Abstract
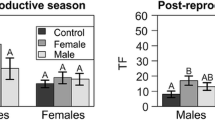
Experimental tests were conducted to determine whether females and males of the tree-dwelling lizard Liolaemus tenuis (Tropiduridae) show intraspecific chemical recognition during breeding and postreproductive seasons. Animals were individually maintained in plastic enclosures for one week. Thereafter, the number of tongue-flicks that a lizard performed in the enclosure of a male, a female, its own home enclosure, and a control (unused) enclosure were recorded. In both seasons, males and females made fewer tongue-flicks in their home enclosures than in any other one, indicating a recognition of a familiar place, probably a chemical self-recognition. Conspecific chemical recognition was season dependent. During the post-reproductive season, lizards tongue-flicked at similar rates in conspecific and control enclosures, while during the breeding season enclosures of females elicited more tongue-flicks by both sexes, and the overall tongue-flick rates were higher than in the postreproductive season. Results are discussed within the context of the social system of the species.
Similar content being viewed by others
REFERENCES
Alberts, A. C. 1989. Ultraviolet visual sensitivity in desert iguanas: Implications for pheromone detection. Anim. Behav. 38:129-137.
Alberts, A. C. 1992. Pheromonal self-recognition in desert iguanas. Copeia 1992:229-232.
Alberts, A. C. 1993. Chemical and behavioral studies of femoral glands secretions in iguanid lizards. Brain Behav. Ecol. 41:255-260.
Copper, W. E. 1994. Chemical discrimination by tongue-flicking in lizards: A review with hypothesis on its origin and its ecological and phylogenetic relationships. J. Chem. Ecol. 20:439-487.
Cooper, W. E. 1995. Foraging models, prey chemical discrimination, and phylogeny in lizards. Anim. Behav. 50:973-985.
Cooper, W. E. 1998. Evaluation of swab and related tests as a bioassay for assessing responses by Squamate reptiles to chemical stimuli. J. Chem. Ecol. 24:841-866.
Cooper, W. E., and Burghardt, G. M. 1990a. A comparative analysis of scoring methods for chemical discrimination of prey by squamate reptiles. J. Chem. Ecol. 16:45-65.
Cooper, W. E., and Burghardt, G. M. 1990b. Vomerolfaction and vomodor. J. Chem. Ecol. 16:103-105.
Cooper, W. E., and Garstka, W. R. 1987. Lingual responses to chemical fractions of urodaeal glandular pheromones of the skink Eumeces laticipes. J. Exp. Zool. 242:249-253.
Cooper, W. E., and Trauth, S. E. 1992. Discrimination of conspecific male and female cloacal chemical stimuli by males and possession of a probable pheromone gland by females in a cordylid lizard, Gerrhosaurus nigrolineatus. Herpetologica 48:229-236.
Cooper, W. E., Garstka, W. R., and Vitt, L. J. 1986. Female sex pheromones in the lizard Eumeces laticeps. Herpetologica 42:361-366.
Cooper, W. E., LÓpez, P., and Salvador, A. 1994. Pheromone detection by an amphisbaenian. Anim. Behav. 47:1401-1411.
De Fazio, A., Simon, C. A., Middendorf, G. A., and Romano, D. 1977. Iguanid substrate licking: A response to novel situations in Sceloporus jarrovi. Copeia 1977:706-709.
De Perno, C. S., and Cooper, W. E. 1995. Prey chemical discrimination and strike-induced chemosensory searching in the lizard Liolaemus zapallarensis. Chemoecology 4:86-72.
Donoso-Barros, R. 1966. Reptiles de Chile. Universidad de Chile, Santiago, 458 pp.
Duvall, D., Graves, B. M., and Carpenter, G. C. 1987. Visual and chemical composite signalling effects of Sceloporus lizards fecal boli. Copeia 1987:1028-1031.
Edwards, A. L. 1968. Experimental Design in Psychological Research, 3rd ed. Holt, Rinehart, and Winston, New York.
