Abstract
Culturing pluripotent embryonic stem cells represents a unique model system for in vitro studies of embryo cell growth and differentiation, and represents a connection between in vitro and in vivo manipulation of genes. To further develop and refine stem cell technology for marine fish, we have established cultures of embryonic stem cells isolated from turbot blastulas. The pluripotent nature of our turbot-ES-like cells was supported by their morphology and elevated levels of alkaline phosphatase enzyme activity, their ability to remain undifferentiated for a prolonged culture period, their spontaneous differentiation potential in vitro and their ability to form embryoid bodies (EB) in response to changes in the extracellular environment. In addition, we show that turbot ES like cells express Oct-4 required for the maintenance of pluripotency of ES cells.
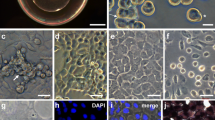
Cells from 100 blastulas (>105 cells/well) were seeded into gelatine coated 24 well cell culture clusters. The cells were polygonal in shape, with dense cytoplasm and large nuclei. The ES-like cells formed colonies within 24 h following seeding, multilayered in a pyramidal fashion, with maximum cell densities in the middle. The cells proliferated vigorously when seeding densities were high and the cells still had not attached to the gelatine-coated surface. Most of the cells became attached to the surface 48 h following seeding. Attached cells grew more slowly and 20% of the plated colonies could be kept stable for 60 days. Eventually, most of the cultures showed extensive differentiation or died. Only a few cultures (4–5%) survived prolonged culturing (>2 months). The cells were stained for alkaline phosphatase activity, a marker of pluripotency and showed intense staining. More specific, turbot ES like cells in culture expressed Oct-4, detected by immunofluorescence staining.
Changing the medium conditions by adding retinoic acid and removing LIF, the proportion of embryoid bodies in our cultures increased. ES-like cells as well as fresh, intact fertilised eggs where successfully cryopreserved. ES cells from the cryopreserved eggs could be isolated and seeded into cultures, forming colonies like the cells from freshly fertilised eggs. Also cryopreserved ES-like cells could be successfully plated. The prolonged survival of these cryopreserved cells has not yet been investigated. The establishment of in vitro cultures of turbot ES-like cells represents a new experimental model for marine flatfish.
Similar content being viewed by others
References
Auernhammer, C. J. and Melmed, S. 2000. Leukaemia-inhibitory factor-neuroimmune modulator of endocrine function. Endocrine Rev. 21: 313-345.
Bejar, J. and Hong, Y. 2002. An ES-like cell line from the mar-ine fish Sparus aurata: Characterisation and chimera production. Transgenic Research 11: 279-289.
Bost, F., Caron, L., Marchetti, I., Dani, C., Le Marchand-Brustel, Y. 2002. Retinoic acid activation of the ERK pathway is required for embryonic stem cell commitment into the adipocyte lineage. Biochem. J. 361: 621-627.
Burdon, T., Chambers, I., Niwa, H., Stracey, C. and Smith, A.G. 1999A). Signalling mechanisms regulating self-renewal and dif-ferentiation of pluripotent embryonic stem cells. Cells Tissues Organs 165: 131-143.
Burdon, T., Stracey, C., Chambers, I., Nichols, J. and Smith, A. 1999B). Suppression of SHP-2 and ERK signalling promotes self-renewal of mouse embryonic stem cells. Dev. Biol. 210: 30-43.
Chen, S-L., Sha, Z-X. and Ye, H-Q. 2002. Establishment of a pluripotent embryonic cell line from sea perch (Lateolabrax japonicus) embryos. Aquaculture 62219: 1-11.
Collodi, P., Kamei, Y., Sharps, A., Weber, D. and Barnes D. 1992. Fish embryo cell cultures for derivation of stem cells and transgenic chimeras. Mol. Mar. Biol. Biotechnol. 1: 257-265.
Doetchman, T., Williams, P., Maeda, N. 1988. Establishment of hamster blastocyst-derived embryonic stem cells. Dev. Biol. 127: 224-227.
