Abstract
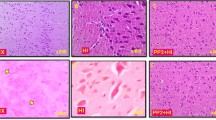
Hypoxia results in generation of nitric oxide (NO) free radicals, activation of caspase-3, and genomic DNA fragmentation. The present study tests the hypothesis that hypoxia-induced caspase-3 activation and DNA fragmentation are nitric oxide mediated. Studies were conducted in newborn piglets, divided into normoxic (n = 5), hypoxic (n = 5), and hypoxic-7-NINA (n = 6). Hypoxic-7-NINA group received the neuronal nitric oxide synthase inhibitor, 7-Nitroindazole (7-NINA). Caspase-3 activity was determined spectrofluorometrically using enzyme-specific substrates. Sections from the neocortex were stained with an antiserum recognizing active caspase-3. Purified DNA was separated by gel electrophoresis. Administration of 7-NINA resulted in decreased immunoreactivity of caspase-3 (mean LI: 20.2%) as compared to the untreated hypoxia group (mean LI: 57.5%) (P < 0.05). 7-NINA attenuated caspase-3 enzymatic activity as well in comparison to the untreated hypoxia group (P < 0.05). Furthermore, multiple low molecular weight bands corresponding to DNA fragments were present in the hypoxic but not in the normoxic or hypoxic-7-NINA groups. Inhibition of nNOS abates the hypoxia-induced increase in active caspase-3 immunoreactivity, as well as enzymatic activity in cortical neurons, and DNA fragmentation in brain homogenates. We conclude that the coordinate increase of capase-3 activity and fragmentation of nuclear DNA in the hypoxic newborn piglet brain are NO mediated.
Similar content being viewed by others
REFERENCES
Mishra, O. P., Zanelli, S. A., and Ohnishi, S. T. 2000. Hypoxia-induced generation of nitric oxide free radicals in cerebral cortex of newborn guinea pigs. Neurochem. Res. 25:1559-1565.
Kumura, E., Yshimine, T., Tanake, S., Hayakawa, T., Shiga, T., and Kosake, H. 1994. Nitrosyl hemoglobin production during reperfusion after focal cerebral-ischemia in rats. Neurosci. Lett. 177:165-167.
Kuppusamy, P., Ohnishi, S. T., Numagami, Y., Ohnishi, T., and Zweiger, J. L. 1995. 3-Dimensional imaging of nitric-oxide production in the rat brain subjected to ischemia-hypoxia. J. Cereb. Blood Flow Metab. 15:899-903.
Olesen, S. P., Moller, A., Morodvintcez, P. I., Busse, R., and Mulsch, A. 1997. Regional measurement of NO formed in vivo during brain ischemia. Acta Neurol. Scand. 95:219-224.
Buisson, A., Margaill, I., Callebert, M., Plotkine, M., and Boulu, R. G. 1993. Mechanisms involved in the neuroprotective activity of a nitric oxide synthase inhibitor during focal cerebral ischemia. J. Neurochem. 61:690-696.
Dawson, V. L., Kizushi, V. M., Huang, P. L., Snyder, S. H., and Dawson, T. M. 1996. Resistance to neurotoxicity in cortical cultures from neuronal nitric oxide synthase-deficient mice. J. Neurosci. 16:2479-2487.
Huang, Z., Huang P. L., Meng, W., Ayata, C., Fishman, M. C., and Moskowitz, M. A. 1996. Enlarged infracts in endothelial nitric oxide synthase knockout mice are attenuated by nitro-L-arginine. J Cereb. Blood Flow Metab. 16:981-987.
Iadecola, C. 1997. Bright and dark sides of nitric oxide in ischemic brain injury. Trends Neurosci. 20:132-139.
Dawson, V. L., Dawson, T. M., Bartley, D. A., Uhl, G. R., and Snyder, S. H. 1993. Mechanisms of nitric oxide-mediated neurotoxicity in primary brain cultures. J. Neurosci. 13:2651-2661.
