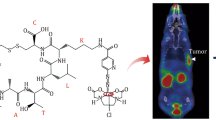
Abstract
The present study explored the 18-kDa translocator protein radioligand 18F-PBR06 as a PET imaging biomarker for diagnosis of inflammation and compared it with 18F-FDG for differentiation of inflammation and lung tumors in animals. 18F-PBR06 was synthesized with an average decay-corrected radiochemical yield of 30–40% (end of synthesis, EOS), and the radiochemical purity was greater than 99%. The inflammation-to-blood ratio of 18F-PBR06 (3.53 ± 0.26) was higher than the tumor-to-blood ratio (1.77 ± 0.35) (P < 0.001). The inflammation-to-muscle ratio of 18F-PBR06 (2.33 ± 0.64) was also higher than the tumor-to-muscle ratio (1.45 ± 0.14) (P = 0.036). Micro-PET/CT images showed high uptake of 18F-FDG in both inflamed muscles and lung tumor tissues. However, 18F-PBR06 uptake in inflamed muscles remained higher than that in the lung tumor tissues, following 90 min of dynamic Micro-PET/CT imaging. Further, macrophages in the inflammatory regions showed a higher fluorescence signal than in lung tumor tissues. Results of the study confirmed that 18F-PBR06 PET/CT imaging allowed for diagnosis of inflammation. Moreover, 18F-PBR06 uptake in the inflammatory regions was significantly higher than in lung tumor tissues, suggesting that 18F-PBR06 PET/CT imaging has potential to differentiate between peripheral lung cancer and inflammation nodules.





Similar content being viewed by others
References
C. Nathan, Points of control in inflammation. Nature 420, 846–852 (2002). https://doi.org/10.1038/nature01320
C. Love, M.B. Tomas, G.G. Tronco et al., FDG PET of infection and inflammation. Radiographics 25, 1357–1368 (2005). https://doi.org/10.1148/rg.255045122
M. Caglar, C. Yener, E. Karabulut, Value of CT, FDG PET-CT and serum tumor markers in staging recurrent colorectal cancer. Int. J. Comput. Assist. Radiol. Surg. 10, 993–1002 (2015). https://doi.org/10.1007/s11548-014-1115-8
N. Seymour, G. Burkill, M. Harries, An analysis of true- and false-positive results of vocal fold uptake in positron emission tomography-computed tomography imaging. J. Laryngol. Otol. 132, 270 (2018). https://doi.org/10.1017/S002221511700247X
I. Vlahos, K. Stefanidis, S. Sheard et al., Lung cancer screening: nodule identification and characterization. Transl. Lung Cancer Res. 7, 288–303 (2018). https://doi.org/10.21037/tlcr.2018.05.02
C. Decotiis, Y. Hu, A.K. Greenberg et al., Inflammatory cytokines and non-small cell lung cancer in a CT-scan screening cohort: background review of the literature. Cancer Biomark. 16, 219–233 (2016). https://doi.org/10.3233/CBM-150559
P. Yang, X.Y. Xu, X.J. Liu et al., The value of delayed (18)F FDG-PET imaging in diagnosis of solitary pulmonary nodules: a preliminary study on 28 patients. Quant. Imaging Med. Surg. 1, 31–34 (2011). https://doi.org/10.3978/j.issn.2223-4292.2011.11.03
L.M. Coussens, Z. Werb, Inflammation and cancer. Nature 420, 860–867 (2014). https://doi.org/10.1038/nature01322
C.J. Van Der Laken, E.H. Elzinga, M.A. Kropholler et al., Noninvasive imaging of macrophages in rheumatoid synovitis using 11C-(R)-PK11195 and positron emission tomography. Arthritis Rheumatol. 58, 3350–3355 (2008). https://doi.org/10.1002/art.23955
Y.Y. Gent, A.E. Voskuyl, R.W. Kloet et al., Macrophage positron emission tomography imaging as a biomarker for preclinical rheumatoid arthritis: findings of a prospective pilot study. Arthritis Rheumatol. 64, 62–66 (2012). https://doi.org/10.1002/art.30655
V. Papadopoulos, M. Baraldi, T.R. Guilarte et al., Translocator protein (18 kDa): new nomenclature for the peripheral-type benzodiazepine receptor based on its structure and molecular function. Trends Pharmacol. Sci. 27, 402–409 (2006). https://doi.org/10.1016/j.tips.2006.06.005
Y. Fujimura, Y. Kimura, F.G. Simeon et al., Biodistribution and radiation dosimetry in humans of a new PET ligand, (18)F-PBR06, to image translocator protein (18 kDa). J. Nucl. Med. 51, 145–149 (2010). https://doi.org/10.2967/jnumed.109.068064
S. Zhang, L. Yang, X. Ling et al., Tumor mitochondria-targeted photodynamic therapy with a translocator protein (TSPO)-specific photosensitizer. Acta Biomater. 28, 160–170 (2015). https://doi.org/10.1016/j.actbio.2015.09.033
M. Imaizumi, E. Briard, S.S. Zoghbi et al., Kinetic evaluation in nonhuman primates of two new pet ligands for peripheral benzodiazepine receptors in brain. Synapse 61, 595–605 (2010). https://doi.org/10.1002/syn.20394
F.M. Lartey, G.O. Ahn, B. Shen et al., PET imaging of stroke-induced neuroinflammation in mice using [18F]PBR06. Mol. Imaging Biol. 16, 109–117 (2014). https://doi.org/10.1007/s11307-013-0664-5
