Abstract
Purpose
Biomarkers of clinical and therapeutic outcome in acromegaly are needed. Polymorphisms or epigenetic changes of detoxification genes, such as those coding for the aryl hydrocarbon receptor (AHR) and the glutathione-S-transferase-P1 (GSTP1), could have a role in GH secreting pituitary tumors’ pathophysiology and clinical expression. In this study, we assessed the contribution of GSTP1 gene promoter methylation status, per se or in combination with the occurrence of the AHR gene rs2066853 variant, on clinical features and response to somatostatin analogs (SSA) treatment in acromegaly patients.
Methods
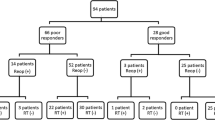
This is an observational, retrospective study, carried out in the Endocrine Unit of an Italian University Hospital. We enrolled 77 wild-type AIP gene acromegaly patients, who have been screened for germline AHR rs2066853 variant and GSTP1 gene promoter methylation. Clinical and biochemical parameters were compared after patients’ stratification according to GSTP1 methylation status and the presence of AHR rs2066853. We also evaluated the response to SSA treatment in 71 cases.
Results
17 patients carried the AHR rs2066853 variant and 26 had methylated GSTP1 (GSTP1-methyl) gene promoter. GSTP1-methyl patients showed a higher prevalence of diabetes mellitus (p = 0.01), colonic polyps (p = 0.05), and were more resistant to SSA (p = 0.02) as compared to GSTP1 unmethylated patients (GSTP1-unmethyl). Patients GSTP1-unmethyl and AHR wild-type were the most sensitive to SSA treatment, while those with both GSTP1-methyl and AHR rs2066853 variant were all resistant to SSA (p = 0.01).
Conclusions
In acromegaly, GSTP1 gene methylation associates with resistance to SSA treatment, especially in patients carrying also the AHR rs2066853 variant, and with increased prevalence of colonic polyps and diabetes mellitus.

Similar content being viewed by others
References
Capatina C, Wass JA (2015) 60 years of neuroendocrinology: acromegaly. J Endocrinol 226(2):T141–T160. https://doi.org/10.1530/JOE-15-0109
Pivonello R, Auriemma RS, Grasso LF, Pivonello C, Simeoli C, Patalano R, Galdiero M, Colao A (2017) Complications of acromegaly: cardiovascular, respiratory and metabolic comorbidities. Pituitary 20(1):46–62. https://doi.org/10.1007/s11102-017-0797-7
Terzolo M, Reimondo G, Berchialla P, Ferrante E, Malchiodi E, De Marinis L, Pivonello R, Grottoli S, Losa M, Cannavo S, Ferone D, Montini M, Bondanelli M, De Menis E, Martini C, Puxeddu E, Velardo A, Peri A, Faustini-Fustini M, Tita P, Pigliaru F, Peraga G, Borretta G, Scaroni C, Bazzoni N, Bianchi A, Berton A, Serban AL, Baldelli R, Fatti LM, Colao A, Arosio M, Italian Study Group of Acromegaly (2017) Acromegaly is associated with increased cancer risk: a survey in Italy. Endocr Relat Cancer 24(9):495–504. https://doi.org/10.1530/erc-16-0553
Giustina A, Chanson P, Kleinberg D, Bronstein MD, Clemmons DR, Klibanski A, van der Lely AJ, Strasburger CJ, Lamberts SW, Ho KK, Casanueva FF, Melmed S, Acromegaly Consensus Group (2014) Expert consensus document: a consensus on the medical treatment of acromegaly. Nat Rev Endocrinol 10(4):243–248. https://doi.org/10.1038/nrendo.2014.21
