Abstract
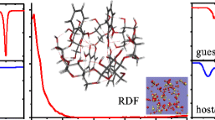
β-Cyclodextrin (β-CD) and p-hydroxybenzaldehyde (p-HB) were studied by 1H-NMR in deuterated aqueous solution and the stoichiometry of the resulting complex (1:1) was determined by the continuous variation method. Inclusion of p-HB in β-CD was confirmed by the observation of NMR shifts for the inside H5 protons of the β-CD cavity. In the solid state X-ray analysis was carried out and revealed the detailed structure of the inclusion complex. Two β-CDs cocrystallize with four p-HB and 9.45 water molecules[2(C6H10O5)7·4C7H6O2·9.45H2O] in the triclinic space group P1 with unit cell parameters: a = 15.262(2), b = 15.728(1), c = 16.350(1) Å, α = 92.67(1)°, β = 96.97(1)°, γ = 103.31(1)°. The anisotropic refinement of 1973 atomic parameters converged at an R-factor = 0.066 for 10157 data with Fo 2 > 2σ (Fo 2). The 2:4 stoichiometry for the β-CD inclusion complex with p-HB in the crystalline state is different from that obtained in solution. β-CD forms dimers stabilized by direct O2(m)1O3(m)1·O2(n)2O3(n)2 hydrogen bonds (intradimer) and by indirect O6(m)1·O6(n)2 hydrogen bonds with one or two bridging water molecules joined in between (interdimer). These dimers are stacked like coins in a roll constructing infinite channels where the p-HB molecules are included. The p-HB molecules direct their polar CHO and OH groups into the nonpolar β-CD cavities and are hydrogen bonded to each other, yielding infinite, antiparallel chains. In addition, crystals of the complex were also investigated with thermogravimetry, vibrational spectroscopy (FTIR), and 13C CP-MAS NMR spectroscopy. The results obtained enabled us to structurally characterize the β-CD inclusion complex with p-HB.
Similar content being viewed by others
References
W. Saenger: Angew. Chem. Int. Ed. Engl. 19, 344 (1980).
J. Szejtli: Chem. Rev. 98, 1743 (1998).
For a recent review on applications of cyclodextrins, see J. Szejtli: Proceedings of the 10th International Symposium on Cyclodextrins, Kluwer Academic Publishers, Dordrecht (2000).
M.V. Rekharsky and Y. Inoue: Chem. Rev. 98, 1875 (1998).
K.-H. Frömming and J. Szejtli: Cyclodextrins in Pharmacy, Kluwer Academic Publishers, Dordrecht (1994), pp. 66.
For examples of inclusion complexes of β-CD and organometallic guests, see (a) S.S. Braga, I.S. Gonçalves, A.D. Lopes, M. Pillinger, J. Rocha, C.C. Romão, and J.J.C. Teixeira-Dias: J. Chem. Soc. Dalton Trans., 2964–2968 (2000); (b) I. Turel, A. Demšar, and J. Košmrlj: J. Incl. Phenom. Macrocycl. Chem. 35, 595 (1999).
L. Szente, Comprehensive Supramolecular Chemistry, J.L. Atwood, J.E.D. Davies, D.D. MacNicol and F. Vögtle Exec. Ed., Vol 3, J. Szejtli and T. Osa (ed.), chap. 7.
K. Harata: Chem. Rev. 98, 1883 (1998).
A. Moreira da Silva, A. Amado, P. Ribeiro-Claro, J. Empis, and J.J.C. Teixeira-Dias: J. Carbohydr. Chem. 14, 677 (1995).
P. Job: Ann. Chim. 9, 113 (1928).
G.M. Sheldrick: SHELXS97, Program for Crystal Structure Solution, University of Göttingen, Germany (1997).
G.M. Sheldrick: SHELXL97, Program for Crystal Structure Refinement, University of Göttingen, Germany (1997).
D.E. McRee: Practical Protein Crystallography, Academic Press, San Diego (1993).
J. Redondo, J. Frigola, A. Torrens, and P. Lupón: Mag. Reson. Chem. 33, 104 (1995).
A.S. Moreira, J. Empis, and J.J.C. Teixeira-Dias: J. Incl. Phenom. Macrocycl. Chem. 33, 81 (1999).
P. V. Demarco and A.L. Thakkar: J. Chem. Soc. Chem. Commun. 2 (1970).
Y. Bahaddi, H. Galons, and N. Rysanek: Bull. Soc. Chim. Fr. 132, 330 (1995).
F. Djedaini, S.Z. Lin, B. Perly, and D. Wouessidjeswe: J. Pharm. Sci. 79, 643 (1987).
A. Ganza-Gonzalez, J.L. Vila-Jato, S. Anguiano-Igea, F.J. Otero-Espinar, and J. Blanco-Méndez: Int. J. Pharm. 106, 179 (1994).
H.-S. Choi, C. Chang, and A.M. Knevel: Pharm. Res. 9, 582 (1992)
M.J. Gidley and S.M. Bociek: J. Am. Chem. Soc. 110, 3820 (1988).
X.-T. Li, J.-H. Li, G.-E. Zhang, G.-X. Xi, and X.-D. Lou: Thermochim. Acta 262, 165 (1995).
D. Cremer and J.A. Pople: J. Am. Chem. Soc. 97, 1354 (1975).
IUPAC-IUB Joint Commision on Biochemical Nomenclature (JCBN) Symbols for specifying the conformation of polysaccharide chains, Recommendations 1981, Eur. J. Biochem. 131, 5 (1983).
F. Iwasaki: Acta Crystallogr. B33, 1646 (1977).
W. Saenger: Isr. J. Chem. 25, 43 (1985).
K. Harata: Crystallographic Studies (Comprehensive Supramolecular Chemistry, Vol. 3, J. Szejtli and T. Osa (eds.)), Pergamon, Oxford (1996), pp. 279.
R. Tokuoka, T. Fujiwara, and K.-I. Tomita: Acta Crystallogr. B37, 1158 (1981).
A. Rontoyianni, I.M. Mavridis, E. Hadjoudis, and A.J.M. Duisenberg: Carbohydr. Res. 252, 19 (1994).
D. Mentzafos, I.M. Mavridis, G. le Bas, and G. Tsoucaris: Acta Crystallogr. B47, 746 (1991).
(a) F.H. Herbstein and R.E. Marsh:Acta Crystallogr. B54, 677 (1998); (b) I.M. Mavridis and E. Hadjoudis: Carbohydr. Res. 229, 1 (1992).
M.R. Caira and D.R. Dodds: J. Incl. Phenom. Macrocycl. Chem. 34, 19 (1999).
J.J. Stezowski, K.H. Jogun, E. Eckle, and K. Bartels: Nature 274, 617 (1978). 125
M.N. Burnett and C.K. Johnson: ORTEPIII, Thermal-Ellipsoid Plot Program for Crystal Structure Illustrations, Oak Ridge Natl. Lab., USA (1996).
P.J. Kraulis: J. Appl. Crystallogr. 24, 946 (1991).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Braga, S.S., Aree, T., Imamura, K. et al. Structure of the β-Cyclodextrin·p-Hydroxybenzaldehyde Inclusion Complex in Aqueous Solution and in the Crystalline State. Journal of Inclusion Phenomena 43, 115–125 (2002). https://doi.org/10.1023/A:1020412412907
Issue Date:
DOI: https://doi.org/10.1023/A:1020412412907