Abstract
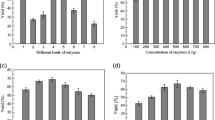
Lipases (E.C. 3.1.1.3) are biological catalysts widely used in industry due to their versatility, selectivity and specificity. For this reason, the interest in their purification has shown great potential. A technique employed in enzymes recovery capable of preserving their biological activity is partitioning using aqueous two-phase systems. Therefore, the objective of this work was to evaluate the partitioning of lipase extracted from pequi seed in aqueous two-phase systems composed of PEG 4000 or 2-propanol with ammonium sulfate and water at 298 K and 308 K. Lipase partitioning in aqueous two-phase systems was statistically analyzed using the F test and comparison of means test, showing a significant effect (P < 0.05) for the interaction between temperature and type of system, as well as the mutual influence of both parameters on each other in the response variables (protein partition coefficient, enzyme partition coefficient, enzyme recovery, selectivity and purification factor). It was verified that the enzyme had a preference for the salt-rich phase (bottom phase) and that the ATPS composed of 2-propanol and ammonium sulfate with crude extract samples at 298 K obtained the best partition results (yield of 86.39%, selectivity of 0.057 and purification factor of 4.48) under the experimental conditions.
Graphic abstract


Similar content being viewed by others
References
Abreu DCA, Figueiredo KC (2017) Separação de proteínas de uma solução salina por membrana de diálise de acetato de celulose. J Eng Exact Sci 3:1023–1033. https://doi.org/10.18540/jcecvl3iss7pp1023-1033
Albertsson PA (1971) Partition of cell particles and macromolecules. Wiley, New York
Angajala G, Pavan P, Subashini R (2016) Lipases: an overview of its current challenges and prospectives in the revolution of biocatalysis. Biocatal Agric Biotechnol 7:257–270. https://doi.org/10.1016/j.bcab.2016.07.001
Anvari M (2015) Extraction of lipase from Rhizopus microsporus fermentation culture by aqueous two-phase partitioning. Biotechnol Biotechnol Equip 29:723–731. https://doi.org/10.1080/13102818.2015.1042406
Barbosa JMP, Souza RL, Fricks AT, Zanin GM, Soares CMF, Lima AS (2011) Purification of lipase produced by a new source of Bacillus in submerged fermentation using an aqueous two-phase system. J Chromatogr B 879:3853–3858. https://doi.org/10.1016/j.jchromb.2011.10.035
Bolinelli RC, Guldvik IJ, Gronberg H, Wiklund F, Mills IG, Thiede B (2015) A differential protein solubility approach for the depletion of highly abundant proteins in plasma using ammonium sulfate. Analyst 140:8109–8117. https://doi.org/10.1039/c5an01560j
Borrelli G, Trono D (2015) Recombinant lipases and phospholipases and their use as biocatalysts for industrial applications. Int J Mol Sci 16:20774–20840. https://doi.org/10.3390/ijms160920774
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye biding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Casas-Godoy L, Gasteazoro F, Duquesne S, Bordes F, Marty A, Sandoval G (2018) Lipases: an overview. Methods Mol Biol 1835:3–38. https://doi.org/10.1007/978-1-4939-8672-9_1
Chaiwut P, Rawdkuen S, Benjakul S (2010) Extraction of protease from Calotropis procera latex by polyethylene glycol-salts biphasic system. Process Biochem 45:1148–1155. https://doi.org/10.1016/j.procbio.2010.04.007
