Abstract
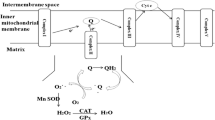
Whether we view the mitochondria as the headquarters for the leader of a crack suicide squad or as a prison for the leader of a militant coup, the role of the mitochondria in the apoptotic process is now well established. During apoptosis the integrity of the mitochondria is breeched, the mitochondrial transmembrane potential drops, the electron transport chain is disrupted, and proteins from the mitochondrial intermembrane space (MIS) such as cytochrome c are released into the cytosol, although not necessarily in that order. In the cytosol, cytochrome c forms part of a proteinaceous complex that directly activates caspase-9, one of the apical enzymes responsible for the dismantling of the cell. In this way a mitochondrial factor which is normally locked away from the rest of the cell can directly trigger apoptosis. The need to regulate the release of cytochrome c suggests that the mitochondria may be the decision center for whether a cell lives or dies. Various hypotheses have been formulated to explain how proteins of the MIS are released and how this process is regulated. These include the Bcl-2-regulated opening of a permeability transition pore or an increase in mitochondrial transmembrane potential followed by outer membrane rupture. It remains to be clarified which mitochondria specific events are essential for apoptosis and which are merely consequences of apoptosis.
Similar content being viewed by others
REFERENCES
Badley AD, Cameron DW, Kravcik S, Phenix BN, Ashby D, Kumar A, Lynch DH, Tschopp J: Dynamic correlation of apoptosis and immune activation during treatment of HIV infection. Cell Death Different 6:420–432, 1999
Ishizaki Y, Cheng L, Mudge AW, Raff MC: Programmed cell death by default in embryonic cells, fibroblasts, and cancer cells. Mol Biol Cell 6:1443–1458, 1995
Ohsako S, Elkon KB: Apoptosis in the effector phase of autoimmune diabetes, multiple sclerosis and thyroiditis. Cell Death Different 6:13–21, 1999
Roy N, Mahadevan MS, McLean M, Shutler G, Yaraghi Z, Farahani R, Baird S, Besner-Johnston A, Lefebvre C, Kang X, Salih M, Aubry H, Tamai K, Guan X, Ioannou P, Crawford T, deJong P, Surh L, Ikeda J, Korneluk R, MacKenzie A: The gene for neuronal apoptosis inhibitory protein is partially deleted in individuals with spinal muscular atrophy. Cell 80:167–178, 1995
Thompson CB: Apoptosis in the pathogenesis and treatment of disease. Science 267:1456–1462, 1995
Waterhouse P, Penninger JM, Timms E, Wakeham A, Shahinian A, Lee KP, Thompson CB, Griesser H, Mak TW: Lymphoproliferative disorders with early lethality in mice deficient in Ctla-4. Science 270:985–988, 1995
Yuan J: Transducing signals of life and death. Curr Opin Cell Biol 9:247–251, 1997
Wolvetang EJ, Johnson KL, Krauer K, Ralph SJ, Linnane AW: Mitochondrial respiratory chain inhibitors induce apoptosis. FEBS Lett 339:40–44, 1994
McCarthy NJ, Whyte MK, Gilbert CS, Evan GI: Inhibition of Ced-3/ICE-related proteases does not prevent cell death induced by oncogenes, DNA damage, or the Bcl-2 homologue Bak. J Cell Biol 136:215–227, 1997
