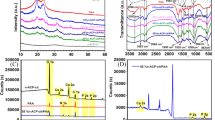
Abstract
Purpose
Because of bone loss, bone complex fractures require medical intervention. Bone grafting may result in a positive recovery from bone loss. As a result, attempts are being made to investigate new bone grafting materials as alternatives, as well as to reproduce bone specificities on a wide scale. Because of its characteristics, Homogeneous Calcium Aluminate Cement (CACH) is a viable candidate for bone substitution. Gold (AuNPs) and silver (AgNPs) nanoparticles can improve CACH efficiency. The biocompatibility of CACH material linked with AuNPs and AgNPs was examined in vitro and in vivo in this study.
Methods
SEM and mitochondrial activity percentage fluctuation were used to analyze materials in vitro for cell adhesion, proliferation, and biocompatibility. Meanwhile, histological examination of samples in vivo searched for bone neoformation.
Results
SEM and mitochondrial activity percentage variation revealed efficient cell adhesion and proliferation when linking biocompatible material. In both samples, histological investigation revealed bone neoformation. CACH linked with AuNPs, on the other hand, produced the most relevant results.
Conclusion
Although both samples showed bone neoformation, CACH combined with AuNPs generated a potentially efficient bone repair material as an enhanced bone substitute.





Similar content being viewed by others
References
Abdala JA, Santos PCO, Vasconcellos LMR, et al. Calcium aluminate cement-based blends for application to fill in bone defects. Res Biomed Eng. 2020;36:429–38. https://doi.org/10.1007/s42600-020-00081-9.
Agarwal R, Garcia AJ. Biomaterial strategies for engineering implants for enhanced osseointegration and bone repair. Adv Drug Deliv Rev. 2015;1(94):53–62. https://doi.org/10.1016/j.addr.2015.03.013.
Bertan A, Cremasco M. Determination of concentration of the Pseudomycelles from fermentation broth via spectroscopy and Neubauer chamber. European Journal of Biological Research. 2020;10:263–70. https://doi.org/10.5281/zenodo.3987017.
Brown AM. A new software for carrying out one-way ANOVA post hoc tests. Computer Methods Progr Biomed. 2005;79(1):89–95. https://doi.org/10.1016/j.cmpb.2005.02.007.
Cohen H, Kugel C, May H, Medlej B, Stein D, Slon V, Hershkovitz I, Brosh T. The impact velocity and bone fracture pattern: forensic perspective. Forensic Sci Int. 2016;266:54–62. https://doi.org/10.1016/j.forsciint.2016.04.035.
DeBaz C, Shamia H, Hahn J, Mithani S, Sadeghi G, Palomo L. Periodontitis impacts quality of life in postmenopausal women. Climact. 2015;18:637–42. https://doi.org/10.3109/13697137.2013.808182.
Drosse I, Volkmer E, Capanna R, Biase P, Mutschler W, Schieker M. Tissue engineering for bone defect healing: an update on a multi-component approach. Inj. 2008;39:S9–20. https://doi.org/10.1016/S0020-1383(08)70011-1.
Goulet JA, Senunas LE, DeSilva GL, Greenfield ML. Autogenous iliac crest bone graft: complications and functional assessment. Clin Orthop Relat Res. 1997;339:76–81. https://doi.org/10.1097/00003086-199706000-00011.
Hatt L, Armiento A, Mys K, et al. Standard in vitro evaluations of engineered bone substitutes are not sufficient to predict in vivo preclinical model outcomes. Acta Biomater. 2023;156:177–89. https://doi.org/10.1016/j.actbio.2022.08.021.
Herdea A, Ionescu A, Dragomirescu M-C, Ulici A. Vitamin D—a risk factor for bone fractures in children: a population-based prospective case–control randomized cross-sectional study. Int J Environ Res Public Health. 2023;20(4):3300. https://doi.org/10.3390/ijerph20043300.
International Organization for Standardization. "ISO 10993–5: 2009-Biological evaluation of medical devices-part 5: tests for in vitro cytotoxicity." (2009): 13.
Komori T. Roles of Runx2 in skeletal development. Adv Exp Med Biol. 2017;962:83–93. https://doi.org/10.1007/978-981-10-3233-2_6.
Li Y, Huang W, Huang S, Du J, Huang C. Screening of anti-cancer agent using zebrafish: comparison with the MTT assay. Biochem Biophys Res Commun. 2012;422:85–90. https://doi.org/10.1016/j.bbrc.2012.04.110.
Li H, Pan S, Xia P, et al. Advances in the application of gold nanoparticles in bone tissue engineering. J Biol Eng. 2020;14:14. https://doi.org/10.1186/s13036-020-00236-3.
Marangoni VS, Paino IM, Zucolotto V. Synthesis and characterization of jacalin-gold nanoparticles conjugates as specific markers for cancer cells. Coll Surf b: Biointerfaces. 2013;112:380–6. https://doi.org/10.1016/j.colsurfb.2013.07.070.
Moussi H, Weiss P, Le Bideau J, Gautier H, Charbonnier B. Injectable macromolecule-based calcium phosphate bone substitutes. Materials Advances. 2022;3(15):6125–41. https://doi.org/10.1039/d2ma00410k.
