Abstract
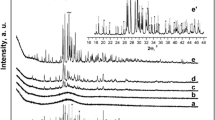
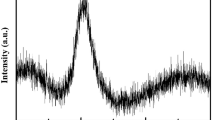
The phase evolution in a calcium phosphate glass with the molar ratio [CaO]/[P2O5] < 1, to which 6.4 mol% TiO2 and 10 mol% Al2O3 were added as nucleation agents, was studied. The results indicate that the primary phase β-Ca2P2O7 is formed during surface nucleation and crystallization which are dominant at T < 930°C. No presence of metastable calcium phosphates was registered during the formation of the primary phase. The growth of β-Ca2P2O7 crystals occurs on the faceted crystal/glass interface with dendritic morphology at a crystal growth rate that is independent of time. The kinetics of β-Ca2P2O7 crystal growth are characterised by a growth activation energy of E a = 426 ± 15 kJ/mol. The parameters of the unit cell of the β-Ca2P2O7 phase decrease with increasing temperature. The secondary TiP2O7 and AlPO4 phases are formed by volume nucleation and crystallization. The temperature of the maximum nucleation rate was determined to be T n = 690°C, and it is higher than the transformation temperature T g. In the temperature interval T < 930°C secondary phases appear during long annealing times. At T > 930°C glass volume crystallization is dominant.
Similar content being viewed by others
References
P. F. James, Y. Iqbal, U. S. Jais, S. Jordery and W. E. Lee, J. Non-Cryst. Solids 219 (1997) 17.
W. Vogel and W. HÖland, Angw.: Chem. Int. Ed. Engl. 26 (1987) 527.
M. B. ToŠiČ, M. M. MitroviĆ and R. Ž Dimitrijevic, J. Mater. Sci. 35 (2000) 3659.
M. B. ToŠiĆ, R. Ž. DimitrijeviĆ and M. M. MitroviĆ, ibid. 37 (2002) 2293.
P. F. James, ibid. 10 (1975) 1802.
R. K. Brow, J. Non-Cryst. Solids 263/264 (2000) 1.
U. Hoppe, ibid. 195 (1996) 138.
I. M. Reaney, P. F. James and W. E. Lee, J. Amer. Ceram. Soc. 79 (1996) 1934.
M. B. ToŠiĆ, M. M. MitroviĆ, R. Ž DimitrijeviĆ and N. S. BlagojeviĆ, J. Mater. Sci. 37 (2002) 4369.
R. Garney, Powd. Diff. 1 (1986) 114.
N. C. Webb, Acta Cryst. 21 (1966) 942.
S. Boudin, ibid. C 49 (1993) 2062.
S. Krumm, Mater. Sci. Forum 228–231 (1996) 183.
A. Marotta, A. Burii and F. Branda, J. Mater. Sci. 16 (1981) 341.
C. S. Ray and D. E. Day, J. Amer. Ceram. Soc. 73 (1990) 439.
E. D. Zanotto and M. C. Weinberg, Physics Chem. Glasses 19 (1989) 186.
M. C. Weinberg, J. Amer. Ceram. Soc. 74 (1991) 1905.
K. F. Kelton, ibid. 75 (1992) 2449.
A. K. Cheetam, G. Ferey and T. Loisean, Angew. Che. Ed. 38 (1999) 3268.
Y. Nan, W. E. Lee and P. F. James, J. Amer. Ceram. Soc. 75 (1992) 1641.
HSC Chemistry (Outokumpu Research Oy, Finland, 1993).
W. D. Kingery, H. K. Bowen and D. R. Uhlmann, “Introduction to Ceramics” (JohnWiley&Sons, NewYork, London, 1976).
Z. Qian, T. L. Tolt and A. R. Cooper, J. Amer. Ceram. Soc. 70 (1987) /1/49.
K. Matusita and S. Saka, Physics Chem. Glasses 20 (1979) 81.
M. C. Weinberg, J. Non-Cryst. Solids 127 (1991) 151.
K. F. Kelton, Mater. Sci. Eng. A 226–228 (1997) 142.
T. Ozawa, Polymer 12 (1971) 150.
J. W. Christian, “The Theory of Transformations in Metals and Alloys” (Pergamon Press, Oxford, 1975).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Tosić, M.B., Dimitrijević, R.Ž. & Mitrović, M.M. The crystallization of calcium phosphate glass with the ratio [CaO]/[P2O5] < 1. Journal of Materials Science 38, 1983–1994 (2003). https://doi.org/10.1023/A:1023585321475
Issue Date:
DOI: https://doi.org/10.1023/A:1023585321475