Abstract
Purpose of review
To describe the host-pathogen factors that impact the management of cryptococcal meningitis (CM) in immunosuppressed patients that lead to the development of cryptococcal immune reconstitution inflammatory syndrome (C-IRIS).
Recent findings
The pre-screening of HIV-infected patients in resource-poor countries with the lateral flow cryptococcal antigen assay could prevent C-IRIS. Delaying ART by 4 weeks is associated with improved survival and recommended by guidelines; this approach remains controversial in wealthier areas as there is limited data. A 5-flucytosine-based combination regimen and avoidance of corticosteroids on initial treatment improve cryptococcus clearance from cerebrospinal fluid. New monoclonal antibodies and kinase inhibitors that alter the immune system, such as ibrutinib and ruxolitinib, have been associated with cryptococcosis.
Summary
It is important to recognize that restoration of the immune system, regardless of the host, can lead to C-IRIS. Prevention of C-IRIS by pre-screening patients, delaying antiretroviral therapy, and using a 5-flucytosine backbone regimen are important in the management of meningitis. C-IRIS management requires the exclusion of therapeutic failure or antifungal resistance. Further research is needed on whether delaying antiretroviral therapy by 4 weeks is necessary in wealthier countries to improve C-IRIS outcomes, and whether C-IRIS can be seen with new immunologic agents.

Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
•Perfect JR, Bicanic T. Cryptococcosis diagnosis and treatment: What do we know now. Fungal Genet Biol. 2015;78:49–54. https://doi.org/10.1016/j.fgb.2014.10.003Excellent paper on diagnosis and management of cryptococcal infections.
•Kwon-Chung KJ, Bennett JE, Wickes BL, Meyer W, Cuomo CA, Wollenburg KR, et al. The Case for Adopting the "Species Complex" Nomenclature for the Etiologic Agents of Cryptococcosis. mSphere. 2017;2:1. https://doi.org/10.1128/mSphere.00357-16This paper describes the reasons for renaming all the cryptococcal species that infect humans.
Day JN, Qihui S, Thanh LT, Trieu PH, Van AD, Thu NH, et al. Comparative genomics of Cryptococcus neoformans var. grubii associated with meningitis in HIV infected and uninfected patients in Vietnam. PLoS Negl Trop Dis. 2017:e0005628. https://doi.org/10.1371/journal.pntd.0005628.
Desjardins CA, Giamberardino C, Sykes SM, Yu CH, Tenor JL, Chen Y, et al. Population genomics and the evolution of virulence in the fungal pathogen Cryptococcus neoformans. Genome Res. 2017;27:1207–19. https://doi.org/10.1101/gr.218727.116.
Knoke M, Schwesinger G. One hundred years ago: the history of cryptococcosis in Greifswald. Medical mycology in the nineteenth century. Mycoses. 1994;37:229–33. https://doi.org/10.1111/j.1439-0507.1994.tb00418x.
Emmons CW. Saprophytic sources of Cryptococcus neoformans associated with the pigeon (Columba livia). Am J Hyg. 1955;62:227–32. https://doi.org/10.1093/oxfordjournals.aje.a119775.
Ruiz A, Neilson JB, Bulmer GS. Control of Cryptococcus neoformans in nature by biotic factors. Sabouraudia. 1982;20:21–9.
Steenbergen JN, Shuman HA, Casadevall A. Cryptococcus neoformans interactions with amoebae suggest an explanation for its virulence and intracellular pathogenic strategy in macrophages. Proc Natl Acad Sci U S A. 2001;26:15245–50. https://doi.org/10.1073/pnas.261418798.
Chen SC, Meyer W, Sorrell TC. Cryptococcus gattii infections. Clin Microbiol Rev. 2014;27:980–1024. https://doi.org/10.1128/CMR.00126-13.
Datta K, Bartlett KH, Baer R, Byrnes E, Galanis E, Heitman J, et al. Spread of Cryptococcus gattii into Pacific Northwest region of the United States. Emerg Infect Dis. 2009;15:1185–91. https://doi.org/10.3201/eid1508.081384.
