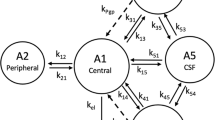
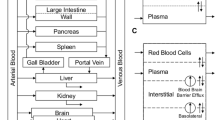
Abstract
Purpose. This study was performed to evaluate the involvement of P-glycoprotein in disposition kinetics of tacrolimus (FK506), a substrate of P-glycoprotein, in the body.
Methods. The blood and tissue concentrations of FK506 after i.v. or p.o. administration (2 mg/kg) to normal andmdrla knockout mice were measured by competitive enzyme immunoassay.
Results. The blood concentrations in knockout mice were significantly higher than those in normal mice. The value of the total clearance (CLtot) for knockout mice (19.3 mL/min/kg) was about 1/3 of that for normal mice (55.8 mL/min/kg)(P < 0.001), although there was no significant difference in the distribution volume at the steady-state (Vdss) (about 4.6 L/kg) between both types of mice. FK506 rapidly penetrated the blood-brain barrier and the brain concentration reached a maximum, which was about 10 times higher in knockout mice than in normal mice, 1 hr after administration. The brain concentration in normal mice thereafter decreased slowly, whereas in knockout mice, an extremely high concentration was maintained for 24 hr.
Conclusions. The pharmacokinetic behavior of FK506 in the tissue distribution is related with the function of P-glycoprotein encoded by themdr la gene. The brain distribution of FK506 is dominated by the P-glycoprotein-mediated drug efflux and presumably also by the binding to FK-binding proteins (immunophilins) in the brain.
Similar content being viewed by others
REFERENCES
F. Thiebaut, T. Tsuruo, H. Hamada, M. M. Gottesman, I. Pastan, and M. C. Willingham. Cellular localization of the multidrug-resistance gene product P-glycoprotein in normal human tissues. Pro. Natl. Acad. Sci. USA 84:7735-7738 (1987).
E. Georges, G. Bradley, J. Gariepy, and V. Ling. Detection of P-glycoprotein isoforms by gene-specific monoclonal antibodies. Pro. Natl. Acad. Sci. USA 87:152-156 (1990).
A. Tsuji, T. Terasaki, Y. Takabatake, Y. Tenda, I. Tamai, T. Yamashita, S. Moritani, T. Tsuruo, and J. Yamashita. P-glycoprotein as the drug efflux pump in primary cultured bovine brain capillary endothelial cells. Life Sci. 51:1427-1437 (1992).
A. De Vaut and P. Gros. Two members of the mouse mdr gene family confer multidrug resistance with overlapping but distinct drug specificities. Mol. Cell. Biol. 10:1652-1663 (1990).
P. Borst, A. H. Schinkel, J. J. M. Smit, E. Wagenaar, L. van Deemter, A. J. Smith, E. W. H. M. Eijdems, F. Baas, and G. J. R. Zaman. Classical and novel forms of multidrug resistance and the physiological functions of P-glycoprotein in mammals. Pharmac. Ther. 60:289-299 (1993).
A. H. Schinkel, J. J. M. Smit, O. van Telligen, J. H. Beijinen, E. Wagenaar, L. van Deemter, C. A. Mol, M. A. van der Valk, E. C. Robanus-Maandag, H. P. te Riele, A. J. M. Berns, and P. Brrst. Disruption of the mouse mdr1a P-glycoprotein gene leads to a deficiency in the blood-brain barrier and to increased sensitivity to drugs. Cell 77:491-502 (1994).
A. H. Schinkel, E. Wagenaar, L. van Deemter, C. A. Mol, and P. Borst. Absence of the mdr1a P-glycoprotein in mice affects tissue distribution and pharmacokinetics of dexamethasone, digoxin, and cyclosporin A. J. Clin. Invest. 96:1698-1705 (1995).
A. Tsuji and I. Tamai, I. Blood-brain barrier function of P-glycoprotein. Adv. Drug Deliv. Rev. 25:287-298 (1997).
K. Ohara, R. Billington, R. W. James, G. A. Dean, M. Nishiyama, and H. Noguchi. Toxicologic evaluation of FK506. Transplant. Proc. 22:83-86 (1990).
B. A. Appignani, R. A. Bhadelia, S. C. Blacklow, A. K. Wang, S. F. Roland, and R. B. Freeman. Neuroimaging finding in patients on immunosuppressive therapy: Experience with tacrolimus toxicity. Am. J. Roentgenol. 166:683-688 (1996).
J. Sharkey and S. P. Butcher. Immunophilins mediate the neuroprotective effects of FK506 in focal cerebral ischemia. Nature (London) 371:336-339 (1994).
S. P. Butcher, D. C. Henshall, Y. Teramura, K. Iwasaki, and J. Sharkey. Neuroprotective actions of FK506 in experimental stroke: In vivo evidence against an antiexcitotoxic mechanism. J. Neurosci. 17:6939-6946 (1997).
