Abstract
1.Animal experiments have shown nitric oxide synthase inhibitors to have antidepressant-like properties. However, the effects of clinically available antidepressants on nitric oxide production in the brain remain unclear. In the present study, we examined whether imipramine, a conventional antidepressant, changes the levels of type-II nitric oxide synthase mRNA and nitrate, a final nitric-oxide-oxidation product measurable in vivo, in the rat brain.
2.Type-II nitric oxide synthase mRNA was detected using a reverse transcription-polymerase chain reaction method and nitrate was measured with a combination of high-performance liquid chromatography and the Griess reaction.
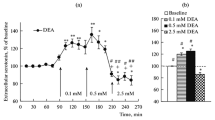
3.In untreated rats, type-II nitric oxide synthase mRNA was not detected in the hypothalamus, hippocampus, cerebral cortex, brain stem, or cerebellum. However, after 28-day oral administration of imipramine, it was detected in every brain region tested. Nitrate levels in the hypothalamus and cerebral cortex increased after 28-day treatment. In the hypothalamus, nitrate levels increased dose-dependently. These dose-dependent nitrate level changes were prevented by pretreatment with a nitric oxide synthase inhibitor. Moreover, the preventive effect of N G-nitro-L-arginine methyl ester was reversed by coadministration of L-arginine, a nitric oxide substrate.
4.These results suggest that chronic imipramine treatment induces nitric oxide synthase gene expression in the brain, followed by augmented NO production.
Similar content being viewed by others
References
Bredt, D. S., Hwang, P. M., Glatt, C. E., Lowenstein, R. R., and Snyder, S. H. (1991). Cloned and expressed nitric oxide synthase structurally resembles cytochrome P-450 reductase. Nature 351:714–718.
Galea, E., Reis, D. J., and Feinstein, D. L. (1994). Cloning and expression of inducible nitric oxide synthase from rat astrocytes. J. Neurosci. Res. 37:406–414.
Garthwaite, J. (1991). Glutamate, nitric oxide and cell–cell signalling in the nervous system. Trends Neurosci. 14:60–67.
Harkin, A. J., Bruce, K. H., Craft, B., and Paul, I. A. (1999). Nitric oxide synthase inhibitors have antidepressant-like properties in mice 1. Acute treatments are active in the forced swim test. Eur. J. Pharmacol. 372:207–213.
Hikida, K., Inoue, Y., Miyazaki, T., and Kojima, N. (1989). Determination of bromperidol in serum by automated column-switching high-performance liquid chromatography. J. Chromatogr. 495:227–234.
Iyengar, R., Stuehr, D. J., and Marletta, M. A. (1987). Macrophage synthesis of nitrite, nitrate, and N-nitrosamines: Precursors and role of the respiratory burst. Proc. Natl. Acad. Sci. U.S.A. 84:6369–6373.
Jopek, R., Kata, M., and Nowak, G. (1999). The activity of rat brain nitric oxide synthase following chronic antidepressant treatment. Acta Pol. Pharm. 56:307–310.
Karatinos, J., Rosse, R. B., and Deutsch, S. I. (1995). The nitric oxide pathway: Potential implications for treatment of neuropsychiatric disorders. Clin. Neuropharmacol. 18:482.
Karolewicz, B., Bruce, K. H., Lee, B., and Paul, I. A. (1999). Nitric oxide synthase inhibitors have antidepressant-like properties in mice 2. Chronic treatment results in downregulation of cortical β-adrenoceptors. Eur. J. Pharmacol. 372:215–220.
Krulik, R., Farska, I., and Jindrova, O. (1981). Plasma imipramine levels and demethylase activity in the liver of stressed amnimals. Neuropsychobiology 7:12–16.
Langer, S. Z. (1981). Presynaptic regulation of the release of catecholamines. Pharmacol. Rev. 32:337–362.
Lipton, S. A., Choi, Y.-B., Pan, Z.-H., Lei, S. Z., Chen, H.-S. V., Sucher, N. J., Loscalzo, J., Singel, D. J., and Stamler, J. S. (1993). A redox-based mechanism for the neuroprotective and neurodestructive effects of nitric oxide and related nitroso-compounds. Nature 364:626–632.
Lorrain, D. S., and Hull, E. M. (1993). Nitric oxide increases dopamine and serotonin release in the medial preoptic area. NeuroReport 5:87–89.
Maj, J., Przegalinski, E., and Mogilnicka, E. (1984). Hypothesis concerning the mechanism of action of antidepressant drugs. Rev. Physiol. Biochem. Pharmacol. 100:1–73.
Moncada, S., and Higgs, A. (1993). The L-arginine-nitric oxide pathway. N. Engl. J. Med. 329:2002–2012.