•Harris JR, Lockhart SR, Debess E, Marsden-Haug N, Goldoft M, Wohrle R, et al. Cryptococcus gattii in the United States: clinical aspects of infection with an emerging pathogen. Clin Infect Dis. 2011;53:1188–95. https://doi.org/10.1093/cid/cir723Important description of Cryptococcal gattii infections in the Northwestern US states.
Phillips P, Galanis E, MacDougall L, Chong MY, Balshaw R, Cook VJ, et al. Longitudinal clinical findings and outcome among patients with Cryptococcus gattii infection in British Columbia. Clin Infect Dis. 2015;60:1368–76. https://doi.org/10.1093/cid/civ041.
Forrest GN, Bhalla P, DeBess EE, Winthrop KL, Lockhart SR, Mohammadi J, et al. Cryptococcus gattii infection in solid organ transplant recipients: description of Oregon outbreak cases. Transpl Infect Dis. 2015;17:467–76. https://doi.org/10.1111/tid.12370.
Perfect JR, Casadevall A. Cryptococcosis. Infect Dis Clin N Am. 2003;16:837–74. https://doi.org/10.1016/s0891-5520(02)00036-3.
•Rajasingham R, Smith RM, Park BJ, Jarvis JN, Govender NP, Chiller TM, et al. Global burden of disease of HIV-associated cryptococcal meningitis: an updated analysis. Lancet Infect Dis. 2017;17:873–81. https://doi.org/10.1016/S1473-3099(17)30243-8This article provides excellent data on the current burden of cryptococcocal meningits and information on patient populations most at risk.
Williamson PR. The relentless march of cryptococcal meningitis. Lancet Infect Dis. 2017;13:790–1. https://doi.org/10.1038/nrneurol.2016.167.
Haugen RK, Baker RD. The pulmonary lesions in cryptococcosis with special reference to subpleural nodules. Am J Clin Pathol. 1954;24:1381–90. https://doi.org/10.1093/ajcp/24.12.1381.
Garcia-Hermoso D, Janbon G, Dromer F. Epidemiological evidence for dormant Cryptococcus neoformans infection. J Clin Microbiol. 1999;37:3204–9.
Kontoyiannis DP, Marr KA, Park BJ, Alexander BD, Anaissie EJ, Walsh TJ, et al. Prospective surveillance for invasive fungal infections in hematopoietic stem cell transplant recipients, 2001-2006: overview of the Transplant-Associated Infection Surveillance Network (TRANSNET) Database. Clin Infect Dis. 2010;50:1091–100. https://doi.org/10.1086/651263.
Sun HY, Wagener MM, Singh N. Cryptococcosis in solid-organ, hematopoietic stem cell, and tissue transplant recipients: evidence-based evolving trends. Clin Infect Dis. 2009;48:1566–76. https://doi.org/10.1086/598936.
Husain S, Wagener MM, Singh N. Cryptococcus neoformans infection in organ transplant recipients: variables influencing clinical characteristics and outcome. Emerg Infect Dis. 2001;7:375–81. https://doi.org/10.3201/eid0703.010302.
Singh N, Alexander BD, Lortholary O, Dromer F, Gupta KL, John GT, et al. Cryptococcus neoformans in organ transplant recipients: impact of calcineurin-inhibitor agents on mortality. J Infect Dis. 2007;195:756–64. https://doi.org/10.1086/511438.
Singh N, Husain S, De Vera M, Gayowski T, Cacciarelli TV. Cryptococcus neoformans Infection in Patients With Cirrhosis, Including Liver Transplant Candidates. Medicine (Baltimore). 2004;83:188–92. https://doi.org/10.1097/01.md.0000126760.45299.69.
Singh N, Huprikar S, Burdette SD, Morris MI, Blair JE, Wheat LJ. Donor-Derived Fungal Infections in Organ Transplant Recipients: Guidelines of the American Society of Transplantation, Infectious Diseases Community of Practice(dagger). Am J Transplant. 2012;12:2414–28. https://doi.org/10.1111/j.1600-6143.2012.04100.x.