Martin SJ, Finucane DM, Amarante-Mendes GP, O'Brien GA, Green DR: Phosphatidylserine externalization during CD95-induced apoptosis of cells and cytoplasts requires ICE/CED-3 protease activity. J Biol Chem 271:28753–28756, 1996
Watters D, Waterhouse N: Proteolytic targets in cell death. Results Probl Cell Different 24:25–44, 1998
Sakahira H, Enari M, Nagata S: Cleavage of CAD inhibitor in CAD activation and DNA degradation during apoptosis. Nature 391:96–99, 1998
Martin SJ, O'Brien GA, Nishioka WK, McGahon AJ, Mahboubi A, Saido TC, Green DR: Proteolysis of fodrin (non-erythroid spectrin) during apoptosis. J Biol Chem 270:6425–6428, 1995
Kothakota S, Azuma T, Reinhard C, Klippel A, Tang J, Chu K, McGarry TJ, Kirschner MW, Koths K, Kwiatkowski DJ, Williams LT: Caspase-3-generated fragment of gelsolin: effector of morphological change in apoptosis. Science 278:294–298, 1997
Kluck RM, Bossy-Wetzel E, Green DR, Newmeyer DD: The release of cytochrome c from mitochondria: A primary site for Bcl-2 regulation of apoptosis. Science 275:1132–1136, 1997
Kluck RM, Martin SJ, Hoffman BM, Zhou JS, Green DR, Newmeyer DD: Cytochrome c activation of CPP32-like proteolysis plays a critical role in a Xenopus cell-free apoptosis system. EMBO J 16:4639–4649, 1997
Gray MW, Burger G, Lang BF: Mitochondrial evolution. Science 283:1476–1481, 1999
Wyllie AH: What is apoptosis? Histopathology 10:995–998, 1986
Hengartner MO, Horvitz HR: Programmed cell death in Caenorhabditis elegans. Curr Opin Genet Dev 4:581–586, 1994
Medema JP, Scaffidi C, Kischkel FC, Shevchenko A, Mann M, Krammer PH, Peter ME: FLICE is activated by association with the CD95 death-inducing signaling complex (DISC). EMBO J 16:2794–2804, 1997
Muzio M, Chinnaiyan AM, Kischkel FC, O'Rourke K, Shevchenko A, Ni J, Scaffidi C, Bretz JD, Zhang M, Gentz R, Mann M, Krammer PH, Peter ME, Dixit VM: FLICE, a novel FADD-homologous ICE/CED-3-like protease, is recruited to the CD95 (Fas/APO-1) death-inducing signaling complex. Cell 85:817–827, 1996
Jacobson MD, Burne JF, King MP, Miyashita T, Reed JC, Raff MC: Bcl-2 blocks apoptosis in cells lacking mitochondrial DNA. Nature 361:365–369, 1993
Nakajima H, Golstein P, Henkart PA: The target cell nucleus is not required for cell-mediated granzyme-or Fas-based cytotoxicity. J Exp Med 181:1905–1909, 1995
Jacobson MD, Burne JF, Raff MC: Programmed cell death and Bcl-2 protection in the absence of a nucleus. EMBO J 13:1899–1910, 1994
Newmeyer DD, Farschon DM, Reed JC: Cell-free apoptosis in Xenopus egg extracts: Inhibition by Bcl-2 and requirement for an organelle fraction enriched in mitochondria. Cell 79:353–364, 1994
Hengartner MO, Horvitz HR: C. elegans cell survival gene ced-9 encodes a functional homolog of the mammalian proto-oncogene bcl-2. Cell 76:665–676, 1994
Nguyen M, Millar DG, Yong VW, Korsmeyer SJ, Shore GC: Targeting of Bcl-2 to the mitochondrial outer membrane by a COOH-terminal signal anchor sequence. J Biol Chem 268:25265–25268, 1993
Susin SA, Lorenzo HK, Zamzami N, Marzo I, Snow BE, Brothers GM, Mangion J, Jacotot E, Costantini P, Loeffler M, Larochette N, Goodlett DR, Aebersold R, Siderovski DP, Penninger JM, Kroemer G: Molecular characterization of mitochondrial apoptosis-inducing factor. Nature 397:441–446, 1999
