Abstract
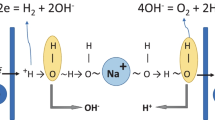
Chronopotentiometry is used as a tool to obtain detailed information on the transport behaviour of the bipolar membrane BP-1 in solutions of high sodium chloride concentration above the limiting current density. We discuss critically the interpretation of the observed transition times. The occurrence of two such polarization times for low to moderate current densities is explained by the membrane asymmetry: the two membrane layers of opposite charge in general have different transport properties such as co-ion concentration and diffusion coefficient. The reversible and irreversible contributions to the transmembrane potential can be distinguished which allows the bipolar membrane energy requirements to be addressed. The experiments indicate that the increased voltage drop across bipolar membranes observed with higher solution concentrations can be explained on the basis of stronger concentration gradients in the membrane layers. The gradients become stronger with increased current density, but here the ohmic resistance under steady state transport conditions (the transport resistance) contributes to the increasing electrical potential. The transport resistance decreases with increasing current density due to the ion-exchange of the salt counter ions with the water splitting products. The experiments show that bipolar membranes should be operated at low current densities and low concentrations to minimize energy requirements. These findings are in contrast to the high current densities required to reduce impurities in the produced acid and base.
Similar content being viewed by others
References
G. Pourcelly, in A.J.B. Kemperman (Ed.), ‘Handbook on Bipolar Membrane Technology’, (Twente University Press, Enschede, Netherlands, 2000), chapter 2.
F.G. Wilhelm, I. Pünt, N.F.A. van der Vegt, M. Wessling and H. Strathmann, J. Membr. Sci. 182 (2001) 13.
V. Cauwenberg, J. Peels, S. Resbeut and G. Pourcelly, ‘Application of Electrodialysis within Fine Chemistry,’ Book of Abstracts, Vol. 1, Euromembrane 1999, 19–22 Sept., Leuven, Belgium (1999), p. 109.
F.G. Wilhelm, N.F.A. van der Vegt, M. Wessling and H. S trathmann, J. Electroanal. Chem. 502 (2001) 152.
H-W. Rösler, F. Maletzki and E. Staude, J. Membr. Sci. 72 (1992) 171.
H-W. Rösler, PhD thesis, Universität GH Essen, Germany (1991).
J.J. Krol, M. Wessling and H. Strathmann, J. Membr. Sci. 162 (1999) 155.
N.P. Gnusin, V.I. Zabolotskii, N.V. Shel'deshov and N.D. Krikunova, English trans. Elektrokhimiya 16 (1980) 49.
J.O'M. Bockris, A.K.N. Reddy, ‘Modern Electrochemistry 1 – Ionics’, 2nd edn (Kluwer Academic/Plenum Publishers, New York 1998).
F.G. Wilhelm, PhD thesis, Universiteit Twente, Enschede (2001).
N.V. Shel'deshov, N.P. Gnusin, V.I. Zabolotskii, N.D. Pis'menskaya, English trans. Elektrokhimiya, 21 (1985) 152.
W. Neubrand PhD thesis, Univeristät Stuttgart, Germany (1999) (also published by Logos, Berlin, 1999).
W. Neubrand and G. Eigenberger, ‘Equilibrium and transport properties of CMX and AMX membranes in aqueous solutions of NaCl and HCl’ Poster printout and Book of Abstracts, Euromembrane, June 1997, University of Twente, Enschede, The Netherlands (1997).
F.G. Wilhelm, N.F.A. van der Vegt, M. Wessling and H. Strathmann, in A.J.B. Kemperman (Ed.), ‘Handbook on Bipolar Membrane Technology’ (Twente University Press, Enschede, Netherlands, 2000), Chapter 4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wilhelm, F., van der Vegt, N., Strathmann, H. et al. Current–voltage behaviour of bipolar membranes in concentrated salt solutions investigated with chronopotentiometry. Journal of Applied Electrochemistry 32, 455–465 (2002). https://doi.org/10.1023/A:1016317503435
Issue Date:
DOI: https://doi.org/10.1023/A:1016317503435