Abstract
Circadian administration of chemotherapy has been reported to decrease toxicity and possibly enhance efficacy. Between March 1991 and December 1993, 18 evaluable patients with progressive, hormone-refractory metastatic prostate cancer were treated in this phase II trial of circadian infusion floxuridine (FUDR). The drug was delivered through a central venous catheter using a CADD-Plus computerized pump such that approximately 70% of the drug was administered between 3 and 9 p.m. and the rest (30%) was administered between 9 p.m. and 3 p.m. The dose of FUDR was 0.15 mg/kg/day × 14 days every 4 weeks. A total of 79 complete cycles was administered.
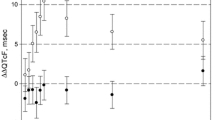
Two of 18 evaluable patients (11.1%) had decreases in PSA lasting five and eight months. No objective responses or improvement in bone scans was noted. The major toxicity observed was diarrhea. Although circadian infusion FUDR is feasible and tolerable, it has limited activity in hormone refractory prostate cancer.
Similar content being viewed by others
References
Landis SH, Murray T, Bolden S, Wingo PA: Cancer statistics, 1998. CA Cancer J Clin 48: 6-29, 1998
Torti FM, Aston D, Lum BL, Kohler M, Williams R, Spaulding JT, Shortliffe L, Freiha FS: Weekly doxorubicin in endocrine-refractory carcinoma of the prostate. J Clin Oncol 1: 477-482, 1983
Devita VT, Hellman S, Rosenberg SA (eds): Cancer: Principles and practice of oncology. 4th ed. Lippincott, 1993, p. 1107
Clarkson B, O'Connor A, Winston L, Hutchison D: The physiologic disposition of 5-FU and 5-flouro-2-deoxyuridine in man. Clin Pharmacol Ther 5: 581-610, 1964
Chabner BA: Pyrimidine antagonists. In Chabner BA (ed) Pharmacologic Principles of Cancer Therapy. WB Saunders, Philadelphia, 1982, pp 183-212
Lokich JJ, Sonneborn H, Paul S, Zipoli T: Phase I study of continuous venous infusion of floxuridine (5-FUDR) chemotherapy. Cancer Treat Rep 67: 791-793, 1983
Daher GC, Zhang RW, Soong SJ, Diasio RB: Circadian variation of fluoropyrimidine catabolic enzymes in rat liver: Possible relevance to 5-fluorodeoxyuridine chemotherapy. Drug Metab Dispos 19: 285-287, 1991
Harris BE, Song R, Soong SJ, Diasio RB: Relationship between dihydropyrimidine dehydrogenase activity and plasma 5-fluorouracil levels with evidence for circadian variation of enzyme activity and plasma drug levels in cancer patients receiving 5-fluorouracil by protracted continuous infusion. Cancer Res 50: 197-201, 1990
Von Roemeling R, Hrushesky WJM: Determination of the therapeutic index of fluxuridine by its circadian infusion pattern. J Natl Cancer Int 82: 386-393, 1990
Hrushesky WJM, Von Roemeling R, Fraley EE, Rabatin JT: Circadian-based infusional chrono-chemotherapy controls progressive metastatic renal cell carcinoma. Semin Surg Oncol 4: 110-115, 1998
Von Roemeling R, Hrushesky WJM: Circadian patterning of continuous Floxuridine infusion reduces toxicity and allows higher dose intensity in patients with widespread cancer. J Clin Oncol 7: 1710-1719, 1989
Scher HI, Kelly WK: Flutamide withdrawal syndrome: its impact on clinical trials in hormone-refractory prostate cancer. J Clin Oncol 11(8): 1566-1572, 1993
Kelly WK, Scher M, Mazumdar M, Schwartz M, Vlamis V: Prostate specific antigen (PSA) as a measure of disease outcome in metastatic hormone refractory prostate cancer (PC). Proc Am Soc Clin Oncol 11: 199, 1992
Scott WW, Gibbons RP, Johnson DE, Prout GR, Schmidt JD, Saroff J, Murphy GP: The continued evaluations of the effects of chemotherapy in patients with advanced carcinoma of the prostate. J Urol 116: 211-213, 1976
Murphy GP, Priore RL, Scardino PT: Hormone refractory metastatic prostate cancer treated with methotrexate, cyclophosphamide plus adriamycin, cis-platinum plus 5-flourouracil plus cyclophosphamide. Urol 32: 33-40, 1988
Tannock IF, Osaba D, Stockler MR, Ernst DS, Neville AJ, Moore MJ, Armitage GR, Wilson JJ, Venner PM, Coppin CM, Murphy KC: Chemotherapy with mitoxantrone plus prednisone or prednisone alone for symptomatic hormone-resistant prostate cancer: a Canadian randomized trial with palliative end points. J Clin Oncol 14: 1756-1764, 1996
Kemeny N, Daly J, Reichman B, Geller N, Botet J, Oderman P: Intrahepatic or systemic infusion of flouordeoxyuridine in patients with liver metastases from colorectal carcinoma. Ann Intern Med 107: 459-465, 1987
Kuzel MK, Tallman MS, Shevrin D, Braud E, Kilton L, Johnson P, Kozlowski J, Vozelgang NJ, Blough R, Benson AB: A phase II study of continuous infusion 5-fluoruracil in advanced hormone refractory prostate cancer. Cancer 72(6): 1965-1968, 1993
Von Roemeling R, Fisher H, DeConti R, Horton J, Wilbur H, Hrushesky W, Ruckdeschel J: Continuous FUDR infusion of hormone refractory prostate cancer. A phase I/II study. Proc Am Soc Clin Oncol 10: 171, 1991
Ohmori H, Matsumura Y, Ochi J, Kobashi K, Akebi N, Saika T, Nanba K, Tanahashi T, Josen T, Nasu Y: Phase II study of 50-DFUR treatment of the bladder and prostate cancer. Jap J Cancer Chemother 18(13): 2307-2314, 1991
Sampaio C, Olencki T, Murthy S, Budd GT, McLain D, Lorenzi V, Tuason L, Bukowski RM: Phase II trial of circadian infusion of the antimetabolite floxuridine in patients with metastatic renal cell carcinoma. J Infus Chemother 4(2): 100-103, 1994
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rajagopalan, K., Peereboom, D., Budd, G.T. et al. Phase II trial of circadian infusion floxuridine (FUDR) in hormone refractory metastatic prostate cancer. Invest New Drugs 16, 255–258 (1998). https://doi.org/10.1023/A:1006195815320
Issue Date:
DOI: https://doi.org/10.1023/A:1006195815320