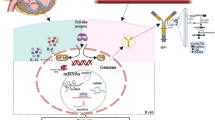
Abstract
Background
There are arguments for individualized treatments and the necessity of non-invasive biomarkers for patients with IgA nephropathy (IgAN) according to gender, but the rationale remains unclear. We aimed to investigate the relationship between urine exosomal circular RNA (circRNA) levels, related genes, clinical features, and renal pathological features in IgA nephropathy patients of different genders.
Methods
Clinicopathological data from patients of different genders from a multicenter cohort were retrospectively analyzed. We used the Oxford classification to examine the severity of pathological damage in these patients. We compared clinical features and renal pathologies between IgAN patients of different genders. Using findings of urine exosomal circRNAs from male IgAN patients, we analyzed the relationship between this factor, the regulated genes located on the sex chromosomes, and renal pathologies.
Results
A total of 502 IgAN patients were included. The proportion of male patients with crescent formation was higher than that of females (p = 0.019). Multivariate logistic regression analysis showed that proteinuria was an independent marker for crescent formation in male and female patients with IgAN, while smoking and higher low-density lipoprotein cholesterol (LDL-C) levels were independent risk factors for crescent formation in males alone. Urine exosomal circRNA chrY:15478147-15481229- located on the Y chromosome in male patients was negatively correlated with the expressions of UTY in specific regions of the Y chromosome.
Conclusion
Compared with female patients, males with IgAN had more severe renal dysfunction and a higher probability of glomerular crescent formation. Urine exosomal circRNA chrY:15478147-15481229- might participate in the pathogenesis of IgAN in male patients by altering UTY expressions.
Graphic abstract



Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this article.
Code availability
Not applicable.
References
Rodrigues JC, Haas M, Reich HN (2017) IgA nephropathy. Clin J Am Soc Nephrol 12(4):677–686
Lv J, Yang Y, Zhang H et al (2013) Prediction of outcomes in crescentic IgA nephropathy in a multicenter cohort study. J Am Soc Nephrol 24(12):2118–2125
Riispere Ž, Laurinavičius A, Kuudeberg A et al (2016) IgA nephropathy clinicopathologic study following the Oxford classification: progression peculiarities and gender-related differences. Medicina (Kaunas) 52(6):340–348
Deng W, Tan X, Zhou Q et al (2018) Gender-related differences in clinicopathological characteristics and renal outcomes of Chinese patients with IgA nephropathy. BMC Nephrol 19(1):31
Peters B, Stegmayr B, Andersson Y et al (2015) Increased risk of renal biopsy complications in patients with IgA-nephritis. Clin Exp Nephrol 19(6):1135–1141
Ma H, Xu Y, Zhang R et al (2019) Differential expression study of circular RNAs in exosomes from serum and urine in patients with idiopathic membranous nephropathy. Arch Med Sci 15(3):738–753
Min QH, Chen XM, Zou YQ et al (2018) Differential expression of urinary exosomal microRNAs in IgA nephropathy. J Clin Lab Anal 32(2):e22226
Solé C, Cortés-Hernández J, Felip ML et al (2015) miR-29c in urinary exosomes as predictor of early renal fibrosis in lupus nephritis. Nephrol Dial Transplant 30(9):1488–1496
Feng Y, Lv LL, Wu WJ et al (2018) Urinary exosomes and exosomal CCL2 mRNA as biomarkers of active histologic injury in IgA nephropathy. Am J Pathol 188(11):2542–2552
Wilusz JE, Sharp PA (2013) Molecular biology. A circuitous route to noncoding RNA. Science 340(6131):440–441
Li Z, Huang C, Bao C et al (2017) Corrigendum: exon-intron circular RNAs regulate transcription in the nucleus. Nat Struct Mol Biol 24(2):194
Jeck WR, Sharpless NE (2014) Detecting and characterizing circular RNAs. Nat Biotechnol 32(5):453–461
Luan R, Tian G, Ci X et al (2021) Differential expression analysis of urinary exosomal circular RNAs in patients with IgA nephropathy. Nephrology (Carlton) 26(5):432–441
Cattran DC, Coppo R, Cook HT et al (2009) The Oxford classification of IgA nephropathy: rationale, clinicopathological correlations, and classification. Kidney Int 76(5):534–545
Trimarchi H, Barratt J, Cattran DC et al (2017) Oxford Classification of IgA nephropathy 2016: an update from the IgA Nephropathy Classification Working Group. Kidney Int 91(5):1014–1021
Wang LP, Peng XY, Lv XQ et al (2019) High throughput circRNAs sequencing profile of follicle fluid exosomes of polycystic ovary syndrome patients. J Cell Physiol 234:15537–15547
Yang C, Wei Y, Yu L et al (2019) Identification of altered circular RNA expression in serum exosomes from patients with papillary thyroid carcinoma by high-throughput sequencing. Med Sci Monit 25:2785–2791
Kaneko Y, Yoshita K, Kono E et al (2016) Extracapillary proliferation and arteriolar hyalinosis are associated with long-term kidney survival in IgA nephropathy. Clin Exp Nephrol 20(4):569–577
Radhakrishnan J, Cattran DC (2012) The KDIGO practice guideline on glomerulonephritis: reading between the (guide)lines–application to the individual patient. Kidney Int 82(8):840–856
