Abstract
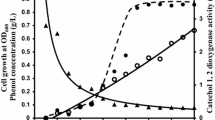
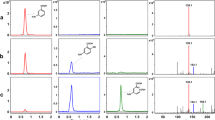
Corynebacterium glutamicum assimilated phenol, benzoate, 4-hydroxybenzoate p-cresol and 3,4-dihydroxybenzoate. Ring cleavage was by catechol 1,2-dioxygenase when phenol or benzoate was used and by protocatechuate 3,4-dioxygenase when the others were used as substrate. The locus ncg12319 of its genome was cloned and expressed in Escherichia coli. Enzyme assays showed that ncg12319 encodes a catechol 1,2-dioxygenase. This catechol 1,2-dioxygenase was purified and accepted catechol, 3-, or 4-methylcatechols, but not chlorinated catechols, as substrates. The optimal temperature and pH for catechol cleavage catalyzed by the enzyme were 30 °C and 9, respectively, and the K m and V max were determined to be 4.24 μmol l−1 and 3.7 μmol l−1 min−1 mg−1 protein, respectively.
Similar content being viewed by others
References
An H-R, Park H-J Kim E-S (2001) Cloning and expression of thermophilic catechol l,2-dioxygenase gene (catA) from Streptomyces setonii. FEMS Microbiol. Lett. 195: 17–22.
Ballou DP, Bull C (1981) Purification and properties of protocatechuate 3,4-dioxygenase from Pseudomonas putida. J. Biol. Chem. 256: 12673–12680.
Briganti F, Pessione E, Giunta C, Scozzafava A (1997) Purification, biochemical properties and substrate specificity of a catechol 1,2-dioxygenase from a phenol degrading Acinetobacter radioresistens. FEBS Lett. 416: 61–64.
Buchan A, Collier LS, Neidle EL, Moran MA (2000) Key aromaticring-cleaving enzyme, protocatechuate 3,4-dioxygenase, in the ecologically important marine Roseobacter lineage. Appl. Environ. Microbiol. 66: 4662–4672.
He Z, Li Y, Zhou P, Liu S-J (2000) Cloning and heterologous expression of a sulfur oxygenase/reductase gene from the thermoacidiophilic archaen Acidianus sp. S5 in Escherichia coli. FEMS Microbiol. Lett. 193: 217–221.
Ikeda M, Nakagawa S (2003) The Corynebacterium glutamicum genome: features and impacts on biotechnological processes. Appl. Microbiol. Biotechnol. 62: 99–109.
Itoh N, Yoshida K, Okada K (1996) Isolation and identification of styrene-degrading Corynebacterium strains, and their styrene metabolism. Biosci. Biotechnol. Biochem. 60: 1826–1830.
Iwagami SG, Yang KQ, Davies J (2000) Characterization of the protocatechuic acid catabolic gene cluster from Streptomyces sp. strain 2065. Appl. Environ. Microbiol. 66: 1499–1508.
Konopka A (1993) Isolation and characterization of subsurface bacterium that degrades aniline and methylanilines. FEMS Microbiol. Lett. 111: 93–100.
Murakami S, Kodama N, Shinke R, Aoki K (1997) Classification of catechol 1,2-dixygenase family: sequence analysis of a gene for the catechol 1,2-dioxygenase showing high specificity for methylcatechols from Gram-positive, aniline-assimilating Rhodococcus erythropolis AN-13. Gene 185: 49–54.
Parales RE, Harwood CS (1996) The ?-ketoadipate pathway and the biology of self-identity. Annu. Rev. Microbiol. 50: 553–590.
Parke D (1997) Acquisition, reorganization, and merger of genes: novel management of the ?-ketoadipate pathway in Agrobacterium tumefaciens. FEMS Microbiol. Lett. 146: 3–12.
Sikkema J, de Bont JA (1993) Metabolism of tetralin (1,2,3,4-tetrahydronaphthalene) in Corynebacterium sp. strain C125. Appl. Environ. Microbiol. 59: 567–572.
Strachan PD, Freer AA, Fewson CA (1998) Purification and characterization of catechol 1,2-dioxygenase from Rhodococcus rhodochrous NCIMB 13259 and cloning and sequencing of its catA gene. Biochem. J. 333: 741–747.
Watanabe K, Teramoto M, Futamata H, Harayama S (1998) Molecular detection, isolation and physiological characterization of functionally dominant phenol-degrading bacteria in activated sludge. Appl. Environ. Microbiol. 64: 4396–4402.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, XH.S.ZP., Liu, SJ. Functional identification of the gene locus (ncg12319 and characterization of catechol 1,2-dioxygenase in Corynebacterium glutamicum . Biotechnology Letters 26, 575–580 (2004). https://doi.org/10.1023/B:BILE.0000021958.86258.08
Issue Date:
DOI: https://doi.org/10.1023/B:BILE.0000021958.86258.08