Susin SA, Zamzami N, Castedo M, Hirsch T, Marchetti P, Macho A, Daugas E, Geuskens M, Kroemer G: Bcl-2 inhibits the mitochondrial release of an apoptogenic protease. J Exp Med 184:1331–1341, 1996
Zou H, Henzel WJ, Liu X, Lutschg A, Wang X: Apaf-1, a human protein homologous to C. elegans CED-4, participates in cytochrome c-dependent activation of caspase-3. Cell 90:405–413, 1997
Zou H, Li Y, Liu X, Wang X: An APAF-1 cytochrome c multimeric complex is a functional apoptosome that activates procaspase-9. J Biol Chem 274:11549–11556, 1999
Hu Y, Benedict MA, Ding L, NG: Role of cytochrome c and dATP/ATP hydrolysis in Apaf-1-mediated caspase-9 activation and apoptosis. EMBO J 18:3586–3595, 1999
Saleh A, Srinivasula SM, Acharya S, Fishel R, Alnemri ES: Cytochrome c and dATP-mediated oligomerization of Apaf-1 is a prerequisite for procaspase-9 activation. J Biol Chem 274:17941–17945, 1999
Stennicke HR, Deveraux QL, Humke EW, Reed JC, Dixit VM, Salvesen GS: Caspase-9 can be activated without proteolytic processing. J Biol Chem 274:8359–8362, 1999
Song Q, Kuang Y, Dixit VM, Vincenz C: Boo, a novel negative regulator of cell death, interacts with Apaf-1. EMBO J 18:167–178, 1999
Inohara N, Gourley TS, Carrio R, Muniz M, Merino J, Garcia I, Koseki T, Hu Y, Chen S, Nunez G: Diva, a Bcl-2 homologue that binds directly to Apaf-1 and induces BH3-independent cell death. J Biol Chem 273:32479–32486, 1998
Tsujimoto Y: Role of Bcl-2 family proteins in apoptosis: Apoptosomes or mitochondria? Genes Cells 3:697–707, 1998
Martinou I, Desagher S, Eskes R, Antonsson B, Andre E, Fakan S, Martinou JC: The release of cytochrome c from mitochondria during apoptosis of NGF-deprived sympathetic neurons is a reversible event. J Cell Biol 144:883–889, 1999
Moriishi K, Huang DC, Cory S, Adams JM: Bcl-2 family members do not inhibit apoptosis by binding the caspase activator Apaf-1. Proc Natl Acad Sci USA 96:9683–9688, 1999
Li H, Zhu H, Xu CJ, Yuan J: Cleavage of BID by caspase 8 mediates the mitochondrial damage in the Fas pathway of apoptosis. Cell 94:491–501, 1998
Kuwana T, Smith JJ, Muzio M, Dixit V, Newmeyer DD, Kornbluth S: Apoptosis induction by caspase-8 is amplified through the mitochondrial release of cytochrome c. J Biol Chem 273:16589–16594, 1998
Susin SA, Lorenzo HK, Zamzami N, Marzo I, Brenner C, Larochette N, Prevost MC, Alzari PM, Kroemer G: Mitochondrial release of caspase-2 and-9 during the apoptotic process. J Exp Med 189:381–394, 1999
Samali A, Cai J, Zhivotovsky B, Jones DP, Orrenius S: Presence of a pre-apoptotic complex of pro-caspase-3, Hsp60 and Hsp10 in the mitochondrial fraction of jurkat cells. EMBO J 18:2040–2048, 1999
Kohler C, Gahm A, Noma T, Nakazawa A, Orrenius S, Zhivotovsky B: Release of adenylate kinase 2 from the mitochondrial intermembrane space during apoptosis. FEBS Lett 447:10–12, 1999
Reed JC: Double identity for proteins of the Bcl-2 family. Nature 387:773–776, 1997
Crompton M: The mitochondrial permeability transition pore and its role in cell death. Biochem J 341:233–249, 1999
