Abstract
Purpose. To demonstrate the applicability of thermally stimulated current (TSC) spectrometry for the detection of low levels of the amorphous phase in crystalline pharmaceutical materials.
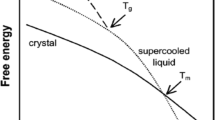
Methods. A crystalline drug substance was melt quenched to produce an amorphous material. Blends of the crystalline and amorphous phases in different ratios (from 75:25 to 99:01) were prepared by serial dilution. TSC studies were performed by applying an electric field at a temperature above the glass transition temperature (Tg) to orient the dipoles, rapidly cooling to 0°C, short circuiting for 1 min, and scanning at 7°C/min to measure the depolarization current. The temperature of the peak in the spectrum corresponds to the Tg of the amorphous phase. Modulated differential scanning calorimtery (DSC) studies were performed using three different test protocols (varying linear heating rate, modulation amplitude, and time period). Powder X-ray diffraction (XRD) studies were performed using a Siemens D500 diffractometer.
Results. The ability to detect the amorphous phase by powder XRD is beset with problems due to indirect inference, orientation effects, and instrument-related intensity variations. Even using a consistent sampling procedure and an internal standard, the XRD could quantify the amorphous phase at a level of 5%. In the conventional or modulated DSC, the amorphous phase manifests itself as a shift in the baseline. Using modulated DSC it was possible to detect the amorphous phase at a level of 5% when tested at a heating rate of 2°C/min and an amplitude of ±1.0°C with a period of 30 s. The moisture sorption method appears to have a similar detection capability. In TSC scans, the glass transition event due to molecular/segmental mobility in the amorphous phase was manifested as a peak/shoulder on the low-temperature side of the depolarization peak of the crystalline phase. The amorphous phase was unambiguously detected at 2% with a lower detection limit of ∼1%.
Conclusions. On the basis of the results of this preliminary investigation, TSC appears to be capable of detecting the amorphous phase at as low as ∼1% in crystalline pharmaceuticals, thus offering a much needed capability in discerning factors.
Similar content being viewed by others
REFERENCES
R. Huttenrauch, S. Fricke, and P. Zieke. Mechanical activation of pharmaceutical systems. Pharm. Res. 2:302-306 (1985).
A. Florence and E. Salole. Changes in crystallinity and solubility on comminution of digoxin and observations on spironolactone and oestradiol. J. Pharm. Pharmac. 28:637-642 (1976).
W. L. Chiou and L. E. Kyle. Differential thermal, solubility and aging studies on various sources of digoxin and digoxin powder: Biopharmaceutical implications. J. Pharm. Sci. 68:1224-1229 (1979).
B. C. Hancock and G. Zografi. Characteristics and significance of the amorphous state in pharmaceutical systems. J. Pharm. Sci. 86:1-12 (1997).
E. Y. Shalaev and G. Zografi. How does residual water affect the solid state degradation of drugs in the compacted state? J. Pharm. Sci. 85:1137-1141 (1996).
A. Salek-Gerhardt, C. Ahlneck, and G. Zagrafi. Assessment of disorder in crystalline solids. Int. J. Pharm. 101:237-247 (1994).
K. Yoshino, T. Sakai, Y. Yamamoto, and Y. Inuishi. Thermally stimulated current in polyacetylene. Jpn. J. Appl. Phys. 20:867-876 (1981).
G. Teyssedre, S. Mezghani, A. Bernes, and C. Lacabanne. Thermally stimulated currents of polymers. In P. Hedvig (ed.), Dielectric Spectroscopy of Polymeric Materials, Adam Hilger Ltd., Bristol, 1997, pp. 227-258.
C. Bucci and R. Fieschi. Ionic thermoconductivity: Method for the investigation of polarization in insulators. Phys. Rev. Lett. 12:16-19 (1964).
N. Fagegaltier, A. Lamure, C. Lacabanne, A. Caron, H. Mifsud, and M. Bauer. Thermal analysis of amorphous phase in a pharmaceutical drug. J. Therm. Anal. 48:459-464 (1997).
B. C. Hancock, S. L. Shamblin, and G. Zografi. Molecular mobility of amorphous pharmaceutical solids below their glass transition temperatures. Pharm. Res. 12:799-806 (1995).
S. P. Duddu and T. D. Sokoloski. Dielectric analysis in the characterization of amorphous pharmaceutical solids. 1. Molecular mobility in poly(vinylpyrrolidone)-water systems in the glassy state. J. Pharm. Sci. 84:773-776 (1995).
C. Gustafsson, H. Lennholm, T. Iversen, and C. Nystrom. Comparison of solid-state NMR and isothermal microcalorimetry in the assessment of the amorphous component of lactose. Int. J. Pharm. 174:243-252 (1998).
L. S. Taylor and G. Zografi. The quantitative analysis of crystallinity using FT-Raman spectroscopy. Pharm. Res. 15:755-761 (1998).
D. B. Black and E. G. Lovering. Estimation of the degree of crystallinity in digoxin by X-ray and infrared methods. J. Pharm. Sci. 29:684-687 (1977).
N. V. Phadnis, R. K. Cavatur, and R. Suryanarayanan. Identification of drugs in pharmaceutical dosage forms by X-ray powder diffractometry. J. Pharm. Biomed. Anal. 135:929-943 (1997).
N. J. Coleman and D. Q. M. Craig. Modulated temperature differential scanning calorimetry: a novel approach to pharmaceutical thermal analysis. Int. J. Pharm. 135:13-29 (1996).
P. G. Royall, D. Q. M. Craig, and C. Doherty. Characterization of the glass transition of an amorphous drug using modulated DSC. Pharm. Res. 15:1117-1121 (1998).
M. Galop and G. L. Collins. Thermally stimulated currents observed in pharmaceutical products. Proceedings of North American Thermal Analysis Society 27th Annual Conference, Sept. 20–22, 1999, Savannah, GA.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Venkatesh, G.M., Barnett, M.E., Owusu-Fordjour, C. et al. Detection of Low Levels of the Amorphous Phase in Crystalline Pharmaceutical Materials by Thermally Stimulated Current Spectrometry. Pharm Res 18, 98–103 (2001). https://doi.org/10.1023/A:1011087012826
Issue Date:
DOI: https://doi.org/10.1023/A:1011087012826