Abstract
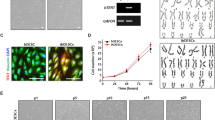
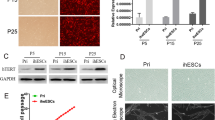
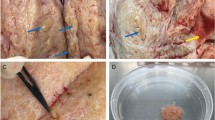
Endometrial-like stromal cells, one of the main components of endometriotic lesions, are an important in vitro model for studying cellular and molecular mechanisms associated with lesion development in endometriosis. However, the short life span of primary endometriotic stromal cells (Ec-ESCs) limits their use. Human telomerase reverse transcriptase (hTERT) plasmids can be used to develop immortalized cell lines. Here we aimed to establish an endometriotic stromal cell line by hTERT immortalization. Primary Ec-ESCs were obtained from a human ovarian endometriotic cyst. The purity was assessed by morphology and the expression of vimentin, cytokeratin, and human interferon-inducible transmembrane protein 1 (hIFITM1). Cells were infected with hTERT lentiviral vector and selected with hygromycin. hTERT mRNA levels were confirmed by RT-qPCR. Immortalized Ec-ESCs (iEc-ESCs) were characterized by examining the expression of morphological markers and key genes of interest, TP53, estrogen receptor β (ERβ), progesterone receptor (PR), and steroidogenic factor-1 (SF-1). Karyotyping and in vitro decidualization studies were also performed. Ec-ESCs were positive for vimentin and hIFITM1 and negative for cytokeratin, indicating that they were representative of Ec-ESC. The fibroblast-like morphology, expression of TP53, ERβ, PR, and SF-1 did not change before and after hTERT immortalization. iEc-ESCs showed an impaired decidualization response like primary Ec-ESCs when compared to normal eutopic stromal cells. Karyotyping showed that 15/19 cells had normal female karyotype, while 4/19 cells had partial trisomy 11q. Collectively, we successfully established and characterized an immortalized endometriotic stromal cell line. It is potentially useful as an in vitro experimental model to investigate endometriosis biology.





Similar content being viewed by others
Data Availability
Not applicable.
References
Giudice LC. Clinical practice. Endometriosis. N Engl J Med. 2010;362(25):2389–98.
Hsu AL, Khachikyan I, Stratton P. Invasive and noninvasive methods for the diagnosis of endometriosis. Clin Obstet Gynecol. 2010;53(2):413–9.
Farquhar C. Endometriosis. Bmj. 2007;334(7587):249–53.
Ballard KD, Seaman HE, de Vries CS, Wright JT. Can symptomatology help in the diagnosis of endometriosis? Findings from a national case-control study—part 1. Bjog. 2008;115(11):1382–91.
Ness RB. Endometriosis and ovarian cancer: thoughts on shared pathophysiology. Am J Obstet Gynecol. 2003;189(1):280–94.
Sugihara K, Kobayashi Y, Suzuki A, Tamura N, Motamedchaboki K, Huang CT, et al. Development of pro-apoptotic peptides as potential therapy for peritoneal endometriosis. Nat Commun. 2014;5:4478.
Sampson JA. Peritoneal endometriosis due to the menstrual dissemination of endometrial tissue into the peritoneal cavity. Am J Obstet Gynecol. 1927;14:422–69.
Noble LS, Takayama K, Zeitoun KM, Putman JM, Johns DA, Hinshelwood MM, et al. Prostaglandin E2 stimulates aromatase expression in endometriosis-derived stromal cells. J Clin Endocrinol Metab. 1997;82(2):600–6.
Bulun SE, Cheng YH, Yin P, Imir G, Utsunomiya H, Attar E, et al. Progesterone resistance in endometriosis: link to failure to metabolize estradiol. Mol Cell Endocrinol. 2006;248(1–2):94–103.
Klemmt PA, Carver JG, Kennedy SH, Koninckx PR, Mardon HJ. Stromal cells from endometriotic lesions and endometrium from women with endometriosis have reduced decidualization capacity. Fertil Steril. 2006;85(3):564–72.
Shen L, Yang S, Huang W, Xu W, Wang Q, Song Y, et al. MicroRNA23a and microRNA23b deregulation derepresses SF-1 and upregulates estrogen signaling in ovarian endometriosis. J Clin Endocrinol Metab. 2013;98(4):1575–82.
Hayashi A, Tanabe A, Kawabe S, Hayashi M, Yuguchi H, Yamashita Y, et al. Dienogest increases the progesterone receptor isoform B/A ratio in patients with ovarian endometriosis. J Ovarian Res. 2012;5(1):31.
Fazleabas AT, Brudney A, Gurates B, Chai D, Bulun S. A modified baboon model for endometriosis. Ann N Y Acad Sci. 2002;955:308–17 discussion 40–2, 96–406.
Joshi N, Su R-W, Fazleabas A. Gene expression in endometriosis. In: Kwak-Kim J, editor. Endometrial gene expression: an emerging paradigm for reproductive disorders. Cham: Springer International Publishing; 2020. p. 159–80.
Fazleabas AT. A baboon model for inducing endometriosis. Methods Mol Med. 2006;121:95–9.
Harirchian P, Gashaw I, Lipskind ST, Braundmeier AG, Hastings JM, Olson MR, et al. Lesion kinetics in a non-human primate model of endometriosis. Hum Reprod. 2012;27(8):2341–51.