Evans, M.J. and Kaufman, M. 1981. Establishment in culture of pluripotential cells from mouse embryos. Nature 292: 154-156.
Graves, K.H. and Moreadith, R.W. 1993. Derivation and char-acterization of putative pluripotent embryonic stem cells from preimplantation rabbit embryos. Mol. Reprod. Dev. 36: 424-433.
Hirobe, T. 1994. Effects of activators (SC-9 and OAG) and inhibitors (staurosporine and H-7) of protein kinase C and the proliferation of mouse epidermal melanoblasts in serum free medium. J. Cell. Science 107: 1679-1686.
Hong, Y., Chen, S. and Schartl, M. 2000. Embryonic stem cells in fish: current status and perspectives. Fish Physiol. Biochem. 22: 165-170.
Hong, Y. and Schartl, M. 1996. Establishment and growth responses of early medakafish (Oryzias latipes) embryonic cells in feeder layer-free cultures. Mol. Mar. Biol. Biotechnol. 5: 93-104.
Hong, Y., Winkler, C. and Schartl, M. 1998a. Production of medakafish chimeras from a stable embryonic stem cell line. Proc. Natl. Acad. Sci. USA 95: 3679-3684.
Hong, Y., Winkler, C. and Schartl, M. 1998b. Efficiency of cell cul-ture derivation from blastula embryos and of chimera formation in the medaka (Oryzias latipes) depends on donor genotype and passage number. Dev. Genes. Evol. 208: 595-602.
Iannaccone, P.M., Taborn, G.U., Garton, R.L., Caplice, M.D. and Brenin, D.R. 1994. Pluripotent embryonic stem cells from rat are capable of producing chimeras. Dev. Biol. 163: 288-292.
Iwamatswu, T. 1994. Stages of normal development in the medaka Oryzias latipes. Zool. Sci. 11: 825-839.
Kawase, E., Suemori, H., Takahashi, N., Okazaki, K. and Nakatsuji, N. 1994. Strain difference in establishment of mouse embryonic stem (ES) cell lines. Int. J. Dev. Biol. 38: 385-390.
Kirchhof, N., Carnwath, J.W., Lemme, E., Anastassiadis, K., Schøler, H. and Niemann, H. 2000. Expression pattern of Oct-4 in preimplantation embryos of different species. Biol. Reprod. 63: 1698-1705.
Kleinsmith, L.J. and Pierce, G.B. 1964. Multipotentiality of single embrional carcinoma cells. Cancer Res. 24: 1544-1549.
Ledermann, B. and Burki, K. 1991. Establishment of a germ-line competent C57BL/6 embryonic stem cell line. Exp. Cell Res. 197: 254-258.
Martin, G.R. 1981. Isolation of pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocar-cinoma stem cells. Proc. Nat. Acad. Sci. USA 78: 7634-7638.
Matsuda, T., Nakamura, T., Nakao, K., Arai, T., Katsuki, M., et al. 1999. STAT3 activation is sufficient to maintain an undif-ferentiated state of mouse embryonic stem cells. EMBO J. 18: 4261-4269.
Nakano, T., Kodama, H. and Honjo, T. 1994. Generation of lymphohematopoietic cells from embryonic stem cells in culture. Science 265: 1098-1101.
Niwa, H., Burdon, T., Chambers, I. and Smith, A.G. 1998. Self renewal of pluripotent embryonic stem cells is mediated via activation of STAT3. Genes Dev. 12: 2048-2060.
Notarianni, E., Galli, C., Laurie, S., Moor, R.M. and Evans, M.J. 1991. Derivation of pluripotent embryonic cell lines from the pig and sheep. J. Reprod. Fert. Suppl. 43: 255-260.
Pain, B., Clark, M.E., Shen, M., Nakazawa, H., Sakurai, M., Samarut, J. and Etches, R.J. 1996. Long term in vitro culture and characterisation of avian embryonic stem cells with multiple morphogenic potentialities. Development 122: 2339-2348.