Lionakis MS, Netea MG, Holland SM. Mendelian genetics of human susceptibility to fungal infection. Cold Spring Harb Perspect Med. 2014;4:6. https://doi.org/10.1101/cshperspect.a019638.
••Gundacker NDB, JW. Fungal Infections in the Era of Biologic Therapies. Curr Clin Micro Rpt. 2015;2:76–83. https://doi.org/10.1007/s40588-015-0018-yThis article provides an important review of the role of new TNF-alpha inhibitors in the role of invasive fungal infections.
Nielsen K, Cox GM, Litvintseva AP, Mylonakis E, Malliaris SD, Benjamin DK Jr, et al. Cryptococcus neoformans {alpha} strains preferentially disseminate to the central nervous system during coinfection. Infect Immun. 2005;73:4922–33. https://doi.org/10.1128/IAI.73.8.4922-4933.2005.
Perfect JR. Cryptococcus neoformans: a sugar-coated killer with designer genes. FEMS Immunol Med Microbiol. 2005;45:395–404. https://doi.org/10.1016/j.femsim.2005.06.005.
Bratton EW, El Husseini N, Chastain CA, Lee MS, Poole C, Sturmer T, et al. Comparison and temporal trends of three groups with cryptococcosis: HIV-infected, solid organ transplant, and HIV-negative/non-transplant. PLoS One. 2012;7:e43582. https://doi.org/10.1371/journal.pone.0043582.
Santangelo R, Zoellner H, Sorrell T, Wilson C, Donald C, Djordjevic J, et al. Role of extracellular phospholipases and mononuclear phagocytes in dissemination of cryptococcosis in a murine model. Infect Immun. 2004;72:2229–39. https://doi.org/10.1128/iai.72.4.2229-2239.2004.
Casadevall A. Cryptococci at the brain gate: break and enter or use a Trojan horse? J Clin Invest. 2010;120:1389–92. https://doi.org/10.1172/JCI42949.
Jarvis JN, Meintjes G, Bicanic T, Buffa V, Hogan L, Mo S, et al. Cerebrospinal fluid cytokine profiles predict risk of early mortality and immune reconstitution inflammatory syndrome in HIV-associated cryptococcal meningitis. PLoS Pathog. 2015;11:e1004754. https://doi.org/10.1371/journal.ppat.1004754.
Haidar G, Singh N. Cryptococcus: Shedding New Light on an Inveterate Yeast. J Fungi (Basel). 2015;1:115–29. https://doi.org/10.3390/jof1020115.
Boulware DR, Meya DB, Bergemann TL, Wiesner DL, Rhein J, Musubire A, et al. Clinical features and serum biomarkers in HIV immune reconstitution inflammatory syndrome after cryptococcal meningitis: a prospective cohort study. PLoS Med. 2010;21:e1000384. https://doi.org/10.1371/journal.pmed.1000384.
•Oshima K, Takazono T, Saijo T, Tashiro M, Kurihara S, Yamamoto K, et al. Examination of cryptococcal glucuronoxylomannan antigen in bronchoalveolar lavage fluid for diagnosing pulmonary cryptococcosis in HIV-negative patients. Med Mycol. 2018;56:88–94. https://doi.org/10.1093/mmy/myx010This article examines the performance of the cryptococcal glucuronoxylomannan (GXM)antigen test from bronchoalveolar lavage fluid (BALF) samples, in an HIV-negative patients. The article suggests the BALF GXM may have a role in the early diagnosis of pulmonary cryptococcosis.
Buchanan KL, Murphy JW. What makes Cryptococcus neoformans a pathogen? Emerg Infect Dis. 1998;4:71–83. https://doi.org/10.3201/eid0401.980109.
Singh N, Perfect JR. Immune reconstitution syndrome associated with opportunistic mycoses. Lancet Infect Dis. 2007;7:395–401. https://doi.org/10.1016/s1473-3099(07)70085-3.
Einsiedel L, Gordon DL, Dyer JR. Paradoxical inflammatory reaction during treatment of Cryptococcus neoformans var. gattii meningitis in an HIV-seronegative woman. Clin Infect Dis. 2004;39:e78–82. https://doi.org/10.1086/424746.