Etheridge, R. 1995. Redescription of Ctenoblepharys adspersa Tschudi, 1845, and the taxonomy of Liolaeminae (Reptilia: Squamata: Tropiduridae). Am. Mus. Novitates 3142:1-34.
Frost, D. R., and Etheridge, R. 1989. A phylogenetic analysis and taxonomy of iguanian lizards (Reptilia: Squamata). Univ. Kans. Mus. Nat. Hist. Mis. Publ. 81.
GÓmez, A., Font, E., and Desfilis, E. 1993. Chemoreception in the Lacertidae: Exploration and conspecific discrimination in the Spanish wall lizard, Podarcis hispanica, pp. 213-230, in E. D. Valakos, W. Böhme, V. Pérez-Mellando, and P. Maragou (eds.). Region: A Biological Approach, Lacetids of the Mediterranean, Hellenic Zoological Society, Athens.
Graves, B. M., and Halpern, M. 1991. Discrimination of self from conspecific chemical cues in Tiliqua scincinoides (Sauria: Scincidae). J. Herpetol. 25:125-126.
Greenberg, N. 1985. Exploratory behavior and stress in the lizard Anolis carolinensis. Z. Tierpsychol. 70:89-102.
Halpern, M. 1992. Nasal chemical senses in reptiles: Structure and function, pp. 423-524, in C. Gans and D. Crews (eds.). Hormones, Brain and Behavior. Biology of Reptilia, Vol. 18.E. The University of Chicago Press, Chicago.
Labra, A. 1998. Selected body temperatures of seven Liolaemus lizards. Rev. Chil. Hist. Nat. 71:349-358.
Maderson, P. F. A. 1972. The structure and evolution of holocrine epidermal glands in sphaerodactyline and eublepharine gekkonid lizards. Copeia 1972:559-571.
Manzur, M. I., and Fuentes, E. R. 1979. Polygyny and agonistic behavior in the tree-dwelling lizard Liolaemus tenuis (Iguanidae). Behav. Ecol. Sociobiol. 6:23-28.
Mason, R. T. 1992. Reptilian pheromones, pp. 114-228, in C. Gans and D. Crews (eds.). Hormones, Brain and Behavior. Biology of Reptilia, Vol. 18E. The University of Chicago Press, Chicago.
Mason, R. T., and Gutzke, W. 1990. Sex recognition in the Leopard gecko, Eublepharis macularis (Sauria: Gekkonidae); Possible mediation by skin-derived semiochemicals. J. Chem. Ecol. 16:27-36.
Moore, M. C., and Lindzey, J. 1992. The physiological basis of sexual behavior in males reptiles, pp. 70-113, in C. Gans and D. Crews (eds.). Hormones, Brain and Behavior. Biology of Reptilia, Vol. 18E. The University of Chicago Press, Chicago.
Steele, L. J., and Cooper, W. E. 1997. Investigations of pheromonal discrimination between conspecific individuals by male and female Leopard geckos (Eublepharis macularis). Herpetologica 53:475-484.
Werner, D. I., Baker, E. M., GonzÁlez, E del C., and Sosa, I. R. 1987. Kinship recognition and grouping in hatchling green iguanas. Behav. Ecol. Sociobiol. 21:83-89.
Whittier, J. M., and Tokarz, R. R. 1992. Physiological regulation of sexual behavior in female reptiles, pp. 24-69, in C. Gans and D. Crews (eds.). Hormones, Brain and Behavior. Biology of Reptilia, Vol. 18E. The University of Chicago Press, Chicago.
Zar, J. H. 1984. Biostatical Analysis. Prentice-Hall International, Englewood Cliffs, New Jersey, 718 pp.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Labra, A., Niemeyer, H.M. Intraspecific Chemical Recognition in the Lizard Liolaemus tenuis . J Chem Ecol 25, 1799–1811 (1999). https://doi.org/10.1023/A:1020925631314
Issue Date:
DOI: https://doi.org/10.1023/A:1020925631314