Abstract
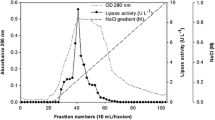
Four Thermus strains produced lipolytic activity when grown in liquid medium for 30 h at 70 °C. The highest total lipase/esterase activity (57 U l−1) was in Thermus aquaticus YT-1, followed by Thermus thermophilus HB27 and HB8 (33 and 25 U l−1, respectively), and finally by Thermus sp. (16 U l−1). Extra-cellular activity was detected in T. aquaticus YT-1 and T. thermophilus HB27 (33 and 17 U l−1). All enzymes were stable at 80 °C over 30 min, and their activity towards fatty acid esters increased as substrate chain-length diminished (i.e. hydrolysis rate was up to 6-fold higher on p-nitrophenyl caproate than on laurate).
Similar content being viewed by others
References
Belo I, Pinheiro R, Mota M (2000) Response of the thermophile Thermus sp. RQ-1 to hyperbaric air in batch and fed-batch cultivation. Appl. Microbiol. Biotechnol. 53: 517–524.
Berger JL, Lee BH, Lacroix C (1995) Identification of new enzyme activities of several strains of Thermus species. Appl. Microbiol. Biotechnol. 44: 81–87.
Cometta S, Sonnleitner B, Fiechter A (1982) The growth behaviour of Thermus aquaticus in continuous cultivation. Eur. J. Appl. Microbiol. Biotechnol. 15: 69–74.
Cowan DA (1992) Enzymes from thermophilic archaebacteria: current and future applications in biotechnology. Biochem. Soc. Sym. 58: 149–169.
Hamid NSA, Zen HB, Tein OB, Halifah YM, Saari N, Fatimah AB (2003) Screening and identification of extracellular lipase-producing thermophilic bacteria from a Malaysian hot spring. World J. Microbiol. Biotechnol. 19: 961–968.
Janssen PH, Monk CR, Morgan HW (1994) A thermophilic, lipolytic Bacillus sp., and continuous assay of its p-nitrophenylpalmitate esterase activity. FEMS Microbiol. Lett. 120: 195–200.
Kademi A, Aït-Abdelkader N, Fakhreddine L, Baratti JC (1999) A thermostable esterase activity from newly isolated moderate thermophilic bacterial strains. Enzyme Microb. Technol. 24: 332–338.
Kim HK, Park SY, Oh TK (1997) Purification and partial characterization of thermostable carboxyl esterase from Bacillus stearothermophilus L1. J. Microbiol. Biotechnol. 7: 37–42.
Lagarde D, Nguyen HK, Ravot G, Wahler D, Reymond JL, Hills G, Veit T, Lefevre F (2002) High-throughput screening of thermostable esterases for industrial bioconversions. Organic Process Res. Develop. 6: 441–445.
Pantazaki AA, Pritsa AA, Kyriadakidis DA (2002) Biotechnologically relevant enzymes from Thermus thermophilus. Appl. Microbiol. Biotechnol. 58: 1–12.
Sellek GA, Chaudhuri JB (1999) Biocatalysis in organic media using enzymes from extremophiles. Enzyme Microb. Technol. 25: 471–482.
Sharma R, Soni SK, Vohra RM, Gupta LK, Gupta JK (2002) Purification and characterisation of a thermostable alkaline lipase from a new thermophilic Bacillus sp. RSJ-1. Proc. Biochem. 37: 1075–1084.
Sigurgísladóttir S, Konrá?sdóttir M, Jónsson A, Kristjánsson JK, Matthiasson E (1993) Lipase activity of thermophilic bacteria from icelandic hot springs. Biotechnol. Lett. 15: 361–366.
Sonnleitner B, Cometta S, Fiechter A (1982) Growth kinetics of Thermus Thermophilus. Eur. J. Appl. Microbiol. Biotechnol. 15: 75–82.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Domínguez, A., Sanromán, A., Fuciños, P. et al. Quantification of intra- and extra-cellular thermophilic lipase/esterase production by Thermus sp.. Biotechnology Letters 26, 705–708 (2004). https://doi.org/10.1023/B:BILE.0000024092.27943.75
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000024092.27943.75