Oliveira IR, Andrade TL, Jacobovitz M, Pandolfelli VC. Bioactivity of calcium aluminate endodontic cement. J Endod. 2013;39:774–8. https://doi.org/10.1016/j.joen.2013.01.013.
Oliveira IR, Barbosa AM, Grancianinov KJS, Origo FD, Santos KW, Leite PMSCM, Raniero LJ, Pandolfelli VC. Surface properties of calcium aluminate cement blends for bone repair applications. Ceram Int. 2020;46(9):14241–51. https://doi.org/10.1016/j.ceramint.2020.02.033.
Oliveira IR, Barbosa AM, Grancianinov KJS, Origo FD, dos Santos KW, Leite PMSCM, Raniero LJ, Pandolfelli VC. Surface properties of calcium aluminate cement blends for bone repair applications. Ceramics International. 2020;46:14241–51. https://doi.org/10.1016/j.ceramint.2020.02.033.
Parreira RM, Andrade TL, Luz AP, Pandolfelli VC, Oliveira IR. Calcium aluminate cement-based compositions for biomaterial applications. Ceram Int. 2016;42:11732–8. https://doi.org/10.1016/j.ceramint.2016.04.092.
Pompeu LLMF, Santos GL, Pandolfelli VC, Oliveira IR. Calcium aluminates potential for endodontics and orthopedic applications. Cerâmica. 2013;59(350):216–24. https://doi.org/10.1590/S0366-69132013000200004.
Rimondini L, Nicoli-Aldini N, Fini M, Guzzardella G, Tschon M, Giardino R. In vivo experimental study on bone regeneration in critical bone defects using an injectable biodegradable PLA/PGA copolymer. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;99(2):148–54. https://doi.org/10.1016/j.tripleo.2004.05.010.
Sani A, Cao C, Cui D. Toxicity of gold nanoparticles (AuNPs): a review. Biochem Biophys Rep. 2021;26:100991. https://doi.org/10.1016/j.bbrep.2021.100991.
Santos AFL, Castilho ML, Veriato TS, Oliveira IR, Raniero L. Compositions of calcium aluminate cement containing gold and silver nanoparticles for biomaterial applications. Res Biomed Eng. 2020;36:139–46. https://doi.org/10.1007/s42600-020-00045-z.
Schröter L, Kaiser F, Stein S, Gbureck U, Ignatius A. Biological and mechanical performance and degradation characteristics of calcium phosphate cements in large animals and humans. Acta Biomater. 2020;117:1–20. https://doi.org/10.1016/j.actbio.2020.09.031.
Serbetci K, Korkusuz F, Hasirci N. Thermal and mechanical properties of hydroxypaptite impregnated acrylic bone cements. Polym Test. 2004;23(2):145–55. https://doi.org/10.1016/s0142-9418(03)00073-4.
Shi Y, Han X, Pan S, et al. Gold nanomaterials and bone/cartilage tissue engineering: biomedical applications and molecular mechanisms. Front Chem. 2021;9:724188. https://doi.org/10.3389/fchem.2021.724188.
Vasconcellos LMR, Camporês KL, Abdala JMA, Vieira MN, Oliveira IR. Biological and microbiological behavior of calcium aluminate cement-based blend for filling of bone defects. J Mater Sci: Mater Med. 2020;31:10. https://doi.org/10.1007/s10856-019-6348-1.
Wähnert D, Greiner J, Brianza S, Kaltschmidt C, Vordemvenne T, Kaltschmidt B. Strategies to improve bone healing: innovative surgical implants meet nano-/micro-topography of bone scaffolds. Biomedicines. 2021;9(7):746. https://doi.org/10.3390/biomedicines9070746.
Younger EM, Chapman MW. Morbidity at bone graft donor sites. J Orthop Trauma. 1989;3:192–5. https://doi.org/10.1097/00005131-198909000-00002.
Zhang D, Liu D, Zhang J, Fong C, Yang M. Gold nanoparticles stimulate differentiation and mineralization of primary osteoblasts through the ERK/MAPK signaling pathway. Mater Sci Eng C. 2014;42:70–7. https://doi.org/10.1016/j.msec.2014.04.042.
Funding
This study was financed by Fundação de Amparo à Pesquisa do Estado de São Paulo (2017/07519–2), Conselho Nacional de Desenvolvimento Científico e Tecnológico (302944/2018–4 and 302158/2022–7), and Financiadora de Estudos e Projetos (01.13.0275.00 and 01.18.0053.00).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by Paula Fonseca Antunes Vieira, Juliani Caroline Ribeiro de Araújo, Luana Marotta Reis de Vasconcellos, Maiara Lima Castilho and Ivone Regina de Oliveira. The original draft of the manuscript was written by Andrea Fernanda Lopes dos Santos and Leandro Raniero. All authors provided feedback on previous drafts of the manuscript. The final manuscript was read and approved by all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
dos Santos, A.F.L., Vieira, P.F.A., de Araújo, J.C.R. et al. Enhancing biocompatibility and bone neoformation with nanostructured calcium aluminate cement. Res. Biomed. Eng. 39, 389–396 (2023). https://doi.org/10.1007/s42600-023-00278-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42600-023-00278-8