Abstract
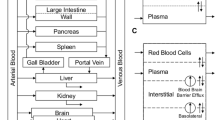
Brain delivery of active anti-HIV compounds is important for successful treatment of the AIDS patient. As an initial step in predicting human brain drug concentrations, hybrid pharmacokinetic models were developed to characterize the disposition of anti-HIV nucleosides following parent and prodrug administrations in mice. Mouse data were obtained following intravenous administration of 3′-azido-2′,3′-dideoxyuridine (AZddU or AZDU), 3′-azido-3′-deoxythymidine (AZT), and their dihydropyridine prodrugs (AZddU-DHP and AZT-DHP). Exponential equations were fitted to the serum concentration–time data for each species, including the pyridinium ion moieties, and subsequently used in differential mass balance equations describing the brain dynamics of each compound. Model parameters for the mass balance equations were estimated by various techniques, including the utilization of in vitro data. In general, model-predicted brain concentrations agreed with the observed data. Similar data in larger animals will permit scale-up of the current model to predict human brain drug concentrations.
Similar content being viewed by others
REFERENCES
A. Brunetti, G. Berg, G. Di Chiro, R. M. Cohen, R. Yarchoan, P. A. Pizzo, S. Broder, J. Eddy, M. J. Fulham, R. D. Finn, and S. M. Larson. Reversal of brain metabolic abnormalities following treatment of AIDS dementia complex with 3′-azido-2′,3′-dideoxythymidine (AZT, Zidovudine): A PET-FDG study. J. Nucl. Med. 30:581–590 (1989).
P. F. Torrence, J. Kinjo, K. Leisak, J. Balzarini, and E. De Clercq. AIDS Dementia: Synthesis and properties of a derivative of 3′-azido-3′-deoxythymidine (AZT) that may become “locked” in the central nervous system. FEBS Lett. 234:135–140 (1988).
E. Palomino, D. Kessel, and J. P. Horwitz. A dihydropyridine carrier system for sustained delivery of 2′,3′-dideoxynucleo-sides to the brain. J. Med. Chem. 32:622–625 (1989).
J. M. Gallo, F. D. Boudinot, K. J. Doshi, J. T. Etse, V. S. Bhadti, R. F. Schinazi, and C. K. Chu. Evaluation of brain targeting of anti-HIV nucleosides delivered via dihydropyridine prodrugs. Pharm. Res. 6:S161 (1989).
R. J. Lutz, R. L. Dedrick, and D. S. Zaharko. Physiological pharmacokinetics: An in vivo approach to membrane transport. Pharmacol. Ther. 11:559–592 (1980).
K. Doshi, J. M. Gallo, F. D. Boudinot, R. F. Schinazi, and C. K. Chu. Comparative pharmacokinetics of 3′-azido-3′-deoxythymidine (AZT) and 3′-azido-2′,3′-dideoxyuridine (AZddU) in mice. Drug Metab. Dispos. 17:590–594 (1989).
NONLIN84, Statistical Consultants, Inc., Lexington, KY, 1985.
L. E. Gerlowski and R. K. Jain. Physiologically based pharmacokinetic modeling: Principles and applications. J. Pharm. Sci. 72:1103–1127 (1983).
Y. Sasaki and H. N. Wagner, Jr. Measurement of the distribution of cardiac output in unanesthetized rats. J. Appl. Physiol. 30:879–884 (1971).
P. A. Altman and D. S. Dittmer. Respiration and Circulation, Federation of American Societies for Experimental Biology, Bethesda, MD, 1971.
J. M. Gallo, F. C. Lam, and D. G. Perrier. Area method for the estimation of partition coefficients for physiological pharmacokinetic models. J. Pharmacokin. Biopharm. 15:271–280 (1987).
SIMUSOLV, Dow Chemical Co., Midland, MI, 1986.
J. Mordenti. Pharmacokinetic scale-up: Predictions of human pharmacokinetic profiles from animal data. J. Pharm. Sci. 74:1097–1099 (1985).
N. Bodor. Redox drug delivery systems for targeting drugs to the brain. In R. L. Juliano (ed.), Biological Approaches to the Controlled Delivery of Drugs, New York Academy of Sciences, New York, 1987, pp. 289–306.
T. P. Zimmerman, W. B. Mahony, and K. L. Prus. 3′-Azido-3′-deoxythymidine. An unusual nucleoside that permeates the membrane of human erythrocytes and lymphocytes by nonfacilitated diffusion. J. Biol. Chem. 262:5748–5754 (1987).
J. Balzarini, R. Pauwels, M. Baba, P. Herdewijn, E. De Clercq, S. Broder, and D. G. Johns. The in vitro and in vivo anti-retrovirus activity, and intracellular metabolism of 3′-azido-2′,3′-dideoxythymidine and 2′,3′-dideoxycytidine are highly dependent on the cell species. Biochem. Pharmacol. 37:897–903 (1988).
R. Spector, A. Z. Spector, and S. R. Snodgrass. Model for transport in the central nervous system. Am. J. Physiol. 232:R73–R79 (1977).
J. M. Collins and R. L. Dedrick. Distributed model for drug delivery to CSF and brain tissue. Am. J. Physiol. 245:R303–R310 (1983).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gallo, J.M., Etse, J.T., Doshi, K.J. et al. Hybrid Pharmacokinetic Models to Describe Anti-HIV Nucleoside Brain Disposition Following Parent and Prodrug Administration in Mice. Pharm Res 8, 247–253 (1991). https://doi.org/10.1023/A:1015808624103
Issue Date:
DOI: https://doi.org/10.1023/A:1015808624103