Arany I, Taylor M, Fülöp T et al (2018) Adverse effects of chronic nicotine exposure on the kidney: potential human health implications of experimental findings. Int J Clin Pharmacol Ther 56(11):501–506
Su H, Chen S, He FF et al (2015) New insights into glomerular parietal epithelial cell activation and its signaling pathways in glomerular diseases. Biomed Res Int 2015:318935
Jain G, Jaimes EA (2013) Nicotine signaling and progression of chronic kidney disease in smokers. Biochem Pharmacol 86(8):1215–1223
Zheng CM, Lee YH, Chiu IJ et al (2020) Nicotine causes nephrotoxicity through the induction of NLRP6 inflammasome and Alpha7 nicotinic acetylcholine receptor. Toxics 8(4):92
Xu D, You X, Wang Z et al (2015) Chinese systemic lupus erythematosus treatment and research group registry VI: effect of cigarette smoking on the clinical phenotype of Chinese patients with systemic lupus erythematosus. PLoS ONE 10(8):e0134451
Jaimes EA, Zhou MS, Siddiqui M et al (2021) Nicotine, smoking, podocytes, and diabetic nephropathy. Am J Physiol Renal Physiol 320(3):F442-f453
Liao D, Ma L, Liu J et al (2019) Cigarette smoking as a risk factor for diabetic nephropathy: a systematic review and meta-analysis of prospective cohort studies. PLoS ONE 14(2):e0210213
Li Z, Yao Y, Han W et al (2015) Smoking prevalence and associated factors as well as attitudes and perceptions towards tobacco control in Northeast China. Int J Environ Res Public Health 12(7):8606–8618
Arany I, Hall S, Reed DK et al (2016) Nicotine enhances high-fat diet-induced oxidative stress in the kidney. Nicotine Tob Res 18(7):1628–1634
Busuioc RM, Covic A, Kanbay M et al (2020) Protein convertase subtilisin/kexin type 9 biology in nephrotic syndrome: implications for use as therapy. Nephrol Dial Transplant 35(10):1663–1674
Ashwal-Fluss R, Meyer M, Pamudurti NR et al (2014) circRNA biogenesis competes with pre-mRNA splicing. Mol Cell 56(1):55–66
Salzman J (2016) Circular RNA expression: its potential regulation and function. Trends Genet 32(5):309–316
Sallustio F, Serino G, Cox SN et al (2016) Aberrantly methylated DNA regions lead to low activation of CD4+ T-cells in IgA nephropathy. Clin Sci (Lond) 130(9):733–746
Walport LJ, Hopkinson RJ, Vollmar M et al (2014) Human UTY(KDM6C) is a male-specific Nϵ-methyl lysyl demethylase. J Biol Chem 289(26):18302–18313
Vendomèle J, Dehmani S, Khebizi Q et al (2018) Subretinal injection of HY peptides induces systemic antigen-specific inhibition of effector CD4(+) and CD8(+) T-cell responses. Front Immunol 9:504
Bai L, Li H, Li J et al (2019) Immunosuppressive effect of artemisinin and hydroxychloroquine combination therapy on IgA nephropathy via regulating the differentiation of CD4+ T cell subsets in rats. Int Immunopharmacol 70:313–323
Wang L, Shilatifard A (2019) UTX mutations in human cancer. Cancer Cell 35(2):168–176
Gažová I, Lengeling A, Summers KM (2019) Lysine demethylases KDM6A and UTY: The X and Y of histone demethylation. Mol Genet Metab 127(1):31–44
Kobatake K, Ikeda KI, Nakata Y et al (2020) Kdm6a deficiency activates inflammatory pathways, promotes M2 macrophage polarization, and causes bladder cancer in cooperation with p53 dysfunction. Clin Cancer Res 26(8):2065–2079
Makita Y, Suzuki H, Kano T et al (2020) TLR9 activation induces aberrant IgA glycosylation via APRIL- and IL-6-mediated pathways in IgA nephropathy. Kidney Int 97(2):340–349
Andricovich J, Perkail S, Kai Y et al (2018) Loss of KDM6A activates super-enhancers to induce gender-specific squamous-like pancreatic cancer and confers sensitivity to BET inhibitors. Cancer Cell 33(3):512-526.e8
Tricarico R, Nicolas E, Hall MJ et al (2020) X- and Y-linked chromatin-modifying genes as regulators of sex-specific cancer incidence and prognosis. Clin Cancer Res 26(21):5567–5578
Molina-Serrano D, Kyriakou D, Kirmizis A (2019) Histone modifications as an intersection between diet and longevity. Front Genet 10:192
Funding
This study was supported by the Hygiene and Health Technology Innovation Project of Health Commission of Jilin Province (No. 2020J037).
Author information
Authors and Affiliations
Contributions
LRM and TG carried out the experiment and wrote the paper. ZH analyzed the clinical data. SXL and ZR collected the clinical data. LJC collected urine samples. LXH conceived the research idea and analyzed the experimental data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Ethical approval
The current study was approved by the Ethics Committee of the Second Hospital of Jilin University (No.2020001) and adhered to the Helsinki Declaration guidelines.
Consent to participate
Informed consent was obtained from participants.
Consent for publication
Consent was obtained from participants regarding the publishing of their data.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Luan, R., Tian, G., Zhang, H. et al. Urinary exosomal circular RNAs of sex chromosome origin are associated with gender-related risk differences of clinicopathological features in patients with IgA nephropathy. J Nephrol 35, 1069–1078 (2022). https://doi.org/10.1007/s40620-021-01118-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-021-01118-7