Abstract
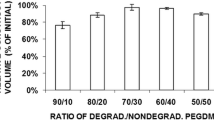
The limited supply of cartilage tissue with appropriate sizes and shapes needed for reconstruction and repair has stimulated research in the area of hydrogels as scaffolds for cartilage tissue engineering. In this study we demonstrate that poly(ethylene glycol) (PEG)-based semi-interpenetrating (sIPN) network hydrogels, made with a crosslinkable poly(ethylene glycol)-dimethacrylate (PEGDM) component and a non-crosslinkable interpenetration poly(ethylene oxide) (PEO) component, and seeded with chondrocytes support cartilage construct growth having nominal thicknesses of 6 mm and relatively uniform safranin-O stained matrix when cultured statically, unlike constructs grown with prefabricated macroporous scaffolds. Even though changing the molecular weight of the PEO from 100 to 20 kDa reduces the viscosity of the precursor polymer solution, we have demonstrated that it does not appear to affect the histological or biochemical characteristics of cartilaginous constructs. Extracellular matrix (ECM) accumulation and the spatial uniformity of the ECM deposited by the embedded chondrocytes decreased, and hydrogel compressive properties increased, as the ratio of the PEGDM:PEO in the hydrogel formulation increased (from 30:70 to 100:0 PEGDM:PEO). Total collagen and glycosaminoglycan contents per dry weight were highest using the 30:70 PEGDM:PEO formulation (24.4±3.5% and 7.1±0.9%, respectively). The highest equilibrium compressive modulus was obtained using the 100:0 PEGDM:PEO formulation (0.32±0.07 MPa), which is similar to the compressive modulus of native articular cartilage. These results suggest that the versatility of PEG-based sIPN hydrogels makes them an attractive scaffold for tissue engineering of cartilage.
© 2001 Kluwer Academic Publishers
Similar content being viewed by others
References
J. A. Buckwalter and H. J. Mankin, Arthritis Rheum. 41 (1998) 1331-1342.
L. J. Soslowsky, G. A. Ateshian and V. C. Mow, in “Biomechanics of Diarthrodial Joints”, edited by V. C. Mow, A. Ratcliffe and S. L. Woo (Springer-Verlag, New York, 1990) pp. 243-268.
G. A. Ateshian and L. J. Soslowsky, in “Basic Orthopaedic Biomechanics”, edited by V. C. Mow and W. C. Hayes (Lippincott-Raven, Philadelphia, 1997) pp. 253-273.
L. E. Freed, G. Vunjak-Novakovic and R. Langer, J. Cell. Biochem. 51 (1993) 257-264.
L. E. Freed, J. C. Marquis, G. Vunjak-Novakovic, J. Emmanual and R. Langer, Biotechnol. Bioeng. 43 (1994) 605-614.
J. Elisseeff, K. Anseth, D. Sims, W. Mcintosh, M. Randolph and R. Langer, Proc. Natl. Acad. Sci. 96 (1999) 3104-3107.
A. T. Metters, K. S. Anseth and C. N. Bowman, Biomed. Sci. Instrum. 35 (1999) 33-38.
A. B. Saim, Y. Cao, Y. Weng, C. N. Chang, M. A. Vacanti, C. A. Vacanti and R. D. Eavey, Laryngoscope 110 (2000) 1694-1697.
P. H. Corkhill, A. S. Trevett and B. J. Tighe, Proc. Inst. Mech. Eng. [H] 204 (1990) 147-155.
J. Clowacki, E. Trepman and J. Folkman, Proc. Soc. Exp. Biol. Med. 172 (1983) 93-98.
Y. Hiraizumi, E. E. Transfeldt, E. Fujimaki and M. Nambu, Spine 20 (1995) 2272-2277.
H. J. Hauselmann, M. B. Aydelotte, B. L. Schumacher, K. E. Kuettner, S. H. Gitelis and E. J. Thonar, Matrix 12 (1992) 116-129.