Perfect JR, Dismukes WE, Dromer F, Goldman DL, Graybill JR, Hamill RJ, et al. Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the infectious diseases society of america. Clin Infect Dis. 2010;50:291–322. https://doi.org/10.1086/649858.
Vidal JE, Boulware DR. Lateral Flow Assay for Cryptococcal Antigen: An Important Advance to Improve the Continuum of Hiv Care and Reduce Cryptococcal Meningitis-Related Mortality. Rev Inst Med Trop Sao Paulo. 2015;15(Suppl19):38–45. https://doi.org/10.1590/S0036-46652015000700008.
Vijayan T, Chiller T, Klausner JD. Sensitivity and specificity of a new cryptococcal antigen lateral flow assay in serum and cerebrospinal fluid. MLO Med Lab Obs. 2013;45:16 18, 20.
Kabanda T, Siedner MJ, Klausner JD, Muzoora C, Boulware DR. Point-of-care diagnosis and prognostication of cryptococcal meningitis with the cryptococcal antigen lateral flow assay on cerebrospinal fluid. Clin Infect Dis. 2014;58:113–6. https://doi.org/10.1093/cid/cit641.
O'Connor L, Livermore J, Sharp AD, Goodwin J, Gregson L, Howard SJ, et al. Pharmacodynamics of liposomal amphotericin B and flucytosine for cryptococcal meningoencephalitis: safe and effective regimens for immunocompromised patients. J Infect Dis. 2013;208:351–61. https://doi.org/10.1093/infdis/jit164.
•Day JN, Chau TT, Wolbers M, Mai PP, Dung NT, Mai NH, et al. Combination antifungal therapy for cryptococcal meningitis. N Engl J Med. 2013;368:1291–302. https://doi.org/10.1056/NEJMoa1110404Important randomized study that showed 5 flucytosine combination therapy reduced mortality and imprved cerebrospinal fluid clearance. Also, fluconazole monotherapy as initial therapy resulted in delayed clearance and increased mortality.
Lawrence DS, Youssouf N, Molloy SF, Alanio A, Alufandika M, Boulware DR, et al. AMBIsome Therapy Induction OptimisatioN (AMBITION): High Dose AmBisome for Cryptococcal Meningitis Induction Therapy in sub-Saharan Africa: Study Protocol for a Phase 3 Randomised Controlled Non-Inferiority Trial. Trials. 2018;19:649. https://doi.org/10.1186/s13063-018-3026-4.
••Jarvis JN, Leeme TB, Molefi M, Chofle AA, Bidwell G, Tsholo K, et al. Short-course High-dose Liposomal Amphotericin B for Human Immunodeficiency Virus-associated Cryptococcal Meningitis: A Phase 2 Randomized Controlled Trial. Clin Infect Dis. 2019;68:393–401. https://doi.org/10.1093/cid/ciy515Early treatment paper with weekly amphotericin B, but importantly daily 5 flucytosine showing early clearance and similar outcomes. Outcomes still better than non 5 flucytosine therapies.
Longley N, Harrison TS, Jarvis JN. Cryptococcal immune reconstitution inflammatory syndrome. Curr Opin Infect Dis. 2013;26:26–34. https://doi.org/10.1097/QCO.0b013e32835c21d1.
Rolfes MA, Hullsiek KH, Rhein J, Nabeta HW, Taseera K, Schutz C, et al. The effect of therapeutic lumbar punctures on acute mortality from cryptococcal meningitis. Clin Infect Dis. 2014;59:1607–14. https://doi.org/10.1093/cid/ciu596.
••Beardsley J, Wolbers M, Kibengo FM, Ggayi AB, Kamali A, Cuc NT, et al. Adjunctive Dexamethasone in HIV-Associated Cryptococcal Meningitis. N Engl J Med. 2016;374:542–54. https://doi.org/10.1056/NEJMoa1509024Critically important paper showing that early dexamethasone therapy resulted in dleayed cryptococcal clearance and increased mortality.