Rexroad, C.E., and Powell, A.M. 1997. Culture of blastomeres from in vitro-matured, fertilised, and cultured bovine embryos. Mol. Reprod. Develop. 48: 238-245.
Robertson, E.J. 1987. In: Teratocarcinomas and Embryonic Stem Cells. A Practical Approach. pp. 71-112. Edited by E.J. Robertson. IRL Press, Oxford.
Rose-John, S. 2002. GP 130 stimulation and the maintenance of stem cells. TRENDS in Biotechnology. 20: 417-419.
Saijoh, Y., Fujii, H., Men, C., Sato, M., Hirota, Y., Nagamatsu, S., Ikeda, M. and Hamada, H. 1996. Identification of putative downstream genes Oct-3, a pluripotent cell-specific transcription factor. Genes Cells 1: 239-252.
Saito, S., Ugai, H., Sawai, K., Yamamoto, Y., Minamihashi, A., Kurosaka, K., Kobayashi, Y., Murata, T., Obata, Y. and Yp-koyama, K. 2002. Isolation of embryonic stem-like cells from equine blastocysts and their differentiation in vitro. FEBS letters 531: 389-396.
Sukoyan, M.A., Vatolin, S.Y., Golubitsa, A.N., Zhelezova, A.I., Semenova, L.A. and Serov, O.L. 1993. Embryonic stem cells derived from morulae, inner cell mass, and blastocysts of mink: Comparison of their pluripotencies. Mol. Reprod. Dev. 36: 148-158.
Sun, L., Bradford, C.S., Ghosh, C., Collodi, P. and Barnes, D.W. 1995. ES-like cell cultures derived from early zebrafish embryos. Mol. Mar. Biol. Biotechnol. 4: 193-199.
Thomson, J.A., Kalishman, J., Golos, T.G., Durning, M., Harris, C.P., Becker, R.A. and Hearn, J.P. 1995. Isolation of a primate embryonic stem cell line. Proc. Nat. Acad. Sci. USA 92: 7844-7848.
Thomson, J.A., Kalishman, J., Golos, T.G., Durning, M., Harris, C.P., Becker, R.A. and Hearn J.P. 1996. Pluripotent cell lines derived from common marmoset (Callithrix jacchus) blastocysts. Biol. Reprod. 55: 254-259.
Thomson, J.A., Itskovitz-Eldor, J., Shapiro, S.S., Waknitz, M.A., Swiergiel, J.J., Marshall, V.S. and Jones, J.M. 1998. Embryonic stem cell lines derived from human blastocysts. Science 282: 1145-1147.
Wakamutsu, Y. and Ozato, K. 1994. Establishment of a pluripo-tent cell line derived from a medaka (Oryzias latipes)blastula embryo. Mol. Mar. Biol. Biotechnol. 3: 185-191.
Wheeler, M.B. 1994. Development and validation of swine em-bryonic stem cells: a review. Repr. Fertil. Dev. 6: 563-568.
Wobus, A., Rohwedel, J., Maltsev, V., Hescheler, J. 1994. In vitro differentiation of embryonic stem cells into cardiomyocytes or skeletal muscle cells is specifically modulated by retinoic acid: Roux's Arch. Dev. Biol. 204, 36-45.
Yeom, Y.I., Fuhrmann, G., Ovitt, C.E., Brehm, A., Ohbo, K., Gross, M., Hubner, K., Schøler, H.R. 1996. Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development 122: 881-894.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Holen, E., Hamre, K. Towards obtaining long term embryonic stem cell like cultures from a marine flatfish, Scophtalmus maximus . Fish Physiology and Biochemistry 29, 245–252 (2003). https://doi.org/10.1023/B:FISH.0000045725.01192.44
Issue Date:
DOI: https://doi.org/10.1023/B:FISH.0000045725.01192.44