect of the phenolic mix. These studies indicate that the compounds quantified are not directly responsible for the observed allelopathic response. It is possible that the amount of phenolic acids may be indirectly related to the chemicals finally responsible for the observed allelopathic effect.
Similar content being viewed by others
REFERENCES
Ando, T., Yoshida, S., and Nishiyama, I. 1983. Nature of oxidizing power of rice roots. Plant Soil 73:57-71.
Appel, H. M. 1993. Phenolics in ecological interactions: The importance of oxidation. J. Chem. Ecol. 19:1521-1552.
Armstrong, W. 1967. The use of polarography in the assay of oxygen diffusion from roots in anaerobic media. Physiol. Plant. 20:540-553.
Belz, R., Duke, S. O., and Hurle, K. 2002. Screening for allelopathy with dose-response? pp. 256-257, in 12th EWRS Symposium, Wageningen, The Netherlands, June 24–27.
Ben-Hammouda, M., Kremer, R. H., Monor, H. C., and Sarwar, M. 1995. A chemical basis for differential allelopathic potential of sorghum hybrids on wheat. J. Chem. Ecol. 21:775-786.
Cartwright, D., Langcake, P., Pryce, R. J., and Leworthy, D. P. 1977. Chemical activation of host defence mechanisms as a basis for crop protection. Nature 267:511-513.
Cartwright, D., Langcake, P., Pryce, R. J., Leworthy, D. P., and Ride, J. P. 1981. Isolation and characterization of two phytoalexins from rice as momilactones A and B. Phytochemistry 20:535-537.
Chou, C. H., Chang, F. J., and Oka, H. I. 1991. Allelopathic potential of a wild rice Oryza perennis. Taiwania 36:201-210.
Dalton, B. R. 1989. Physiochemical and biological processes affecting the recovery of exogeneously applied ferulic acid from tropical soils. Plant Soil 115:13-22.
Duke, S. O. 2002. The poison is in the dose: Hormesis, dose-response, and time courses, p. 55, in Third World Congress on Allelopathy: Challenge for the New Millenium–Book of Abstracts. Tsukuba, Japan, August 26–30.
Duke, S. O., Williams, R. D., and Markhart, A. H. 1983. Interaction of moisture stress and three phenolic compounds on lettuce seed germination. Ann. Bot. 52:923-926.
Einhellig, F. A. 1996. Interactions involving allelopathy in cropping systems. Agron. J. 88:886-893.
Einhellig, F. A. and Leather, G. A. 1988. Potentials for exploiting allelopathy to enhance crop production. J. Chem. Ecol. 14:1829-1844.
Foy, C. L. 1999. How to make bioassays for allelopathymore relevant to field conditions with particular reference to cropland weeds, pp. 25-34, in Inderjit, K. M. M. Dakshini, and C. L. Foy (eds.). Principles and Practices in Plant Ecology: Allelochemical Interactions. CRC Press, New York.
Hall, A. B., Blum, U., and Fites, R. 1982. Stress modification of allelopathy of Helianthus annuus L. debris on seed germination. Am. J. Bot. 69:776-783.
Hassan, S. M., Rao, A. N., Bastawisi, A. O., and Aidy, I. R. 1994. Weed management in broadcast seeded rice in Egypt, in The InternationalWorkshop on Constraints, Opportunities and Innovations for wet seeded rice. IRRI, Thailand.
Huang, Z., Haig, T., Wu, H., An, M., and Pratley, J. 2003. Correlation between phytotoxicity on annual ryegrass (Lolium rigidum) and production dynamics of allelochemicals within root exudates of an allelopathic wheat. J. Chem. Ecol. 29:2263-2280.
Kobayashi, A., Kim, M. J., and Kawazu, K. 1996. Uptake and exudation of phenolic compounds by wheat and antimicrobial components of the root exudate. Z. Naturforsch. 51c:527-533.