••Beardsley J, Hoang NLT, Kibengo FM, Tung NLN, Binh TQ, Hung LQ, et al. Do Intracerebral Cytokine Responses Explain the Harmful Effects of Dexamethasone in Human Immunodeficiency Virus-associated Cryptococcal Meningitis? Clin Infect Dis. 2019;68:1494–501. https://doi.org/10.1093/cid/ciy725Another paper showing how dexamethasone delays clearance of cryptococcus through Interleukin-10.
Shelburne SA 3rd, Darcourt J, White AC Jr, Greenberg SB, Hamill RJ, Atmar RL, et al. The role of immune reconstitution inflammatory syndrome in AIDS-related Cryptococcus neoformans disease in the era of highly active antiretroviral therapy. Clin Infect Dis. 2005, 40:1049–52. https://doi.org/10.1086/428618.
Lortholary O, Fontanet A, Memain N, Martin A, Sitbon K, Dromer F, et al. Incidence and risk factors of immune reconstitution inflammatory syndrome complicating HIV-associated cryptococcosis in France. AIDS. 2005;19:1043–9. https://doi.org/10.1097/01.aids.000174450.70874.30.
Shelburne SA, Visnegarwala F, Darcourt J, Graviss EA, Giordano TP, White AC Jr, et al. Incidence and risk factors for immune reconstitution inflammatory syndrome during highly active antiretroviral therapy. AIDS. 2005;19:399–406. https://doi.org/10.1097/01aids.000161769.06158.8a.
Murdoch DM, Venter WD, Feldman C, Van Rie A. Incidence and risk factors for the immune reconstitution inflammatory syndrome in HIV patients in South Africa: a prospective study. AIDS. 2008;22:601–10. https://doi.org/10.1097/QAD.0b013e3282f4a607.
Michelet C, Arvieux C, Francois C, Besnier JM, Rogez JP, Breux JP, et al. Opportunistic infections occurring during highly active antiretroviral treatment. AIDS. 1998;12:1815–22. https://doi.org/10.1097/00002030-199814000-00013.
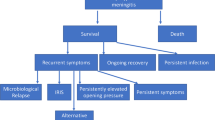
Haddow LJ, Colebunders R, Meintjes G, Lawn SD, Elliott JH, Manabe YC, et al. Cryptococcal immune reconstitution inflammatory syndrome in HIV-1-infected individuals: proposed clinical case definitions. Lancet Infect Dis. 2010;10:791–802. https://doi.org/10.1016/S1473-3099(10)70170-5.
Boulware DR, Bonham SC, Meya DB, Wiesner DL, Park GS, Kambugu A, et al. Paucity of initial cerebrospinal fluid inflammation in cryptococcal meningitis is associated with subsequent immune reconstitution inflammatory syndrome. J Infect Dis. 2010;202:962–70. https://doi.org/10.1086/655785.
Meintjes G, Lawn SD, Scano F, Maartens G, French MA, Worodria W, et al. Tuberculosis-associated immune reconstitution inflammatory syndrome: case definitions for use in resource-limited settings. Lancet Infect Dis. 2008;8:516–23. https://doi.org/10.1016/S1473-3099(08)70184-1.
Phillips P, Bonner S, Gataric N, Bai T, Wilcox P, Hogg R, et al. Nontuberculous mycobacterial immune reconstitution syndrome in HIV-infected patients: spectrum of disease and long-term follow-up. Clin Infect Dis. 2005;41:1483–97. https://doi.org/10.1086/497269.
Manfredi R, Pieri F, Pileri SA, Chiodo F. The changing face of AIDS-related opportunism: cryptococcosis in the highly active antiretroviral therapy (HAART) era. Case reports and literature review. Mycopathologia. 1999;148:73–8. https://doi.org/10.1023/a:1007156027134.
Boelaert JR, Goddeeris KH, Vanopdenbosch LJ, Casselman JW. Relapsing meningitis caused by persistent cryptococcal antigens and immune reconstitution after the initiation of highly active antiretroviral therapy. AIDS. 2004;18:1223–4. https://doi.org/10.1097/00002030-200405210-00023.