M. Naito, T. Ohhara, A. Yamazaki, T. Danki, and T. Tsuruo. Reversal of multidrug resistance by an immunosuppressive agent FK506. Cancer Chemother. Pharmacol. 29:195-200 (1992).
T. Saeki, K. Ueda, Y. Tanigawara, R. Hori, and T. Komano. Human P-glycoprotein transports cyclosporine A and FK506. J. Biol. Chem. 267:6077-6080 (1993).
M. Kobayashi, K. Tamura, N. Katayama, K. Nakamura, K. Nagase, K. Hane, T. Tutumi, M. Niwa, H. Tanaka, K. Iwasaki, and M. Kohsaka. FK506 assay past and present — characteristics of FK506 ELISA. Transplant. Proc. 23:2725-2729 (1991).
K. Yamaoka, T. Nakagawa, and T. Uno. Moment analysis for disposition kinetics of several cephalosporin antibiotics in rats. J. Pharm. Pharmacol. 35:19-22 (1983).
R. G. Blasberg, J. D. Fenstermacher, and C. S. Patlak. Transport of α-aminoisobutyric acid across brain capillary and cellular membranes. J. Cereb. Blood Flow Metab. 3:8-32 (1983).
U. Christians, C. Kruse, R. Kownatzki, H. M. Schiebel, R. Schwinzer, M. Sattler, R. Schottmann, A. Linck, V. M. F. Almeida, F. Braun, F., and K. Fr. Sewing. Measurement of FK506 by HPLC and isolation and characterization of its metabolites. Transplant. Proc. 23:940-941 (1991).
T. Shiraga, H. Matsuda, K. Nagase, K. Iwasaki, K. Noda, H. Yamazaki, T. Shimada, and Y. Funae. Metabolism of FK506, a potent immunosappressive agent, by cytochrome P450 3A enzymes in rat, dog, and human liver microsomes. Biochem. Pharmacol. 47:7275 (1994).
A. M. Alak. Measurement of tacrolimus (FK506) and its metabolites: A review of assay development and application in therapeutic drug monitoring and pharmacokinetic studies. Ther. Drug Monitor. 19:338-351 (1997).
A. M. Alak and S. Moy. Biological activity of tacrolimus (FK506) and its metabolites from whole blood of kidney transplant patients. Transplant. Proc. 29:2487-2490 (1997).
K. Iwasaki, T. Shiraga, K. Nagase, K. Hirano, K. Nozaki, and K. Noda. Pharmacokinetic study of FK506 in the rat. Transplant. Proc. 23:2757-2759 (1991).
K. Iwasaki, T. Shiraga, H. Matsuda, Y. Teramura, A. Kawamura, T. Hata, S. Ninimiya, and Y. Esumi. Absorption, distribution, metabolism and excretion of tacrolimus (FK506) in the rat. Xenobio. Metabol. Dispos. 13:259-265 (1998).
R. Venkataramanan, A. Swaminatha, T. Prasad, A. Jain, S. Zuckerman, V. Warty, J. McMichael, J. Lever, G. Burckart, and T. Starzl. Clinical pharmacokinetics of tacrolimus. Clin. Pharmacokinet. 29:404-430 (1995).
N. Takeguchi, K. Ichimura, M. Koike, W. Matsui, T. Kashiwagura, and K. Kawahara. Inhibition of the multidrug efflux pump in isolated hepatocyte couplets by immunosuppressants FK506 and cyclosporine. Transplantation 55:646-650 (1993).
F. Ichimura, K. Yokogawa, T. Yamana, A. Tsuji, and Y. Mizukami. Physiological pharmacokinetic model for pentazocine. I. Tissue distribution and elimination in the rat. Int. J. Pharm. 15:321-333 (1983).
J. Liu, J. D. Farmer, W. S. Lane, J. Friedman, I. Weissman, and S. L. Schreiver. Calcineurin is a common target of cyclophillin-cyclosporin A and FKBP-FK506 complexes. Cell 66:807-815 (1991).
J. P. Steiner, T. M. Dawson, M. Fotuhi, C. E. Glatt, A. M. Snowman, N. Cohen, and S. H. Snyder. High brain densities of the immunophilin FKBP colocalized with calcineurin. Nature (London) 358:584-587 (1992).
M. Asami, T. Kuno, H. Mukai, and C. Tanaka. Detection of the FK506-FKBP-calcineurin complex by a simple binding assay. Biochem. Biophys. Res. Commun. 192:1388-1394 (1993).
T. Terao, E. Hisanaga, Y. Sai, I. Tamai, and A. Tsuji. Active secretion of drugs from the small intestinal epithelium in rats by P-glycoprotein functioning as an absorption barrier. J. Pharm. Pharmacol. 48:1083-1089 (1996).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yokogawa, K., Takahashi, M., Tamai, I. et al. P-Glycoprotein-Dependent Disposition Kinetics of Tacrolimus: Studies in mdr la Knockout Mice. Pharm Res 16, 1213–1218 (1999). https://doi.org/10.1023/A:1018993312773
Issue Date:
DOI: https://doi.org/10.1023/A:1018993312773