Abstract
Background and Objectives
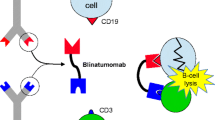
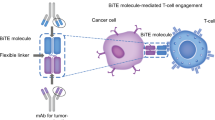
Blinatumomab is a bispecific T-cell engager (BiTE®) antibody construct that transiently links CD19-positive B cells to CD3-positive T cells, resulting in induction of T-cell-mediated serial lysis of B cells and concomitant T-cell proliferation. Blinatumomab showed anti-leukemia activity in clinical trials and was approved by the US Food and Drug Administration for the treatment of Philadelphia chromosome-negative relapsed/refractory B-cell precursor acute lymphoblastic leukemia (r/r ALL). The objectives of this work were to characterize blinatumomab pharmacokinetics and pharmacodynamics and to evaluate dosing regimens.
Methods
Data from six phase I and II trials in patients with r/r ALL, minimal residual disease-positive ALL, and non-Hodgkin’s lymphoma (NHL) were analyzed. Blinatumomab pharmacokinetics was characterized by non-compartmental and population pharmacokinetic analyses and pharmacodynamics was described graphically.
Results
Blinatumomab exhibited linear pharmacokinetics under continuous intravenous infusion for 4–8 weeks per cycle over a dose range of 5–90 µg/m2/day, without target-mediated disposition. Estimated mean (standard deviation) volume of distribution, clearance, and elimination half-life were 4.52 (2.89) L, 2.72 (2.71) L/h, and 2.11 (1.42) h, respectively. Pharmacokinetics was similar in patients with ALL and NHL and was not affected by patient demographics, supporting fixed dosing in adults. Although creatinine clearance was a significant covariate of drug clearance, no dose adjustment was required in patients with mild or moderate renal impairment. Incidence of neutralizing antidrug antibodies was <1 %. Blinatumomab pharmacodynamics featured T-cell redistribution and activation, B-cell depletion, and transient dose-dependent cytokine elevation. Blinatumomab did not affect cytochrome P450 enzymes directly; cytokines may trigger transient cytochrome P450 suppression with low potential for inducing drug interactions.
Conclusions
Blinatumomab has unique pharmacokinetic and immunological features that require indication-dependent dosing regimens. Stepped dosing is required to achieve adequate efficacy and minimize cytokine release in diseases with high tumor burden.





Similar content being viewed by others
References
Bargou R, Leo E, Zugmaier G, Klinger M, Goebeler M, Knop S, et al. Tumor regression in cancer patients by very low doses of a T cell-engaging antibody. Science. 2008;321:974–7.
Nagorsen D, Bargou R, Ruttinger D, Kufer P, Baeuerle PA, Zugmaier G. Immunotherapy of lymphoma and leukemia with T-cell engaging BiTE antibody blinatumomab. Leuk Lymphoma. 2009;50:886–91.
Blincyto™ (blinatumomab) prescribing information. Thousand Oaks, CA, USA: Amgen Inc.; 2014.
Przepiorka D, Ko C-W, Deisseroth A, Yancey CL, Candau-Chacon R, Chiu H-J, et al. FDA approval: blinatumomab. Clin Cancer Res. 2015;21:4035–9.
Klinger M, Brandl C, Zugmaier G, Hijazi Y, Bargou RC, Topp MS, et al. Immunopharmacologic response of patients with B-lineage acute lymphoblastic leukemia to continuous infusion of T cell-engaging CD19/CD3-bispecific BiTE antibody blinatumomab. Blood. 2012;119:6226–33.
Hijazi Y, Klinger M, Schub A, Wu B, Zhu M, Kufer P, et al. Blinatumomab exposure and pharmacodynamic response in patients with non-Hodgkin lymphoma (NHL). J Clin Oncol. 2013;31(15 suppl):Abstract 3051.
Zimmerman Z, Maniar T, Nagorsen D. Unleashing the clinical power of T cells: CD19/CD3 bi-specific T cell engager (BiTE®) antibody construct blinatumomab as a potential therapy. Int Immunol. 2015;27:31–7.