Huser J, Rechenmacher CE, Blatter LA: Imaging the permeability pore transition in single mitochondria. Biophys J 74:2129–2137, 1998
Reed PW: Inophores. Methods Enzymol 55:435–454, 1979
Zamzami N, Susin SA, Marchetti P, Hirsch T, Gomez-Monterrey I, Castedo M, Kroemer G: Mitochondrial control of nuclear apoptosis. J Exp Med 183:1533–1544, 1996
Marchetti P, Castedo M, Susin SA, Zamzami N, Hirsch T, Macho A, Haeffner A, Hirsch F, Geuskens M, Kroemer G: Mitochondrial permeability transition is a central coordinating event of apoptosis. J Exp Med 184:1155–1160, 1996
Hirsch T, Susin SA, Marzo I, Marchetti P, Zamzami N, Kroemer G: Mitochondrial permeability transition in apoptosis and necrosis. Cell Biol Toxicol 14:141–145, 1998
Lemasters JJ, Nieminen AL, Qian T, Trost LC, Elmore SP, Nishimura Y, Crowe RA, Cascio WE, Bradham CA, Brenner DA, Herman B: The mitochondrial permeability transition in cell death: A common mechanism in necrosis, apoptosis and autophagy. Biochim Biophys Acta 1366:177–196, 1998
Bradham CA, Qian T, Streetz K, Trautwein C, Brenner DA, Lemasters JJ: The mitochondrial permeability transition is required for tumor necrosis factor alpha-mediated apoptosis and cytochrome c release. Mol Cell Biol 18:6353–6364, 1998
Szabo I, Zoratti M: The mitochondrial permeability transition pore may comprise VDAC molecules. I. Binary structure and voltage dependence of the pore. FEBS Lett 330:201–205, 1993
Krajewski S, Tanaka S, Takayama S, Schibler MJ, Fenton W, Reed JC: Investigation of the subcellular distribution of the bcl-2 oncoprotein: Residence in the nuclear envelope, endoplasmic reticulum, and outer mitochondrial membranes. Cancer Res 53:4701–4714, 1993
Shimizu S, Narita M, Tsujimoto Y: Bcl-2 family proteins regulate the release of apoptogenic cytochrome c by the mitochondrial channel VDAC. Nature 399:483–487, 1999
Marzo I, Zamzami BC, Jurgensmeier JM, Susin SA, Vieira HL, Prevost MC, Xie Z, Matsuyama S, Reed JC, Kroemer G: Bax and adenine nucleotide translocator cooperate in the mitochondrial control of apoptosis. Science 281:2027–2031, 1998
Murphy AN, Bredesen DE, Cortopassi G, Wang E, Fiskum G: Bcl-2 potentiates the maximal calcium uptake capacity of neural cell mitochondria. Proc Natl Acad Sci USA 93:9893–9898, 1996
Andreyev AY, Fiskum G: Cytochrome c release from brain mitochondria is independent of the mitochondrial permeability transition. FEBS Lett 439:373–376, 1998
Bossy-Wetzel E, Newmeyer DD, Green DR: Mitochondrial cytochrome c release in apoptosis occurs upstream of DEVD-specific caspase activation and independently of mitochondrial transmembrane depolarization. EMBO J 17:37–49, 1998
Finucane DM, Bossy-Wetzel E, Waterhouse NJ, Cotter TG, Green DR: Bax-induced caspase activation and apoptosis via cytochrome c release from mitochondria is inhibitable by Bcl-xL. J Biol Chem 274:2225–2233, 1999
Eskes R, Antonsson B, Osen-Sand A, Montessuit S, Richter C, Sadoul R, Mazzei G, Nichols A, Martinou JC: Bax-induced cytochrome C release from mitochondria is independent of the permeability transition pore but highly dependent on Mg2+ ions. J Cell Biol 143:217–224, 1998