Chong KY, Stefanova R, Zhang J, Brooks MSL (2020) Aqueous two-phase extraction of bioactive compounds from haskap leaves (Lonicera caerulea): comparison of salt/ethanol and sugar/propanol systems. Sep Purif Technol 252:1–12. https://doi.org/10.1016/j.seppur.2020.117399
Coelho DF, Silveira E, Pessoa A Jr, Tambourgi EB (2013) Bromelain purification through unconventional aqueous two-phase system (PEG/ammonium sulphate). Bioprocess Biosyst Eng 36:185–192. https://doi.org/10.1007/s00449-012-0774-5
Fekete S, Guillarme D, Sandra P, Sandra K (2016) Chromatographic, electrophoretic, and mass spectrometric methods for the analytical characterization of protein biopharmaceuticals. Anal Chem 88:480–507. https://doi.org/10.1021/acs.analchem.5b04561
Gandolfi ORR, Gonçalves GRF, Batista IC, Fontan RCI, Veloso CM, Bonomo RCF (2020) Aqueous two-phase system (polyethylene glycol + ionic liquid) for extraction of α-amylase: phase diagrams, systems characterization and partition study. Braz J Chem Eng 37:595–606. https://doi.org/10.1007/s43153-020-00043-9
Gonçalves Filho D, Silva AG, Guidini CZ (2019) Lipases: sources, immobilization methods, and industrial applications. Appl Microbiol Biotechnol 103:7399–7423. https://doi.org/10.1007/s00253-019-10027-6
González-Amado M, Rodil E, Arce A, Soto A, Rodríguez O (2016) The effect of temperature on polyethylene glycol (4000 or 8000) e (sodium or ammonium) sulfate aqueous two phase systems. Fluid Phase Equilib 428:95–101. https://doi.org/10.1016/j.fluid.2016.06.019
Jiang B, Feng Z, Liu C, Xu Y, Li D, Ji G (2015) Extraction and purification of wheat-esterase using aqueous two-phase systems of ionic liquid and salt. Int J Food Sci Technol 52:2878–2885. https://doi.org/10.1007/s13197-014-1319-5
Kapoor M, Gupta MN (2012) Lipase promiscuity and its biochemical applications. Process Biochem 47:555–569. https://doi.org/10.1016/j.procbio.2012.01.011
Khayati G, Gholitabar A (2016) Liquid-liquid equilibrium of hydrophilic alcohols with three different salts of chloride: experiment and correlation. J Chem Eng Data 61:1454–1461. https://doi.org/10.1021/acs.jced.5b00771
Lehninger AL, Nelson DL, Cox MM (2006) Princípios de bioquímica de Lehninger,4th edn. Sarvier, São Paulo
Luechau F, Ling TC, Lyddiatt A (2010) Two-step process for initial capture of plasmid DNA and partial removal of RNA using aqueous two-phase systems. Process Biochem 45:1432–1436. https://doi.org/10.1016/j.procbio.2010.05.015
Mathiazakan P, Shing SY, Ying SS, Kek HK, Tang MSY, Show PL, Ooi CW, Ling TC (2016) Pilot-scale aqueous two-phase floatation for direct recovery of lipase derived from Burkholderia cepacia strain ST8. Sep Purif Technol 171:206–213. https://doi.org/10.1016/j.seppur.2016.07.017
Mazzeu CJ, Ramos EZ, Da Silva Cavalcanti MH, Hirata DB, Virtuoso LS (2015) Partitioning of Geotrichum candidum lipase from fermentative crude extract by aqueous two-phase system of polyethylene glycol and sodium citrate. Sep Purif Technol 156:158–164. https://doi.org/10.1016/j.seppur.2015.09.069
Mistry SL, Kaul A, Merchuk JC, Asenjo JA (1996) Mathematical modelling and computer simulation of aqueous two-phase continuous protein extraction. J Chromatogr A 741:151–163. https://doi.org/10.1016/0021-9673(96)00179-3
