Abstract
Oral piritrexim (PTX), a second generation antimetabolite, has been shown to be an active agent against methotrexate refractory transitional cell cancer (TCC) of the bladder in phase I trials. We conducted a phase II trial of this drug in patients with TCC of the bladder who failed a first line chemotherapy regimen.
Methods: Oral PTX was started at the dose of 25 mg three times per day for 5 days weekly for 3 weeks followed by one week of rest. If this was tolerated the dose was increased to 50 mg three times a day. Patients were monitored for response rate and toxicity.
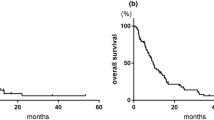
Results: Seventeen patients were entered into the trial. Two patients did not complete the required 2 courses of treatment to be evaluable. There were 13 evaluable patients. Among the 13 no one achieved a complete response (CR), however, there were 3 partial responses (PRs=RR: 23%) and 5 stable diseases (SDs). The responses lasted 2, 8 and 14 months. The major dose limiting toxicity was myelosuppression. Two patients died on treatment. One death was due to neutropenic fever and the cause of death in the second patient is thought to be a cerebral vascular accident (CVA).
Conclusion: PTX is an active drug in the treatment of TCC of the bladder. Bone marrow suppression is the most common dose limiting toxicity. In view of the observed responses and toxicities in this study and other studies, we suggest that the role of PTX be further investigated in the following clinical settings:
1. Palliative initial treatment in patients with TCC of bladder who are not candidates for more aggressive chemotherapy.
2. As first line chemotherapy in combination with other active drugs.
Similar content being viewed by others
References
Wingo PA, Tong T, Bolden S: Cancer statistics 1995. CA 45:8–30, 1995
Loehrer PJ, Einhorn LH, Elson LH, Crawford PJ, Kuebler ED, Tannock P, Raghavan I, Stuart-Harris DA, Sarosdy R, Lowe MF, Blumenstein BA, Trump D: Randomized comparison of cisplatin alone or in combination with methotrexate, vinblastine and doxorubicin in patients with metastatic urothelial carcinoma: A cooperative group study. J Clin Oncol 10:1066, 1992
Roth BJ, Bajorin DF: Advanced bladder cancer: The need to identify new agents in the post-M-VAC (methotrexate, vinblastine, doxorubicin and cisplatin. World J Urol 153:894–900, 1995
Fischer GA: Defective transport of amethopterin (methotrexate) as a mechanism of resistance to the antimetabolite in L5178y leukemia cells. Biochem Pharmacol 11:1233–1234, 1962
Flintoff WF, Davidson SV, Siminovitch L: Isolation and partial characterization of three methotrexate-resistant phenotypes from Chinese hamster ovary cells. Somatic Cell Gen 2:245–262, 1976
Galivan J: Transport and metabolism of methotrexate in normal and resistant cultured rat hepatoma cells. Cancer Res 39:735–743, 1979
Niethammer D, Jackson RC: Changes of molecular properties associated with the development of resistance against methotrexate in human lymphoblastoid cells. Eur J Cancer 11:845–854, 1975
Sirotnak FM, Moccio DM, Kelleher LE, Goutas LJ: Relative frequency and kinetic properties of transport-defective phenotypes among methotrexate-resistant L1210 clonal cell lines derived in vivo. Cancer Res 41:4447–4452, 1981
Albrecht AM, Biedier JL and Hitchinson DJ: Two different species of dihydrofolate reductase in mammalian cells differentially resistant to anwthoptetin and methasquin. Cancer Res 32:1539–1546, 1972
Alt FW, Kellems RE, Bertino JR, Schimke RT: Selective multiplication of dihydrofolate reductase genes in methotrexate-resistant variants of cultured murine cells. J Biol Chem 253:1357–1370, 1978
Hakala MT, Zakrzewski SF, Nichol CA: Relation of folic reductase to amethopterin resistance in cultured mammalian cells. J Biol Chem 236:952–958, 1961
Littlefield J: Hybridization of hamster cells with high and low folate reductase activity. Proc Natl Acad Sci USA 62:88–95, 1969
Rosowsky A, Mota CE, Queener SF, Waltham M, Ercikan-Abali E, Bertino JR: 2,4-Diamino-5-substituted-quinazolines as inhibitors of a human dihydrofolate reductase with a sitedirected mutation at position 22 and of the dihydrofolate reductases from Pneumocystis carinii and Toxoplasma gondii. J Med Chem 38(5):745–52, 1995
Flintoff WF, Essani K: Methotrexate-resistant Chinese hamster ovary cells contain a dihydrofolate reductase with an altered affinity for methotrexate. Biochemistry 19:4321–4327, 1980
Jackson RC, Hart LI, Harrap KR: Intrinsic resistance to methotrexate of cultured mamrnalian cells in relation to the inhibition kinetics of their dihydrofolate reductase. Cancer Res 36:1991–1997, 1976
Lewis WS, Cody V, Galisky N, Luft JY, Pangborn W, Chunduru SK, Spencer HT, Appleman JR, Blakely RL: Methotrexate resistant variants of human dihydrofolate reductase with substitution of leucine 22. J Biol Chem 270(10):5057–5064, 1995