Finucane DM, Waterhouse NJ, Amarante-Mendes GP, Cotter TG, Green DR: Collapse of the inner mitochondrial transmembrane potential is not required for apoptosis of HL60 cells. Exp Cell Res 251:166–174, 1999
Li P, Nijhawan D, Budihardjo I, Srinivasula SM, Ahmad M, Alnemri ES, Wang X: Cytochrome c and dATP-dependent formation of Apaf-1/caspase-9 complex initiates an apoptotic protease cascade. Cell 91:479–489, 1997
Kuida K, Haydar TF, Kuan CY, Gu Y, Taya C, Karasuyama H, Su MS, Rakic P, Flavell RA: Reduced apoptosis and cytochrome c-mediated caspase activation in mice lacking caspase 9. Cell 94:325–337, 1998
Chandel NS, Schumacker PT: Cells depleted of mitochondrial DNA (rho0) yield insight into physiological mechanisms. FEBS Lett 454:173–176, 1999
La Piana G, Fransvea E, Marzulli D, Lofrumento NE: Mitochondrial membrane potential supported by exogenous cytochrome c oxidation mimics the early stages of apoptosis. Biochem Biophys Res Commun 246:556–561, 1998
Ravagnan L, Marzo I, Costantini P, Susin SA, Zamzami N, Petit PX, Hirsch F, Goulbern M, Poupon MF, Miccoli L, Xie, Z, Reed JC, Kroemer G: Lonidamine triggers apoptosis via a direct, Bcl-2-inhibited effect on the mitochondrial permeability transition pore. Oncogene 18:2537–2546, 1999
Vander Heiden MG, Chandel NS, Williamson EK, Schumacker PT, Thompson CB: Bcl-xL regulates the membrane potential and volume homeostasis of mitochondria. Cell 91:627–637, 1997
Heiden MG, Chandel NS, Schumacker PT, Thompson CB: Bcl-xL prevents cell death following growth factor withdrawal by facilitating mitochondrial ATP/ADP exchange. Mol Cell 3:159–167, 1999
Muchmore SW, Sattler M, Liang H, Meadows RP, Harlan JE, Yoon HS, Nettesheim D, Chang BS, Thompson CB, Wong SL, Ng SL, Fesik SW: X-ray and NMR structure of human Bcl-xL, an inhibitor of programmed cell death. Nature 381:335–341, 1996
Antonsson B, Conti F, Ciavatta A, Montessuit S, Lewis S, Martinou I, Bernasconi L, Bernard A, Mermod JJ, Mazzei G, Maundrell K, Gambale F, Sadoul R, Martinou JC: Inhibition of Bax channel-forming activity by Bcl-2. Science 277:370–372, 1997
Minn AJ, Velez P, Schendel SL, Liang H, Muchmore SW, Fesik SW, Fill M, Thompson CB: Bcl-x(L) forms an ion channel in synthetic lipid membranes. Nature 385:353–357, 1997
Schendel SL, Xie Z, Montal MO, Matsuyama S, Montal M, Reed JC: Channel formation by antiapoptotic protein Bcl-2. Proc Natl Acad Sci USA 94:5113–5118, 1997
Schendel SL, Azimov R, Pawlowski K, Godzik A, Kagan BL, Reed JC: Ion channel activity of the BH3 only bcl-2 family member, BID. J Biol Chem 274:21932–21936, 1999
Schendel SL, Montal M, Reed JC: Bcl-2 family proteins as ion-channels. Cell Death Different 5:372–380, 1998
Basanez G, Nechushtan A, Drozhinin O, Chanturiya A, Choe E, Tutt S, Wood KA, Hsu Y, Zimmerberg J, Youle RJ: Bax, but not Bcl-xL, decreases the lifetime of planar phospholipid bilayer membranes at subnanomolar concentrations. Proc Natl Acad Sci USA 96:5492–5497, 1999
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Waterhouse, N.J., Green, D.R. Mitochondria and Apoptosis: HQ or High-Security Prison?. J Clin Immunol 19, 378–387 (1999). https://doi.org/10.1023/A:1020550716138
Issue Date:
DOI: https://doi.org/10.1023/A:1020550716138