Lawn SD, Bekker LG, Myer L, Orrell C, Wood R. Cryptococcocal immune reconstitution disease: a major cause of early mortality in a South African antiretroviral programme. AIDS. 2005;19:2050–2. https://doi.org/10.1097/01.aids.0000191232.16111.f9.
Boulware DR, Meya DB, Muzoora C, Rolfes MA, Huppler Hullsiek K, Musubire A, et al. Timing of antiretroviral therapy after diagnosis of cryptococcal meningitis. N Engl J Med. 2014;37:2487–98. https://doi.org/10.1056/NEJMoa1312884.
••Eshun-Wilson I, Okwen MP, Richardson M, Bicanic T. Early versus delayed antiretroviral treatment in HIV-positive people with cryptococcal meningitis. Cochrane Database Syst Rev. 2018;7:CD009012. https://doi.org/10.1002/14651858.CD009012.pub3This is a review of 4 studies in patients with cryptococcal meningitis from low and middle-income countries. It outlines the relative risk for development of C-IRIS when starting ART early vs. delayed initiation of ART (>4 weeks). It provides the basis for concern for too early an initiation of ART in the setting of cryptococcal meningitis in low income countries.
••Delliere S, Guery R, Candon S, Rammaert B, Aguilar C, Lanternier F, et al. Understanding Pathogenesis and Care Challenges of Immune Reconstitution Inflammatory Syndrome in Fungal Infections. J Fungi (Basel). 2018;4:E139. https://doi.org/10.3390/jof4040139This is an excellent review of the pathophysiology of C-IRIS and the role of various cytokines as well as the role the predominant T-Helper cells play in both disease control and C-IRIS.
Martin-Blondel G, Mars LT, Liblau RS. Pathogenesis of the immune reconstitution inflammatory syndrome in HIV-infected patients. Curr Opin Infect Dis. 2012;25:312–20. https://doi.org/10.1097/QCO.0b013e328352b664.
Bellocchio S, Gaziano R, Bozza S, Rossi G, Montagnoli C, Perruccio K, et al. Liposomal amphotericin B activates antifungal resistance with reduced toxicity by diverting Toll-like receptor signalling from TLR-2 to TLR-4. The Journal of Antimicrob Chemother. 2005;55:214–22. https://doi.org/10.1093/jac/dkh542.
Ben-Ami R, Lewis RE, Kontoyiannis DP. Immunocompromised hosts: immunopharmacology of modern antifungals. Clin Infect Dis. 2008;47:226–35. https://doi.org/10.1086/589290.
Meya DB, Okurut S, Zziwa G, Cose S, Boulware DR, Janoff EN. HIV-Associated Cryptococcal Immune Reconstitution Inflammatory Syndrome Is Associated with Aberrant T Cell Function and Increased Cytokine Responses. J Fungi (Basel). 2019;5:E42. https://doi.org/10.3390/jof5020042.
••Rajasingham R, Wake RM, Beyene T, Katende A, Letang E, Boulware DR. Cryptococcal Meningitis Diagnostics and Screening in the Era of Point-of-Care Laboratory Testing. J Clin Microbiol. 2018;57:e01238–18. https://doi.org/10.1128/JCM.01238-18Important paper on the lateral flow assay and pre-screening HIV-infected patients for cryptococcal infection prior to starting antiretrovial therapy to minimize IRIS.
Singh N, Lortholary O, Alexander BD, Gupta KL, John GT, Pursell K, et al. An immune reconstitution syndrome-like illness associated with Cryptococcus neoformans infection in organ transplant recipients. Clin Infect Dis. 2005;40:1756–61. https://doi.org/10.1086/430606.
Lanternier F, Chandesris MO, Poiree S, Bougnoux ME, Mechai F, Mamzer-Bruneel MF, et al. Cellulitis revealing a cryptococcosis-related immune reconstitution inflammatory syndrome in a renal allograft recipient. Am J Transplant. 2007;7:2826–8. https://doi.org/10.1111/j.1600-6143.2007.01994x.