E. Briard, S.S. Zoghbi, F.G. Simeon et al., Single-step high-yield radiosynthesis and evaluation of a sensitive 18F-labeled ligand for imaging brain peripheral benzodiazepine receptors with PET. J. Med. Chem. 52, 688–699 (2009). https://doi.org/10.1021/jm8011855
M. Wang, M. Gao, K.D. Miller et al., Synthesis of [11C]PBR06 and [18F]PBR06 as agents for positron emission tomographic (PET) imaging of the translocator protein (TSPO). Steroids 76, 1331–1340 (2011). https://doi.org/10.1016/j.steroids.2011.06.012
S. Vaidyanathan, C.N. Patel, A.F. Scarsbrook et al., FDG PET/CT in infection and inflammation—current and emerging clinical applications. Clin. Radiol. 70, 787–800 (2015). https://doi.org/10.1016/j.crad.2015.03.010
A.M. Chacko, S. Watanabe, K.J. Herr et al., 18F-FDG as an inflammation biomarker for imaging dengue virus infection and treatment response. JCI Insight (2017). https://doi.org/10.1172/jci.insight.93474
M. Meyer, P. Fernandez, P. Zanotti-Fregonara, 18F-FDG imaging of chronic inflammation associated with promontofixation of the uterus. Clin. Nucl. Med. 42, 42–43 (2017). https://doi.org/10.1097/RLU.0000000000001420
S. Han, S. Woo, H.S. Chong et al., A systematic review of the prognostic value of texture analysis in 18F-FDG PET in lung cancer. Ann. Nucl. Med. (2018). https://doi.org/10.1007/s12149-018-1281-9
J. Toyohara, P.H. Elsinga, K. Ishiwata et al., Evaluation of 4′-[methyl-11C] thiothymidine in a rodent tumor and inflammation model. J. Nucl. Med. 53, 488–494 (2012). https://doi.org/10.2967/jnumed.111.098426
S. Wagner, H.J. Breyholz, C. Holtke et al., A new 18F-labelled derivative of the MMP inhibitor CGS 27023A for PET: radiosynthesis and initial small-animal PET studies. Appl. Radiat. Isot. 67, 606–610 (2009). https://doi.org/10.1016/j.apradiso.2008.12.009
O. Devinsky, A. Vezzani, S. Najjar et al., Glia and epilepsy: excitability and inflammation. Trends Neurosci. 36, 174–184 (2013). https://doi.org/10.1016/j.tins.2012.11.008
H. Boutin, C. Prenant, R. Maroy et al., [18F]DPA-714: direct comparison with [11C]PK11195 in a model of cerebral ischemia in rats. PLoS ONE 8, e56441 (2013). https://doi.org/10.1371/journal.pone.0056441
D. Harhausen, V. Sudmann, U. Khojasteh et al., Specific imaging of inflammation with the 18 kDa translocator protein ligand DPA-714 in animal models of epilepsy and stroke. PLoS ONE 8, e69529 (2013). https://doi.org/10.1371/journal.pone.0069529
O. Gaemperli, J. Shalhoub, D.R. Owen et al., Imaging intraplaque inflammation in carotid atherosclerosis with 11C-PK11195 positron emission tomography/computed tomography. Eur. Heart J. 33, 1902–1910 (2012). https://doi.org/10.1093/eurheartj/ehr367
A. Hatori, J. Yui, T. Yamasaki et al., PET imaging of lung inflammation with [18F]FEDAC, a radioligand for translocator protein (18 kDa). PLoS ONE 7, e45065 (2012). https://doi.org/10.1371/journal.pone.0045065
M.A. Ingersoll, A.M. Platt, S. Potteaux et al., Monocyte trafficking in acute and chronic inflammation. Trends Immunol. 32, 470–477 (2011). https://doi.org/10.1016/j.it.2011.05.001
J. Toyohara, M. Sakata, K. Oda, Longitudinal observation of [11C]4DST uptake in turpentine-induced inflammatory tissue. Nucl. Med. Biol. 40, 240–244 (2013). https://doi.org/10.1016/j.nucmedbio.2012.10.008
C. Wu, X. Yue, L. Lang et al., Longitudinal PET imaging of muscular inflammation using 18F-DPA-714 and 18F-Alfatide II and differentiation with tumors. Theranostics 4, 546–555 (2014). https://doi.org/10.7150/thno.8159
C.H. Xu, J.S. Xue, X.W. Zhang et al., The value of macrophage inhibitory cytokine-1 level in differentiating benign from malignant solitary pulmonary nodules. Clin. Respir. J. 12, 1473–1478 (2018). https://doi.org/10.1111/crj.12693
A.J. Khan, R.P. Akhtar, Z.S. Faruqui, Turpentine oil inhalation leading to lung necrosis and empyema in a toddler. Pediatr. Emerg. Care 22, 355–357 (2006). https://doi.org/10.1097/01.pec.0000215371.48859.fd
Author information
Authors and Affiliations
Corresponding authors
Additional information
This work was funded in part by the National Natural Science Foundation of China (Nos. 11875114, 81471706, and 81871407), Science and Technology Commission of Shanghai Municipality (No. 16410722700), and sponsored by the Shanghai Sailing Program (No. 17YF1417400).
Rights and permissions
About this article
Cite this article
Zhang, H., Tan, H., Mao, WJ. et al. 18F-PBR06 PET/CT imaging of inflammation and differentiation of lung cancer in mice. NUCL SCI TECH 30, 83 (2019). https://doi.org/10.1007/s41365-019-0597-z
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41365-019-0597-z