Chen G, Wright JE, Rosowsky A: Dihydrofolate reductase binding and cellular uptake of nonpolyglutamatable antifolates. Mol Pharmacol 48(4):758–765, 1995
Rosenblatt DS, Whitehead VM: Methotrexate polyglutamates in cultured human cells. Adv Exp Med Biol 163:275–285, 1983
Duch DS, Edelstein MP, Bowers SW, Nichols CA: Biochemical and chemotherapeutic studies in 2,4-diamino6 (1,5-dimethoxybenzyl)-5-methyl pyfido (2,3d) pyrimidine (BW 301u). A novel lipid-soluble inhibitor of dihydrofolate reductase. Cancer Res 42:3987–3994, 1982
Sigel CW, Macklin AW, Woolley JL, Johnson NW, Collier MA, Blum MR, Clendeninn NJ, Everm BIM, Grebe G, Mackars A, Foss R, Duch DS, Bowers SW, Nichol CA: Preclinical biochemical pharmacology and toxicology of piritrexim, a lipophile inhibitor of dihydrofolate reductase. NCI Monogr 5:111–120, 1987
Taylor IW, Slowiaczek P, Friedlander ML, Tattersall MHN: Selective toxicity of a new lipophilic antifolate, BW301U, for methotrexate-resistant cells with reduced drug uptake. Cancer Res 45:978–982, 1985
Feun LG, Gonzales R, Savaraj N, Hanlon J, Collier M, Robinson WA, Clendeninn NJ: Phase II trial of piritrexim in metastatic melanoma using a intermittent, low dose administration. J Clin Oncol 9:464–467, 1991
Vokes EE, Dimery IW, Jacobs CD, Karp D, Molina A, Collier MA, Eble ML, Clendeninn NJ: A phase I study of piritrexim in combination with methotrexate in recurrent and metastatic head and neck cancer. Cancer 67:2253–2257, 1991
Schiesel JD, Carabasi M, Magill G, Casper E, Cheng E, Marks L, Feyzi J, Clendeninn NJ, Smalley R: Oral piritrexim-A phase II study in patients with advanced soft tissue sarcoma. Inv New Drugs 10:97–98, 1992
de Wit R, Kaye SB, Roberts JT, Stoter G, Scott J, Verweij J: Oral piritrexim, an effective treatment for metastatic urothelial cancer. Brit J Cancer 67:388–390, 1993
de Vries EG, Gietema JA, Workman P, Scott JE, Crawshaw A, Dobbs HT, Dennis, Mulder N, Sleijfer D, Willemse PH: A phase II and pharmacokinetic study with oral piritrexim for metastatic breast cancer. Brit J Cancer 68(3):641–644, 1993
Bleehen NM, Newman HV, Rampling RP, Ramsay JR, Roberts JT, Bedford P, Nethersell AB: A phase II study of oral piritrexim in recurrent high-grade (III, IV) glioma. Brit J Cancer 72(3):766–768, 1995
Feun LG, Savaraj N, Bandito P, Waldman S, Collier M, Glendennin N: Oral piritrexim in advanced bladder cancer: An effective drug after progression on MVAC chemotherapy? Am J Clin Oncol 17(15):448–451, 1994
Feun LG, Savaraj N, Benedetro P, Hanlon J, Sridhar KS, Collier M, Richman S, Liao SH, Clendeninn NJ: Phase I trial of piritrexim capsules using prolonged, low-dose oral administration for the treatment of advanced malignancies. J Natl Cancer Inst 83:51–55, 1991a
Laszlo J, Brenckman WD, Morgan E, Clendeninn NJ, Williams T, Currie V, Young C: Initial clinical studies of piritrexim NCI Monogr 5:121–125, 1987
Yagoda A: Chemotherapy of the urothelial tract tumors. Cancer 60:574, 1987
Troner M, Birch R, Omura GA, Williams S: Phase III comparison of cisplatin versus cisplatin doxorubicin and cyclophosphamide in the treatment of bladder (U Jrothelia) cancer: a Southeastern Cancer Study Group trial. J Urol 37(4):660–662, 1987
Soloway MS, Einstein A, Corder MP, Bonney W, Prout GR Jr, Coombs J: A comparison of cisplatin and the combination of cisplatin and cyclophosphamide in advanced urothelial cancer. A national bladder cancer collaborative group A study. Cancer 1,52(5):767–772, 1983
Hillcoat BL, Raghavan D, Matthews J, Kefford R, Yuen K, Woods R, Olver I, Bishop J, Pearson B, Coorey G: A randomized trial of cisplatin versus cisplatin plus methotrexate in advanced cancer of the urothelial tract. J Clin Oncol 7(6):706–709, 1989
Khandekar JD, Elson PJ, DeWys WD, Slayton RE, Harris DT: Comparative activity and toxicity of cis-diamminedichloroplatinum (DDP) and a combination of doxorubicin cyclophosphamide and DDP in disseminated transitional cell carcinomas of the urinary tract. J Clin Oncol 3(4):539–545, 1985
Roth BJ, Dreicer R, Einhorn LH, et al.: Significant activity of Paclitaxel in advanced transitional cell carcinoma of the urothelium: A phase II trial of the Eastern Cooperative Oncology Group (E1892). J Clin Oncol 12:2264–2270, 1994
Pollera CF, Ceribelli A, Crecco M, Calabresi F: Weekly gemcitabinc in advanced bladder cancer: A preliminary report from a phase I study. Ann Oncol 5(2):182–184, 1994
Einhorn LH, Roth BJ, Ansari R, Dreicer R, Gonin R, Loehrer PJ: Phase II trial of vinblastine, ifosfamide, and gallium combination chemotherapy in metastatic urothelial carcinoma. J Clin Oncol 12(11):2271–2276, 1994
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Khorsand, M., Lange, J., Feun, L. et al. Phase II trial of oral piritrexim in advanced, previously treated transitional cell cancer of bladder. Invest New Drugs 15, 157–163 (1997). https://doi.org/10.1023/A:1005860806635
Issue Date:
DOI: https://doi.org/10.1023/A:1005860806635