Giustina A, Arnaldi G, Bogazzi F, Cannavo S, Colao A, De Marinis L, De Menis E, Degli Uberti E, Giorgino F, Grottoli S, Lania AG, Maffei P, Pivonello R, Ghigo E (2017) Pegvisomant in acromegaly: an update. J Endocrinol Invest 40(6):577–589. https://doi.org/10.1007/s40618-017-0614-1
Maffezzoni F, Formenti AM, Mazziotti G, Frara S, Giustina A (2016) Current and future medical treatments for patients with acromegaly. Expert Opin Pharmacother 17(12):1631–1642. https://doi.org/10.1080/14656566.2016.1199687
Paragliola RM, Corsello SM, Salvatori R (2017) Somatostatin receptor ligands in acromegaly: clinical response and factors predicting resistance. Pituitary 20(1):109–115. https://doi.org/10.1007/s11102-016-0768-4
Kasuki L, Wildemberg LE, Gadelha MR (2018) Management of endocrine disease: personalized medicine in the treatment of acromegaly. Eur J Endocrinol 178(3):R89–R100. https://doi.org/10.1530/EJE-17-1006
Cannavo S, Ragonese M, Puglisi S, Romeo PD, Torre ML, Alibrandi A, Scaroni C, Occhi G, Ceccato F, Regazzo D, De Menis E, Sartorato P, Arnaldi G, Trementino L, Trimarchi F, Ferrau F (2016) Acromegaly is more severe in patients with AHR or AIP gene variants living in highly polluted areas. J Clin Endocrinol Metab 101(4):1872–1879. https://doi.org/10.1210/jc.2015-4191
Ibanez-Costa A, Korbonits M (2017) AIP and the somatostatin system in pituitary tumours. J Endocrinol 235(3):R101–R116. https://doi.org/10.1530/JOE-17-0254
Cannavo S, Ferrau F, Ragonese M, Romeo PD, Torre ML, Puglisi S, De Menis E, Arnaldi G, Salpietro C, Cotta OR, Albani A, Ruggeri RM, Trimarchi F (2014) Increased frequency of the rs2066853 variant of aryl hydrocarbon receptor gene in patients with acromegaly. Clin Endocrinol (Oxf) 81(2):249–253. https://doi.org/10.1111/cen.12424
Wolinski K, Stangierski A, Dyrda K, Nowicka K, Pelka M, Iqbal A, Car A, Lazizi M, Bednarek N, Czarnywojtek A, Gurgul E, Ruchala M (2017) Risk of malignant neoplasms in acromegaly: a case-control study. J Endocrinol Invest 40(3):319–322. https://doi.org/10.1007/s40618-016-0565-y
Schnekenburger M, Karius T, Diederich M (2014) Regulation of epigenetic traits of the glutathione S-transferase P1 gene: from detoxification toward cancer prevention and diagnosis. Front Pharmacol 5:170. https://doi.org/10.3389/fphar.2014.00170
Yuan Y, Qian ZR, Sano T, Asa SL, Yamada S, Kagawa N, Kudo E (2008) Reduction of GSTP1 expression by DNA methylation correlates with clinicopathological features in pituitary adenomas. Mod Pathol 21(7):856–865. https://doi.org/10.1038/modpathol.2008.60
Katznelson L, Laws ER Jr, Melmed S, Molitch ME, Murad MH, Utz A, Wass JA, Endocrine S (2014) Acromegaly: an endocrine society clinical practice guideline. J Clin Endocrinol Metab 99(11):3933–3951. https://doi.org/10.1210/jc.2014-2700
Ferrau F, Romeo PD, Puglisi S, Ragonese M, Torre ML, Scaroni C, Occhi G, De Menis E, Arnaldi G, Trimarchi F, Cannavo S (2016) Analysis of GPR101 and AIP genes mutations in acromegaly: a multicentric study. Endocrine 54(3):762–767. https://doi.org/10.1007/s12020-016-0862-4
Boguszewski CL, Ayuk J (2016) Management of endocrine disease: acromegaly and cancer: an old debate revisited. Eur J Endocrinol 175(4):R147–R156. https://doi.org/10.1530/EJE-16-0178
