Abstract
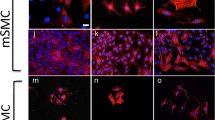
Smooth muscle cells (SMC) and endothelial cells (EC) play a pivotal role in arteriogenesis and atherosclerosis. We evaluated the role of EC on the growth of SMC and neonatal cardiomyocytes (NEO) by using serum-free EC-supernatant (AoCM). Five percent fetal calf serum was used in order to mimic growth effects of blood. EC and SMC purities were 99% as determined by absence or presence of markers such as CD31, desmin, α-smooth muscle actin and tropomyosin using immunostaining and FACS analysis. AoCM markedly influenced the morphology of NEO as determined by α-actinin staining but showed only little effect on the phenotype of SMC. Protein synthesis after 2 days increased 2.5-fold in SMC and 3.7-fold in NEO as determined by tritium incorporation. The values for serum (2.8 and 2.3-fold, respectively) were comparable. The induction of DNA-synthesis by serum in NEO was twice that of AoCM (3.9-fold). However, proliferative effects of serum and AoCM on SMC differed markedly: Serum induced a 66-fold increase in DNA-synthesis resulting in a 54% higher cell number. DNA-synthesis after AoCM treatment lead to a nonsignificant small increase and no proliferation was detected. Platelet derived growth factor (PDGF-AB), present in blood, induced a 47-fold increase in DNA-synthesis and a 38% increase in cell number. Our data suggest that EC in the absence of physical forces exert strong morphogenic effects on cardiomyocytes but they lack specific effects on smooth muscle cells. In vessels EC might function as a border to isolate SMC from key regulators in blood such as PDGFs.
Similar content being viewed by others
References
Schaper W, Piek JJ, Munoz-Chapuli R, Wolf C, Ito W: Collateral circulation of the heart. In: J.A. Ware, M. Simons (eds). Angiogenesis and Cardiovascular Disease. Oxford University Press, New York, Oxford, 1999, pp 159-198
Kubin T, Ando H, Scholz D, Bramlage P, Kostin S, van Veen A, Hein S, Fischer S, Breier A, Schaper J, Schaper W: Microvascular endothelial cells remodel cultured adult cardiomyocytes and increase survival. Am J Physiol 45: H2179-H2187, 1999
Ando H, Kubin T, Schaper W, Schaper J: Porcine coronary microvascular endothelial cells express α-smooth muscle actin and show low NOS III activity. Am J Physiol 45: H1755-H1768, 1999
Smirnov VN, Orekhov AN: Smooth muscle cells from adult human aorta. In: H.M. Piper (ed). Cell Culture Techniques in Heart and Vessel Research. Springer, Berlin, 1990, pp 271-289
Shah AM: Paracrine modulation of heart cell function by endothelial cells. Cardiovasc Res 31: 847-867, 1996
Elsässer A, Schlepper M, Klövekorn WP, Cai WJ, Zimmermann R, Müller KD, Strasser R, Kostin S, Gagel C, Münkel B, Schaper W, Schaper J: Hibernating myocardium. Circulation 96: 2920-2931, 1997
Schwartz SM, deBlois D, O'Brien ERM: The intima-soil for atherosclerosis and restenosis. Circ Res 77: 445-465, 1995
Owens GK: Regulation of differentiation of vascular smooth muscle cells. Physiol Rev 75: 487-517, 1995
Kassab GS, Rider CA, Tang NJ, Fung YCB: Morphometry of pig coronary arterial trees. Am J Physiol 265: H350-H365, 1993
Kassab GS, Fung YCB: Topology and dimensions of pig coronary capillary network. Am J Physiol 267: H319-H325, 1994
Shay JW, Wright WE: Hayflick, his limit, and cellular aging. Nature Rev 1: 72-76, 2000
Hughes AD, Clunn GF, Refson J, Demoliou-Mason C: Platelet-derived growth factor (PDGF): Actions and mechanisms in vascular smooth muscle. Gen Pharmac 27: 1079-1089, 1996
Bowen-Pope DF, Hart CE, Seifert RA: Sera and conditioned media contain different isoforms of platelet-derived growth factor (PDGF) which bind to different classes of PDGF receptor. J Biol Chem 264: 2502-2508, 1989
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kubin, T., Vogen, S., Wetzel, J. et al. Porcine aortic endothelial cells show little effects on smooth muscle cells but are potent stimulators of cardiomyocyte growth. Mol Cell Biochem 242, 39–45 (2003). https://doi.org/10.1023/A:1021177326151
Issue Date:
DOI: https://doi.org/10.1023/A:1021177326151