Abstract
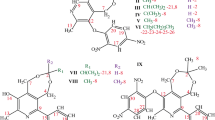
The solution conformations of the opioid phenylmorphan (5-m-hydroxyphenyl-2-methylmorphan) and its 9α-methyl analogue were studied using one- and two-dimensional high resolution NMR techniques. The NMR spectra were analyzed by interpreting the phase-sensitive 2-D COSY and double quantum filtered COSY spectra, 1H –1H vicinal coupling constants, and nuclear Overhauser effects in the phase-sensitive 2-D NOESY spectra. The results show that, for both compounds, a chair–chair conformation of the cyclohexane and piperidine rings is exclusively preferred with some distortion of the rings from perfectly staggered chairs. For phenylmorphans, the phenyl ring is oriented to fit into the cleft formed by the cyclohexane and piperidine rings. Thus, for the ( + )-enantiomer, the phenyl group assumes the same orientation with regard to the piperidine ring as morphine consistent with the morphine-like properties of the compound. For the 9α-methyl analogue, the plane of the phenyl ring essentially bisects the piperidine ring to which it is attached and is outside of the required range of opioid agonists. This is consistent with the atypical properties of the two enantiomers. The NMR results are compared to the conformations of (–)-phenylmorphan and the ( + )-9α-methyl analogue in the crystal state and to the results of molecular mechanics (MM2) studies.
Similar content being viewed by others
REFERENCES
E. L. May and M. Takeda. Optical isomers of miscellaneous strong analgetics. J. Med. Chem. 13:805–807 (1970).
H. H. Ong, T. Oh-shi, and E. L. May. Phenylmorphan agonists-antagonists. J. Med. Chem. 17:987–989 (1974).
M. Froimowitz, C. G. Pick, and G. W. Pasternak. Phenylmorphans and analogues: Opioid receptor subtype selectivity and effect of conformation on activity. J. Med. Chem. 35:1521–1525 (1992).
H. Awaya, E. L. May, M. D. Aceto, H. Merz, M. E. Rogers, and L. S. Harris. Racemic and optically active 2,9-dimethyl-5-(m-hydroxyphenyl)morphans and pharmacological comparisons with the 9-demethyl homologues. J. Med. Chem. 27:536–539 (1984).
T. G. Cochran. Stereochemistry and absolute configuration of the analgesic agonist-antagonist (−)-5-m-hydroxyphenyl-2-methylmorphan. J. Med. Chem. 17:987–989 (1974).
M. Froimowitz, W. Pangborn, and V. Cody. Absolute configuration and conformation of the pure opioid antagonist (+)-2,9α-methyl-5-(m-hydroxyphenyl)morphan. Chirality 4:377–383 (1992).
H. Tecle and G. Hite. Narcotic antagonists—The azabicyclanes. I. New insights into the molecular modes of receptor interactions of narcotic analgetics and narcotic antagonists. In Problems of Drug Dependence, 1976, National Academy of Sciences, Washington, DC, 1976, pp. 464–470.
D. S. Fries and P. S. Portoghese. Stereochemical studies of medicinal agents. 20. Absolute configuration and analgetic potency of α-promedol enantiomers. The role of the C-4 chiral center in conferring stereoselectivity in axial-and equatorial-phenyl prodine congeners. J. Med. Chem. 19:1155–1158 (1976).
M. Froimowitz, P. Salva, G. J. Hite, G. Gianutsos, P. Suzdak, and R. Heyman. Conformational properties of α-and β-azabicyclane opiates. The effect of conformation on pharmacological activity. J. Comput. Chem. 5:291–298 (1984).
M. Froimowitz. Preferred conformers for the pharmacologically typical and atypical antipodes of phenylmorphan opiates. J. Med. Chem. 27:1234–1237 (1984).
W. R. Croasmun and R. M. K. Carlson (eds.). Methods in Stereochemical Analysis, Vol. 9, VCH, New York, 1987.
M. Karplus. Contact electron-spin coupling of nuclear magnetic moments. J. Chem. Phys. 30:11–15 (1959).
J. A. Pople, W. G. Schneider, and H. J. Bernstein. High Resolution Nuclear Magnetic Resonance Spectroscopy, Academic Press, New York, 1969.
H. Booth. Applications of hydrogen nuclear magnetic resonance to the conformational analysis of cyclic compounds. Prog. NMR Spectrosc. 5:149–153 (1969).
E. W. Garbisch and M. G. Griffen. Proton coupling constants in cyclohexane. J. Am. Chem. Soc. 90:6543–6544 (1968).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
DiMeglio, C.M., Froimowitz, M. & Makriyannis, A. Conformational Analysis of the Opioid Phenylmorphan and Its 9α-Methyl Analogue in Solution Using High-Resolution Nuclear Magnetic Resonance Spectroscopy. Pharm Res 10, 1200–1205 (1993). https://doi.org/10.1023/A:1018928604461
Issue Date:
DOI: https://doi.org/10.1023/A:1018928604461