•Sun HY, Alexander BD, Huprikar S, Forrest GN, Bruno D, Lyon GM, et al. Predictors of immune reconstitution syndrome in organ transplant recipients with cryptococcosis: implications for the management of immunosuppression. Clin Infect Dis. 2015;60:36–44. https://doi.org/10.1093/cid/ciu711This is an important case series looking at variables that pose a risk for development of C-IRIS in Solid Organ Transplant recipients.
Singh N, Lortholary O, Alexander BD, Gupta KL, John GT, Pursell K, et al. Allograft loss in renal transplant recipients with cryptococcus neoformans associated immune reconstitution syndrome. Transplantation. 2005;80:1131–3. https://doi.org/10.1097/01.tp.0000180530.17683.02.
D'Elios MM, Josien R, Manghetti M, Amedei A, de CM, Cuturi MC, et al. Predominant Th1 cell infiltration in acute rejection episodes of human kidney grafts. Kidney Int. 1997;51:1876–84. https://doi.org/10.1038/ki.1997.256.
Wiesner DL, Boulware DR. Cryptococcus-Related Immune Reconstitution Inflammatory Syndrome(IRIS): Pathogenesis and Its Clinical Implications. Curr Fungal Infect Rep. 2011;5:252–61. https://doi.org/10.1007/s12281-011-0064-8.
Ingram PR, Howman R, Leahy MF, Dyer JR. Cryptococcal immune reconstitution inflammatory syndrome following alemtuzumab therapy. Clin Infect Dis. 2007;44:e115–e7. https://doi.org/10.1086/518168.
•Liao TL, Chen YM, Chen DY. Risk factors for cryptococcal infection among patients with rheumatoid arthritis receiving different immunosuppressive medications. Clin Microbiol Infect. 2016;22:815 e1–3. https://doi.org/10.1016/j.cmi.2016.05.030This is a review of the role immunomodulatory agents, in particular monoclonal anti-tumour necrosis factor (TNF) antibodies, may play in increasing risk of cryptococcosis.
Hage CA, Wood KL, Winer-Muram HT, Wilson SJ, Sarosi G, Knox KS. Pulmonary cryptococcosis after initiation of anti-tumor necrosis factor-alpha therapy. Chest. 2003;124:2395–7. https://doi.org/10.1378/chest.124.6.2395.
Marchand T, Revest M, Tattevin P, Chevrier S, Poullot E, Lamy T, et al. Early cryptococcal meningitis following treatment with rituximab, fludarabine and cyclophosphamide in a patient with chronic lymphocytic leukemia. Leuk Lymphoma. 2013;54:643–5. https://doi.org/10.3109/10428194.2012.717081.
Ettahar N, Legout L, Ajana F, Patoz P, Massongo M, Rose C, et al. Cryptococcal osteomyelitis in a patient with a lymphocytic leukemia treated with fludarabine-cyclophosphamide-rituximab. J Mycol Med. 2013;23:57–63. https://doi.org/10.1016/j.mycmed.2012.12.049.
Wingfield T, Jani M, Krutikov M, Mayer J, Uriel A, Marks J, et al. Cryptococcal meningitis in an HIV-negative patient with rheumatoid arthritis treated with rituximab. Rheumatology (Oxford). 2011;50:1725–7. https://doi.org/10.1093/rheumatology/ker210.
Sanchez-Ojanguren J, Isern-Segura I, Chico-Chumillas C, Javaloyas-de-Morlius M. Cryptococcus neoformans meningoencephalitis in a patient treated with rituximab. Med Clin (Barc). 2009;133:157–8. https://doi.org/10.1016/j.medcli.2008.09.036.
Okamoto K, Proia LA, Demarais PL. Disseminated Cryptococcal Disease in a Patient with Chronic Lymphocytic Leukemia on Ibrutinib. Case Rep Infect Dis. 2016;2016:4642831. https://doi.org/10.1155/2016/4642831.
Messina JA, Maziarz EK, Spec A, Kontoyiannis DP, Perfect JR. Disseminated Cryptococcosis With Brain Involvement in Patients With Chronic Lymphoid Malignancies on Ibrutinib. Open Forum Infect Dis. 2017;4:ofw261. https://doi.org/10.1093/ofid/ofw261.