Cannavo S, Condurso R, Ragonese M, Ferrau F, Alibrandi A, Arico I, Romanello G, Squadrito S, Trimarchi F, Silvestri R (2011) Increased prevalence of restless legs syndrome in patients with acromegaly and effects on quality of life assessed by Acro-QoL. Pituitary 14(4):328–334. https://doi.org/10.1007/s11102-011-0298-z
Ragonese M, Alibrandi A, Di Bella G, Salamone I, Puglisi S, Cotta OR, Torre ML, Ferrau F, Ruggeri RM, Trimarchi F, Cannavo S (2014) Cardiovascular events in acromegaly: distinct role of Agatston and Framingham score in the 5-year prediction. Endocrine 47(1):206–212. https://doi.org/10.1007/s12020-013-0115-8
Cannavo S, Trimarchi F, Ferrau F (2017) Acromegaly, genetic variants of the aryl hydrocarbon receptor pathway and environmental burden. Mol Cell Endocrinol 457:81–88. https://doi.org/10.1016/j.mce.2016.12.019
Formosa R, Borg J, Vassallo J (2017) Aryl hydrocarbon receptor (AHR) is a potential tumour suppressor in pituitary adenomas. Endocr Relat Cancer 24(8):445–457. https://doi.org/10.1530/ERC-17-0112
Formosa R, Vassallo J (2017) The complex biology of the aryl hydrocarbon receptor and its role in the pituitary gland. Horm Cancer 8(4):197–210. https://doi.org/10.1007/s12672-017-0300-y
Dietrich C, Kaina B (2010) The aryl hydrocarbon receptor (AhR) in the regulation of cell-cell contact and tumor growth. Carcinogenesis 31(8):1319–1328. https://doi.org/10.1093/carcin/bgq028
Feng S, Cao Z, Wang X (1836) Role of aryl hydrocarbon receptor in cancer. Biochim Biophys Acta 2:197–210. https://doi.org/10.1016/j.bbcan.2013.05.001
Daly AF, Beckers A (2017) The role of AIP mutations in pituitary adenomas: 10 years on. Endocrine 55(2):333–335. https://doi.org/10.1007/s12020-016-1194-0
Hernandez-Ramirez LC, Gabrovska P, Denes J, Stals K, Trivellin G, Tilley D, Ferrau F, Evanson J, Ellard S, Grossman AB, Roncaroli F, Gadelha MR, Korbonits M, International FIPA Consortium (2015) Landscape of familial isolated and young-onset pituitary adenomas: prospective diagnosis in AIP mutation carriers. J Clin Endocrinol Metab 100(9):E1242–E1254. https://doi.org/10.1210/jc.2015-1869
Tang ST, Wang CJ, Tang HQ, Zhang Q, Wang Y (2013) Evaluation of glutathione S-transferase genetic variants affecting type 2 diabetes susceptibility: a meta-analysis. Gene 530(2):301–308. https://doi.org/10.1016/j.gene.2013.08.043
Shen X, Wang J, Yan X, Ren X, Wang F, Chen X, Xu Y (2016) Predictive value of GSTP1 Ile105Val polymorphism in clinical outcomes of chemotherapy in gastric and colorectal cancers: a systematic review and meta-analysis. Cancer Chemother Pharmacol 77(6):1285–1302. https://doi.org/10.1007/s00280-016-3047-1
Funding
This study has been supported by a grant of the Ministry of Education, University and Research (PRIN 2015, Prot. 2015ZHKFTA) and of the Ministry of Health (Progetto Ordinario Ricerca Finalizzata RF-2013-02356201) of the Italian Government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
On behalf of all authors, the corresponding author states that there is no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Ferraù, F., Romeo, P.D., Puglisi, S. et al. GSTP1 gene methylation and AHR rs2066853 variant predict resistance to first generation somatostatin analogs in patients with acromegaly. J Endocrinol Invest 42, 825–831 (2019). https://doi.org/10.1007/s40618-018-0988-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40618-018-0988-8