Muneeruddin K, Nazzaro M, Kaltashov IA (2015) Characterization of intact protein conjugates and biopharmaceuticals using ion-exchange chromatography with online detection by native electrospray ionization mass spectrometry and top-down tandem mass spectrometry. Anal Chem 87:10138–10145. https://doi.org/10.1021/acs.analchem.5b02982
Nandini KE, Rastogi NK (2011) Liquid-liquid extraction of lipase using aqueous two-phase system. Food Process Technol 4:295–303. https://doi.org/10.1007/s11947-008-0160-0
Nascimento PA, Alves AN, dos Santos KA, Veloso CM, Santos LS, Fontan RCI, Sampaio VS, Bonomo RCF (2021) Optimization of lipase extraction from pequi seed (Caryocar brasiliense Camb.). J Food Process Preserv. https://doi.org/10.1111/jfpp.15616
Nian R, Zhang W, Tan L, Lee J, Bi X, Yang Y, Gan HT, Gagnon P (2016) Advance chromatin extraction improves capture performance of protein A affinity chromatography. J Chromatogr A 1431:1–7. https://doi.org/10.1016/j.chroma.2015.12.044
Ooi CW, Tey BT, Hii SL, Kamal SMM, Lan JCW, Ariff A, Ling TC (2009) Purification of lipase derived from Burkholderia pseudomallei with alcohol/salt-based aqueous two-phase systems. Process Biochem 44:1083–1108. https://doi.org/10.1016/j.procbio.2009.05.008
Padilha GS, Osório WR (2019) Economic method for extraction/purification of a Burkholderia cepacia lipase with potential biotechnology application. Appl Biochem Biotechnol. https://doi.org/10.1007/s12010-019-03041-x
Padilha CEA, Oliveira Júnior SD, Souza DFS, Oliveira JA, Macedo GR, Santos ES (2016) Baker’s yeast invertase purification using aqueous two phase system—modeling and optimization with PCA/LS-SVM. Food Bioprod Process 101:157–165. https://doi.org/10.1016/j.fbp.2016.11.004
Passos H, Trindade MP, Vaz TSM, da Costa LP, Freire MGF, Coutinho JAP (2013) The impact of self-aggregation on the extraction of biomolecules in ionic-liquid-based aqueous two-phase systems. Sep Purif Technol 108:174–180. https://doi.org/10.1016/j.seppur.2013.02.008
Pastore GM, Costa VSR, Koblitz MGB (2003) Purificação parcial e caracterização bioquímica de lipase extracelular produzida por nova linhagem de Rhizopus sp. Ciênc Tecnol Aliment 23:135–140. https://doi.org/10.1590/S0101-20612003000200006
Picó A, Colavizza D, Benaissa M, Maes P, Tartar A, Montreuil J, Spik G (2006) Poly(ethylene glycol) molar mass and polydispersivity effect on protein partitioning in aqueous two-phase systems. J Chromatogr B 830:286–292. https://doi.org/10.1016/j.jchromb.2005.11.008
Ramakrishnan V, Goveas LC, Suralikerimath N, Jampani C, Halami PM, Narayan B (2016) Extraction and purification of lipase from Enterococcus faecium MTCC5695 by PEG/phosphate aqueous-two phase system (ATPS) and its biochemical characterization. Biocatal Agric Biotechnol 6:19–27. https://doi.org/10.1016/j.bcab.2016.02.005
Reis IAO, Santos SB, Pereira FDS, Sobral CRS, Freire MG, Freitas LS, Soares CMF, Lima AS (2014) Extraction and recovery of rutin from acerola waste using alcohol-salt-based aqueous two-phase systems. Sep Sci Technol 49:656–663. https://doi.org/10.1080/01496395.2013.860461
Rodrigues RC, Virgen-Ortíz JJ, Santos JCS, Berenguer-Murcia A, Alcantara AR, Barbosa O, Ortiz C, Fernandez-Lafuente R (2019) Immobilization of lipases on hydrophobic supports: immobilization mechanism, advantages, problems, and solutions. Biotechnol Adv 37:746–770. https://doi.org/10.1016/j.biotechadv.2019.04.003