Bruner-Tran KL, Mokshagundam S, Herington JL, Ding T, Osteen KG. Rodent models of experimental endometriosis: identifying mechanisms of disease and therapeutic targets. Curr Womens Health Rev. 2018;14(2):173–88.
Zeitvogel A, Baumann R, Starzinski-Powitz A. Identification of an invasive, N-cadherin-expressing epithelial cell type in endometriosis using a new cell culture model. Am J Pathol. 2001;159(5):1839–52.
Boccellino M, Quagliuolo L, Verde A, La Porta R, Crispi S, Piccolo MT, et al. In vitro model of stromal and epithelial immortalized endometriotic cells. J Cell Biochem. 2012;113(4):1292–301.
Redwine DB. Ovarian endometriosis: a marker for more extensive pelvic and intestinal disease. Fertil Steril. 1999;72(2):310–5.
Arnold JT, Kaufman DG, Seppala M, Lessey BA. Endometrial stromal cells regulate epithelial cell growth in vitro: a new co-culture model. Hum Reprod. 2001;16(5):836–45.
Joshi NR, Miyadahira EH, Afshar Y, Jeong JW, Young SL, Lessey BA, et al. Progesterone resistance in endometriosis is modulated by the altered expression of MicroRNA-29c and FKBP4. J Clin Endocrinol Metab. 2017;102(1):141–9.
Sun H, Fukuda S, Hirata T, Arakawa T, Ma S, Neriishi K, et al. IFITM1 is a novel, highly sensitive marker for endometriotic stromal cells in ovarian and extragenital endometriosis. Reprod Sci 2019:1933719119831782.
Yilmaz BD, Bulun SE. Endometriosis and nuclear receptors. Hum Reprod Update. 2019;25(4):473–85.
Chen Y, Yi Y, Han H, Cui H, Shen D, Wei L, Cheng H, et al. Establishment of human endometriosis-derived immortalized eutopic endometrium stromal and epithelial cell lines. Int J Clin Exp Med. 2016;9(8):16450–8.
Holdsworth-Carson SJ, Colgrave EM, Donoghue JF, Fung JN, Churchill ML, Mortlock S, et al. Generation of immortalized human endometrial stromal cell lines with different endometriosis risk genotypes. Mol Hum Reprod. 2019;25(4):194–205.
Toouli CD, Huschtscha LI, Neumann AA, Noble JR, Colgin LM, Hukku B, et al. Comparison of human mammary epithelial cells immortalized by simian virus 40 T-antigen or by the telomerase catalytic subunit. Oncogene. 2002;21(1):128–39.
Liu X, Roberts J, Dakic A, Zhang Y, Schlegel R. HPV E7 contributes to the telomerase activity of immortalized and tumorigenic cells and augments E6-induced hTERT promoter function. Virology. 2008;375(2):611–23.
Körner M, Burckhardt E, Mazzucchelli L. Higher frequency of chromosomal aberrations in ovarian endometriosis compared to extragonadal endometriosis: a possible link to endometrioid adenocarcinoma. Mod Pathol. 2006;19(12):1615–23.
Han SJ, Jung SY, Wu SP, Hawkins SM, Park MJ, Kyo S, et al. Estrogen receptor beta modulates apoptosis complexes and the inflammasome to drive the pathogenesis of endometriosis. Cell. 2015;163(4):960–74.
Jiang L, Zhang M, Wang S, Han Y, Fang X. Common and specific gene signatures among three different endometriosis subtypes. PeerJ. 2020;8:e8730.
Liu X, Zhang Q, Guo SW. Histological and ommunohistochemical characterization of the similarity and difference between ovarian endometriomas and deep infiltrating endometriosis. Reprod Sci. 2018;25(3):329–40.
Sui Cong EM, Charles OA, Omwandho AS-P, Stammler A, Tinneberg H-R, Konrad L. PAI-1 secretion of endometrial and endometriotic cells is Smad2/3- and ERK1/2-dependent and influences cell adhesion. Am J Transl Res. 2016;8(5):2394–402.
Banu SK, Lee J, Speights VO Jr, Starzinski-Powitz A, Arosh JA. Cyclooxygenase-2 regulates survival, migration, and invasion of human endometriotic cells through multiple mechanisms. Endocrinology. 2008;149(3):1180–9.
Acknowledgments
The authors thank Spectrum Health Universal Biorepository and Prisma Healthcare for the sample collection.
Funding
This research was supported by NIH grants RO1 HD042280 and HD083273 to A.T.F.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Ethics Approval
This study has been approved by Michigan State University, Spectrum Health, and Prisma Healthcare Institutional Review Boards.
Consent to Participate (Include Appropriate Statements)
Written informed consent has been obtained from the patients.
Consent for Publication (Include Appropriate Statements)
The authors declared that the work has not been published in whole or in part elsewhere.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 95 kb)
Rights and permissions
About this article
Cite this article
Song, Y., Joshi, N.R., Vegter, E. et al. Establishment of an Immortalized Endometriotic Stromal Cell Line from Human Ovarian Endometrioma. Reprod. Sci. 27, 2082–2091 (2020). https://doi.org/10.1007/s43032-020-00228-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s43032-020-00228-0