Kong, C., Xu, X., Hu, F., Chen, X., Bing, L., and Tan, Z. 2002. Using specific secondary metabolites as markers to evaluate allelopathic potentials of rice varieties and individual plants. Chin. Sci. Bull. 47:839-843.
Liebl, R. and Worsham, A. 1983. Inhibition of pitted morning glory (Ipomoea lacunosa L.) and certain other weed species by phytotoxic components of wheat (Triticum aestivum L.) straw. J. Chem. Ecol. 9:1027-1043.
Olofsdotter, M., Rebulanan, M., Madrid, A., Dali, W., Navarez, D., and Olk, D. C. 2002. Why phenolic acids are unlikely primary allelochemicals in rice. J. Chem. Ecol. 28:229-242.
Purvis, C. E. 1990. Allelopathy: A new direction in weed control. P. Prot. Q. 5:55-59.
Putnam, A. R. 1985. Allelopathic research in agriculture: Past highlights and potential, pp. 1-8, in A. C. Thompson (ed.). The Chemistry of Allelopathy: Biological Interactions Among Plants. American Chemical Society Symposium Series No. 268, ACS, Washington, DC.
Rasmussen, H. A. and Einhellig, F. A. 1977. Synergistic effects of p-coumaric and ferulic acids on germination and growth of grain sorghum. J. Chem. Ecol. 3:197-205.
Rasmussen, H. A. and Einhellig, F. A. 1979. Inhibitory effects of combinations of three phenolic acids on grain sorghum growth. P. Sci. Lett. 14:15-109.
Rimando, A. M., Olofsdotter, M., Dayan, F. E., and Duke, S. O. 2001. Searching for rice allelochemicals: An example of bioassay guided isolation. Agron. J. 93:16-20.
Rizvi, S. J. H. and Rizvi, V. 1992. Allelopathy: Basic and Applied Aspects. Chapman and Hall: London.
Romeo, J. T. and Weidenhamer, J. D. 1998. Bioassays for allelopathy in terrestrial plants, pp. 179-211, in K. F. Haynes and J. G. Millar (eds.). Methods in Chemical Ecology II: Bioassay Methods. Chapman and Hall, Sydney.
Seal. A. N., Pratley, J. E., Haig, T., and An, M. 2004. Identification and quantitation of compounds in a series of allelopatic and non-allelopathic rice root exudates. J. Chem. Ecol. 30:1629-1644.
Seigler, D. 1996. Chemistry and mechanisms of allelopathic interactions. Agron. J. 88:876-885.
Tanaka, F., Ono, S., and Hayasaka, T. 1990. Identification and evaluation of toxicity of rice root elongation inhibitors in flooded soils with added wheat straw. Soil Sci. Plant Nutr. 36:97-103.
Weidenhamer, J. D. and Romeo, J. T. 1989. Allelopathic properties of Polygonella myriophylla: Field evidence and bioassays. J. Chem. Ecol. 15:1957-1970.
Worsham, A. D. 1989. Current and potential techniques using allelopathy as an aid in weed management, pp. 275-292, in C. H. Chou and G. R. Waller (eds.). Phytochemical Ecology: Allelochemicals, Mycotoxins and Insect Pheromones and Allomones. Institute of Botany, Academia Sinica Monograph Series No. 9, Taipei.
Wu, H., Haig, T., Pratley, J., Lemerle, D., and An, M. 2002. Biochemical basis for wheat seedling allelopathy on the suppression of annual ryegrass. J. Agric. Food Chem. 50:4567-4571.
Wu, H., Pratley, J., Lemerle, D., and Haig, T. 1999. Crop cultivars with allelopathic capability. Weed Res. 39:171-181.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Seal, A.N., Haig, T. & Pratley, J.E. Evaluation of Putative Allelochemicals in Rice Root Exudates for Their Role in the Suppression of Arrowhead Root Growth. J Chem Ecol 30, 1663–1678 (2004). https://doi.org/10.1023/B:JOEC.0000042075.96379.71
Issue Date:
DOI: https://doi.org/10.1023/B:JOEC.0000042075.96379.71