M. Demoor-Fossard, M. Boittin, F. Redini and J. P. Pujol, Mol. Cell Biochem. 199 (1999) 69-80.
M. D. Buschmann, Y. A. Gluzband, A. J. Grodzinsky and E. B. Hunziker, J. Cell Sci. 108 (1995) 1497-1508.
K. T. Paige, L. G. Cima, M. J. Yaremchuk, B. L. Schloo, J. P. Vacanti and C. A. Vacanti, Plast. Reconstr. Surg. 97 (1996) 168-178.
J. L. Van Susante, P. Buma, G. J. Van Osch, D. Versleyen, P. M. Van Der Kraan, W. B. Van Der Berg and G. N. Homminga, Acta Orthop. Scand. 66 (1995) 549-556.
P. Bulpitt and D. Aeschlimann, J. Biomed. Mater. Res. 47 (1999) 152-169.
G. M. Cruise, D. S. Scharp and J. A. Hubbell, Biomaterials 19 (1998) 1287-1294.
Y. Inada, M. Furukawa, H. Sasaki, Y. Kodera, M. Hiroto, H. Nishimura and A. Matsushima, Trends Biotechnol. 13 (1995) 86-91.
J. Eelisseeff, W. McINTOSH, K. ANSETH, S. RILEY, P. RAGAN and R. Langer, J. Biomed. Mater. Res. 51 (2000) 164-171.
J. Elisseef, K. Anseth, D. Sims, W. McINTOSH, M. RANDOLPH, M. YAREMCHUK and R. Langer, Plast. Reconstr. Surg. 104 (1999) 1014-1022.
D. B. Brewer, Proc. Roy. Soc. Med. 44 (1951) 561.
R. E. Schreiber and A. Ratcliffe, in “Methods in Molecular Biology, vol. 139: Extracellular Matrix Protocols”, edited by C. Streuli and M. Grant, (Humana Press, Inc., Totowa, NJ, 2000).
Y. J. Kim, R. L. Sah, J. Y. Doong and A. J. Grodzinsky, Anal. Biochem. 174 (1988) 168-176.
R. W. Farndale, D. J. Buttle and A. J. Barrett, Biochim. Biophys. Acta 883 (1986) 173-177.
J. F. Woessner, Arch. Biochem. Biophys. 93 (1961) 440.
R. E. Schreiber, N. S. Dunkelman, G. Naughton and A. Ratcliffe, Ann. N.Y. Acad. Sci. 875 (1999) 398-404.
R. M. Schinagl, D. Gurskis, A. C. Chen and R. L. Sah, J. Orthop. Res. 15 (1997) 499-506.
M. K. Kwan, W. M. Lai and V. C. Mow, J. Biomech. 23 (1990) 145-155.
S. J. Bryant and K. Anseth, Biomaterials 22 (2001) 619-626.
L. E. Freed, A. P. Hollander, I. Martin, J. R. Barry, R. Langer and G. Vunjak-Novakovic, Exp. Cell Res. 240 (1998) 58-65.
P. J. Flory, “Principles of polymer chemistry” (Cornell University Press Ithaca, NY, 1953).
E. W. Merrill, K. A. Dennison and C. Sung, Biomaterials 14 (1993) 1117-1126.
T. Canal and N. A. Peppas, J. Biomed. Mater. Res. 23 (1989) 1183-1193.
K. S. Anseth, C. N. Bowman and L. Brannon-Peppas, Biomaterials 17 (1996) 1647-1657.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Riley, S.L., Dutt, S., de la Torre, R. et al. Formulation of PEG-based hydrogels affects tissue-engineered cartilage construct characteristics. Journal of Materials Science: Materials in Medicine 12, 983–990 (2001). https://doi.org/10.1023/A:1012817317296
Issue Date:
DOI: https://doi.org/10.1023/A:1012817317296