Nakaki, T., Mishima, A., Fujii, T., Suzuki, E., and Shintani, F. (1997). Nitric oxide and neurodegenerative diseases. Curr. Top. Pulm. Pharmacol. Toxicol. 3:157–163.
Nowak, G., Kata, M., Jopek, R., and Siedlecki, A. (1997). Chronic electroconvulsive treatment increases the activity of nitric oxide synthase in the rat brain. Pol. J. Pharmacol. 49:379.
Pögün, S., and Kuhar, M. J. (1994). Regulation of neurotransmitter reuptake by nitric oxide. Ann. N.Y. Acad. Sci. 17:305–315.
Ryan, N. D., Puig-Antich, J., Cooper, T., Rabinovich, H., Ambrosini, P., Davies, M., King, J., Torres, D., and Fried, J. (1986). Imipramine in adolescent major depression: Plasma level and clinical response. Acta Psychiatr. Scand. 73:275–288.
Schmidt, H. H., and Walter, U. (1994). NO at work. Cell 78:919–925.
Seilicovich, A., Lasaga, M., Befumo, M., Duvilanski, B. H., del Carman, Diaz M., Rettori, V., and McCann, S. M. (1995). Nitric oxide inhibits the release of norepinephrine and dopamine from the medial hypothalamus of the rat. Proc. Natl. Acad. Sci. U.S.A. 92:11299–11302.
Sessa, W. C. (1994). The nitric oxide synthase family of proteins. J. Vasc. Res. 31:131–143.
Sugita, S., Kobayashi, A., Suzuki, S., Yoshida, T., and Nakazawa, K. (1987). High-performance liquid chromatographic determination of imipramine and its metabolites in rat brain. J. Chromatogr. 421:412–417.
Sulser, F., Janowsky, A. J., Okada, F., Manier, D. H., and Mobley, P. L. (1983). Regulation of recognition and action function of the norepinephrine (NE) receptor-coupled adenylate cyclase system in brain. Implications for the therapy of depression. Neuropsychopharmacology 22:425–431.
Suzuki, E., Kageyama, H., Nakaki, T., Kanba, S., Inoue, S., and Miyaoka, H. (2003). Nitric oxide induced heat shock protein 70 mRNA in rat hypothalamus during acute restraint stress under sucrose diet. Cell. Mol. Neurobiol. 23:XXX-XXX.
Suzuki, E., Nakaki, T., Shintani, F., Kanba, S., and Miyaoka, H. (2002). Antipsychotic, antidepressant, anxiolytic, and anticonvulsant drugs induce Type II nitric oxide synthase mRNA in rat brain. Neurosci. Lett. 333:217–219.
Suzuki, E., Shintani, F., Kanba, S., Asai, M., and Nakaki, T. (1996). Induction of interleukin-1β and interleukin-1 receptor antagonist mRNA by chronic treatment with various psychotropics in widespread area of rat brain. Neurosci. Lett. 215:201–204.
Suzuki, E., Shintani, F., Kanba, S., Asai, M., and Nakaki, T. (1997). Immobilization stress increases mRNA levels of interleukin-1 receptor antagonist in various rat brain regions. Cell. Mol. Neurobiol. 17:557–562.
Suzuki, E., Yagi, G., Nakaki, T., Kanba, S., and Asai, M. (2001). Elevated plasma nitrate levels in depressive states. J. Affect. Disord. 63:221–224.
Wegener, G. W., Volke, V., Harvey, B. H., and Rosenberg, R. (2003). Local, but not systemic, administration of serotonergic antidepressants decreases hippocampal nitric oxide synthase activity. Brain Res. 959:128–134.
Xie, Q.-W., Cho, H. J., Calaycay, J., Mumford, R. A., Swiderek, K. M., Lee, T. D., Ding, A., Troso, T., and Nathan, C. (1992). Cloning and characterization of inducible nitric oxide synthase from mouse macropharge. Science 256:225.
Yamada, K., and Nabeshima, T. (1997). Simultaneous measurement of nitrite and nitrate levels as indices of nitric oxide release in the cerebellum of conscious rats. J. Neurochem. 68:1234–1243.
Yildiz, F., Erden, B. F., Ulak, G., Utkan, T., and Gacar, N. (2000). Antidepressant-like effect of 7-nitroindazole in the forced swimming test in rats. Psychopharmacology 149:41–44.
Zhu, X.-Z., and Luo, L.-G. (1992). Effect of nitroprusside (nitric oxide) on endogenous dopamine release from rat striatal slices. J. Neurochem. 59:932–935.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Suzuki, E., Nakaki, T., Kanba, S. et al. Long-Term Imipramine Treatment Increases Nitrate Levels in the Rat Hypothalamus. Cell Mol Neurobiol 23, 953–962 (2003). https://doi.org/10.1023/B:CEMN.0000005323.10335.93
Issue Date:
DOI: https://doi.org/10.1023/B:CEMN.0000005323.10335.93