Dawson, V. L. and Dawson, T. M. 1996. Nitric oxide neurotoxicity. J. Chem. Neuroanat. 10:179-190.
Cohen, G. 1997. Caspases: The executioners of apoptosis. Biochem. J. 326:1-16.
Graeber, M. B. and Moran, L. B. 2002. Mechanisms of cell death in neurodegenerative diseases: Fashion, fiction, and facts. Brain Pathol. 12:385-390.
Kuida, K., Zheng, T. S., Na, S., Kuan, C., Yang, D., Karasuyama, H., Rakic, P., and Flavell, R. A. 1996. Decreased apoptosis in the brain and premature lethality in CPP32-deficient mice. Nature 384:368-372.
Akhter, W., Ashraf, Q. M., Zanelli, S. A., Mishra, O. P., and Delivoria-Papadopoulos, M. 2001. Effect of graded hypoxia on cerebral cortical genomic DNA fragmentation in newborn piglets. Biol. Neonate. 79:187-193.
Ashraf, Q. M., Zubrow, A. B., Mishra, O. P., and Delivoria-Papadopoulos, M. 2002. Nitration of Bax and Bcl-2 proteins during hypoxia in cerebral cortex of newborn piglets and the effect of nitric oxide synthase inhibition. Biol. Neonate. 81:65-72.
Rossiter, J. P., Anderson, L. L., Yang, F., and Cole, G. M. 2002. Caspase-3 activation and caspase-like proteolytic activity in human perinatal hypoxicischemic brain injury. Acta Neuropathol. (Berl.) 103:66-73.
Lamprecht, W., Stein, P., Heinz, F., and Weisser, H. 1974. Creatine phoshate. Pages 1777-1778, in Bergmeyer, H. U. (ed.), Methods of enzymatic analysis, 4th ed., New York, Academic Press.
Bredt, D. S. and Snyder, S. H. 1989. Nitric oxide mediates glutamate-linked enhancement of cGMP levels in the cerebellum. Proc. Natl. Acad. Sci. USA 86:9030-9033.
Dwyer, M. A., Bredt, D. S., and Snyder, S. H. 1991. Nitric oxide synthase: Irreversible inhibition by L-NG-nitroarginine in brain in vitro and in vivo. Biochem. Biophys. Res. Commun. 176:1136-1141.
Guiffrida, A. M., Cox, D., and Mathias, A. P. 1975. RNA polymerase activity in various classes of nuclei from different regions of rat brain during postnatal development. J. Neurochem. 24:749-755.
Austoker, J., Cox, D., and Mathias, A. P. 1972. Fractionation of nuclei from brain by zonal centrifugation and a study of the ribonucleic acid polymerase activity in the various classes of nuclei. Biochem. J. 129:1139-1155.
Higuchi, Y. and Lynn, S. 1995. Purification of all forms of HeLa cell mitochondrial DNA and assessment of damage to it caused by hydrogen peroxide treatment of mitochondria or cells. J. Biol Chem. 270:7950-7956.
Katsetos, C. D., Frankfurter, A., Christakos, S., Mancall, E.L., Vlachos, I., and Ulrich, H. 1993. Differential localization of class-III β-tubulin isotype (β III) and calbindin-D28k defines distinct neuronal types in the developing human cerebellar cortex. J. Neuropathol. Exp. Neurol. 52:655-666.
Katsetos, C. D., Spandou, E., Legido, A., Taylor, M. L., Zanelli, S. A., Chadaverian, J. P., Christakos, S., Mishra, O. P., and Delivoria-Papadopoulos, M. 2001. Acute hypoxia-induced alterations of calbindin-D28k immunoreactivity in cerebellar Purkinje cells of the guinea pig fetus at term. J. Neuropathol. Exp. Neurol. 60:470-482.
Mishra, O. P. and Delivoria-Papadopoulos, M. 1999. Cellular mechanisms of hypoxic injury in the developing brain. Brain Res. Bull. 48:233-238.
