Abstract
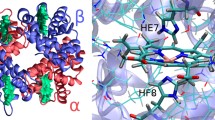
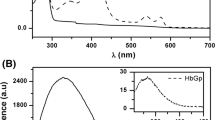
The kinetics of CNProto- and CNDeutero-hemin binding to apohemoglobin A2 was investigated in a stopped-flow device in 0.05 M potassium phosphate buffer, pH 7, at 10°C. The overall kinetic profile exhibited multiple phases: Phases I–IV corresponding with heme insertion (8.5−13 × 107 M−1 s−1), local structural rearrangement (0.21−0.23 s−1), global αδ structural event (0.071−0.098 s−1), and formation of the Fe–His bond (0.009−0.012 s−1), respectively. Kinetic differences observed between apohemoglobin A2 and apohemoglobin A (previously studied) prompted an analysis of the structures of β and δ chains through molecular modeling. This revealed a structural repositioning of the residues not only at, but also distant from the site of the amino acid substitutions, specifically those involved in the heme contact and subunit interface. A significant global change was observed in the structure of the exon-coded 3 region and provided additional evidence for the designation of this as the subunit assembly domain.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vasudevan, G., McDonald, M.J. Analysis of the Global Architecture of Hemoglobin A2 by Heme Binding Studies and Molecular Modeling. J Protein Chem 17, 319–327 (1998). https://doi.org/10.1023/A:1022551131455
Published:
Issue Date:
DOI: https://doi.org/10.1023/A:1022551131455