Abid MB, Stromich J, Gundacker ND. Is ibrutinib associated with disseminated cryptococcosis with CNS involvement? Cancer Biol Ther. 2018;20:1–3. https://doi.org/10.1080/15384047.2018.1508622.
••Chamilos G, Lionakis MS, Kontoyiannis DP. Call for Action: Invasive Fungal Infections Associated With Ibrutinib and Other Small Molecule Kinase Inhibitors Targeting Immune Signaling Pathways. Clin Infect Dis. 2018;66:140–8. https://doi.org/10.1093/cid/cix687Excellent review the role on ibrutinib and other small molecule kinase inhibitors may play in increasing the risk of cryptococcosis and other invasive fungal infections. A neglected area of knowledge expanded by this paper.
Wysham NG, Sullivan DR, Allada G. An opportunistic infection associated with ruxolitinib, a novel janus kinase 1,2 inhibitor. Chest. 2013;143:1478–9. https://doi.org/10.1378/chest.12-1604.
Prakash K, Richman D. A case report of disseminated histoplasmosis and concurrent cryptococcal meningitis in a patient treated with ruxolitinib. BMC Infect Dis. 2019;19:287. https://doi.org/10.1186/s12879-019-3922-6.
Liu J, Mouhayar E, Tarrand JJ, Kontoyiannis DP. Fulminant Cryptococcus neoformans infection with fatal pericardial tamponade in a patient with chronic myelomonocytic leukaemia who was treated with ruxolitinib: Case report and review of fungal pericarditis. Mycoses. 2018;61:245–55. https://doi.org/10.1111/myc.12735.
Hirano A, Yamasaki M, Saito N, Iwato K, Daido W, Funaishi K, et al. Pulmonary cryptococcosis in a ruxolitinib-treated patient with primary myelofibrosis. Respir Med Case Rep. 2017;22:87–90. https://doi.org/10.1016/j.rmcr.2017.06.015.
Chen CC, Chen YY, Huang CE. Cryptococcal meningoencephalitis associated with the long-term use of ruxolitinib. Ann Hematol. 2016;95:361–2. https://doi.org/10.1007/s00277-015-2532-7.
•Dioverti MV, Abu Saleh OM, Tande AJ. Infectious complications in patients on treatment with Ruxolitinib: case report and review of the literature. Infect Dis (Lond). 2018:50, 381–357. https://doi.org/10.1080/23744235.2017.1390248This is an excellent review on the role of Ruxolitinib, a Janus kinase 2 inhibitor in increasing the risk of invasive fungal infections.
Reinwald M, Boch T, Hofmann WK, Buchheidt D. Risk of Infectious Complications in Hemato-Oncological Patients Treated with Kinase Inhibitors. Biomark Insights. 2016;10(Suppl 3):55–68. https://doi.org/10.4137/BMI.S22430.
Maziarz EK, Perfect JR. Cryptococcosis. Infect Dis Clin N Am. 2016;30:179–206. https://doi.org/10.1016/j.idc.2015.10.006.
Graybill JR, Sobel J, Saag M, Van der HC, Powderly W, Cloud G, et al. Diagnosis and management of increased intracranial pressure in patients with AIDS and cryptococcal meningitis. The NIAID Mycoses Study Group and AIDS Cooperative Treatment Groups. Clin Infect Dis. 2000;30:47–54. https://doi.org/10.1086/313603.
Sitapati AM, Kao CL, Cachay ER, Masoumi H, Wallis RS, Mathews WC. Treatment of HIV-related inflammatory cerebral cryptococcoma with adalimumab. Clin Infect Dis. 2010;50:e7–10. https://doi.org/10.1086/649553.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Imran Mohamedy declares that he has no conflict of interest.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Fungal Infections
Rights and permissions
About this article
Cite this article
Mohamedy, I., Forrest, G.N. Cryptococcal Immune Reconstitution Inflammatory Syndrome: a Paradoxical Response to a Complex Organism. Curr Treat Options Infect Dis 12, 13–29 (2020). https://doi.org/10.1007/s40506-020-00210-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-020-00210-z