Moore, P. K., Wallace, P., Gaffen, Z., Hart, S. L., and Babbedge, R. C. 1993. Characterization of the novel nitric oxide synthase inhibitor 7-nitro indazole and related indazoles: Antinociceptive and cardiovascular effects. Br. J. Pharmacol. 110:219-224.
Moore, P. K., and Handy, R. L. C. 1997. Selective inhibitors of neuronal nitric oxide synthase—is no NOS really good NOS for the nervous system? Trends Pharmacol. 18:204-211.
Rairigh, R. L., Storme, L., Parker, T. A., Le Cras, T. D., Markham, N., Jakkula, M., and Abman, S. H. 2000. Role of neuronal nitric oxide synthase in regulation of vascular and ductus arteriosus tone in the ovine fetus. Am. J. Physiol. Lung Cell. Mol. Physiol. 278:L105-110.
Leker, R. R., Teichner, A., Ovadia, H., Keshet, E., Reinherz, E., and Ben-Hur, T. 2001. Expression of endothelial nitric oxide synthase in the ischemic penumbra: Relationship to expression of neuronal nitric oxide synthase and vascular endothelial growth factor. Brain Res. 909:1-7.
Mishra, O. P. and Delivoria-Papadopoulos, M. 2001. Effect of graded hypoxia on high-affinity Ca2+-ATPase activity in cortical neuronal nuclei of newborn piglets. Neurochem. Res. 26:1335-1341.
Bolanos, J. P. and Almeida, A. 1999. Roles of nitric oxide in brain hypoxia-ischemia. Biochem. Biophys. Acta 1411:415-436.
Zanelli, S. A., Ashraf, Q. M., and Mishra, O. P. 2002. Nitration is a mechanism of regulation of the NMDA receptor function during hypoxia. Neuroscience 112:869-877.
Tamatani, M., Ogawa, S., Niitsu, Y., and Tohyama, M. 1998. Involvement of Bcl-2 family and caspase-3-like protease in NO-mediated neuronal apoptosis. J. Neurochem. 71:1588-1596.
Zubrow, A. B., Delivoria-Papadopoulos, M., Ashraf, Q. M., Ballesteros, J. R., Fritz, K. I., and Mishra, O. P. 2001. Nitric oxide-mediated expression of Bax protein and DNA fragmentation during hypoxia in neuronal nuclei from newborn piglets. Brain Res. 954:60-67.
Wolf, B. B., Schuler, M., Echeverri, F., and Green, D. R. 1999. Caspase-3 is the primary activator of apoptotic DNA fragmentation via DNA fragmentation factor-45/Inhibitor of caspase-activated DNAse inactivation. J. Biol. Chem. 274:30651-30656.
Enari, M., Sakahira, H., Yokoyama, H., Okawa, K., Iwamatsu, A., and Nagata, S. 1998. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 391:43-50.
Sakahira, H., Enari, M., and Nagata, S. 1998. Cleavage of CAD inhibitor in CAD activation and DNA degradation during apoptosis. Nature 391:96-99.
Liu, X., Zou, H., Slaughter, C., and Wang, X. 1997. DFF, a heterodimeric protein that functions downstream of caspase-3 to trigger DNA fragmentation during apoptosis. Cell 89:175-184.
Wood, D. E., Thomas, A., Devi, L. A., Berman, Y., Beavis, R. C., Reed, J. C., and Newcomb, E. W. 1998. Bax cleavage is mediated by calpain during drug-induced apoptosis. Oncogene 17:1069-1078.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Parikh, N.A., Katsetos, C.D., Ashraf, Q.M. et al. Hypoxia-Induced Caspase-3 Activation and DNA Fragmentation in Cortical Neurons of Newborn Piglets: Role of Nitric Oxide. Neurochem Res 28, 1351–1357 (2003). https://doi.org/10.1023/A:1024992214886
Issue Date:
DOI: https://doi.org/10.1023/A:1024992214886