Sanglard MG, Farias FO, Sosa FHB, Santos TPM, Igarashi-Mafra L, Mafra MR (2018) Measurement and correlation of aqueous biphasic systems composed of alcohol (1-propanol/2-propanol/tert-butanol) + (NH4)2SO4 + H2O at 298 K and a textile dye partition. Fluid Phase Equilib 466:7–13. https://doi.org/10.1016/j.fluid.2018.03.009
Sankaran R, Show PL, Yap YJ, Lam HL, Ling TC, Pan GT, Yang TCK (2018) Sustainable approach in recycling of phase components of large scale aqueous two-phase flotation for lipase recovery. J Clean Prod 184:938–948. https://doi.org/10.1016/j.jclepro.2018.02.301
Selvakumar P, Ling TC, Walker S, Lyddiatt A (2010) A practical implementation and exploitation of ATPS for intensive processing of biological feedstock: a novel approach for heavily biological feedstock loaded ATPS. Sep Purif Technol 75:323–331. https://doi.org/10.1016/j.seppur.2010.08.022
Senske M, Smith AE, Pielak GJ (2016) Protein stability in reverse micelles. Angew Chem 128:3650–3653. https://doi.org/10.1002/ange.201508981
Show PL, Ooi CW, Anuar MS, Ariff A, Yusof YA, Chen SK, Anuar MSM, Ling TC (2013) Recovery of lipase derived from Burkholderia cenocepacia ST8 using sustainable aqueous two-phase flotation composed of recycling hydrophilic organic solvent and inorganic salt. Sep Purif Technol 110:112–118. https://doi.org/10.1016/j.seppur.2013.03.018
Show PL, Ling TC, Lan JCW, Tey BT, Ramanan RN, Yong ST, Ooi CW (2015) Review of microbial lipase purification using aqueous two-phase systems. Curr Org Chem 19:19–29
Soodam K, Guinee TP (2018) The case for milk protein standardisation using membrane filtration for improving cheese consistency and quality. Int J Dairy Technol 71:1–15. https://doi.org/10.1111/1471-0307.12502
Souza RL, Lima RA, Coutinho JAP, Soares CMF, Lima AS (2015) Aqueous two-phase systems based on cholinium salts and tetrahydrofuran and their use for lipase purification. Sep Purif Technol 155:118–126. https://doi.org/10.1016/j.seppur.2015.05.021
Tan Z, Li F, Zhao C, Teng Y, Liu Y (2017) Chiral separation of mandelic acid enantiomers using an aqueous two-phase system based on a thermo-sensitive polymer and dextran. Sep Purif Technol 172:382–387. https://doi.org/10.1016/j.seppur.2016.08.039
Wang Y, Yan Y, Hu S, Han J, Xu X (2010) Phase diagrams of ammonium sulfate + ethanol/1-propanol/2-propanol + water aqueous two-phase systems at 298.15 K and correlation. J Chem Eng Data 55:876–881. https://doi.org/10.1021/je900504e
Winkler UK, Stuckmann M (1979) Glycogen, hyaluronate, and some other polysaccharides greatly enhance the formation of exolipase by Serratia marcescens. J Bacteriol 3:663–670. https://doi.org/10.1128/jb.138.3.663-670.1979
Yagmurov ER, Kozlov GV, Pushkarev MA (2017) Lipase purification: the review of conventional and novel methods. J Hyg Eng Des 20:60–69
Acknowledgements
The authors gratefully acknowledge the financial support provided for this work by Foundation for Research Support of Bahia State (FAPESB) and Coordination for the Improvement of Higher Education Personnel (CAPES, finance code 001) agencies of Brazil.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nascimento, P.A., Alves, A.N., dos Santos, K.A. et al. Partitioning of pequi seed (Caryocar brasiliense Camb.) lipase in aqueous two-phase systems composed of PEG/2-propanol + ammonium sulfate + water. Braz. J. Chem. Eng. 38, 957–965 (2021). https://doi.org/10.1007/s43153-021-00154-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43153-021-00154-x