Topp MS, Gökbuget N, Stein AS, Zugmaier G, O’Brien S, Bargou RC, et al. Safety and activity of blinatumomab for adult patients with relapsed or refractory B-precursor acute lymphoblastic leukaemia: a multicentre, single-arm, phase 2 study. Lancet Oncol. 2015;16:57–66.
Huehls AM, Coupet TA, Sentman CL. Bispecific T-cell engagers for cancer immunotherapy. Immunol Cell Biol. 2015;93:290–6.
Raponi S, De Propris MS, Intoppa S, Milani ML, Vitale A, Elia L, et al. Flow cytometric study of potential target antigens (CD19, CD20, CD22, CD33) for antibody-based immunotherapy in acute lymphoblastic leukemia: analysis of 552 cases. Leuk Lymphoma. 2011;52:1098–107.
Nagorsen D, Baeuerle PA. Immunomodulatory therapy of cancer with T cell-engaging BiTE antibody blinatumomab. Exp Cell Res. 2011;317:1255–60.
Haas C, Krinner E, Brischwein K, Hoffmann P, Lutterbuse R, Schlereth B, et al. Mode of cytotoxic action of T cell-engaging BiTE antibody MT110. Immunobiology. 2009;214:441–53.
Anthony DA, Andrews DM, Watt SV, Trapani JA, Smyth MJ. Functional dissection of the granzyme family: cell death and inflammation. Immunol Rev. 2010;235:73–92.
Ewen CL, Kane KP, Bleackley RC. A quarter century of granzymes. Cell Death Differ. 2012;19:28–35.
Voskoboinik I, Dunstone MA, Baran K, Whisstock JC, Trapani JA. Perforin: structure, function, and role in human immunopathology. Immunol Rev. 2010;235:35–54.
Kontermann RE. Dual targeting strategies with bispecific antibodies. mAbs. 2012;4:182–197.
Rathi C, Meibohm B. Clinical pharmacology of bispecific antibody constructs. J Clin Pharmacol. 2015;55:S21–8.
Portell CA, Wenzell CM, Advani AS. Clinical and pharmacologic aspects of blinatumomab in the treatment of B-cell acute lymphoblastic leukemia. Clin Pharmacol. 2013;5:5–11.
Wu B, Sun YN. Pharmacokinetics of peptide-Fc fusion proteins. J Pharm Sci. 2014;103:53–64.
Fielding AK, Richards SM, Chopra R, Lazarus HM, Litzow MR, Buck G, et al. Outcome of 609 adults after relapse of acute lymphoblastic leukemia (ALL); an MRC UKALL12/ECOG 2993 study. Blood. 2007;109:944–50.
Oriol A, Vives S, Hernandez-Rivas JM, Tormo M, Heras I, Rivas C, et al. Outcome after relapse of acute lymphoblastic leukemia in adult patients included in four consecutive risk-adapted trials by the PETHEMA Study Group. Haematologica. 2010;95:589–96.
Nagorsen D. Blinatumomab (Blincyto™) indicated for the treatment of relapsed or refractory B-cell precursor acute lymphoblastic leukemia (Amgen, Inc.). In: Presented at: Accelerating anticancer agent development and validation workshop; May 6–8, 2015; Bethesda, MD, USA.
Cockcroft DW, Gault MH. Prediction of creatinine clearance from serum creatinine. Nephron. 1976;16:31–41.
Xu Y, Hijazi Y, Wolf A, Wu B, Sun YN, Zhu M. Physiologically based pharmacokinetic model to assess the influence of blinatumomab-mediated cytokine elevations on cytochrome P450 enzyme activity. CPT Pharmacomet Syst Pharmacol. 2015;4:507–15.
Schoen T, Blum J, Paccaud F, Burnier M, Bochud M, Conen D, et al. Factors associated with 24-hour urinary volume: the Swiss salt survey. BMC Nephrol. 2013;14:246.
Lobo ED, Hansen RJ, Balthasar JP. Antibody pharmacokinetics and pharmacodynamics. J Pharm Sci. 2004;93:2645–68.
Zhu M, Kratzer A, Johnson J, Holland C, Brandl C, Singh I, et al. Pharmacokinetics/pharmacodynamics (PKPD) of blinatumomab in patients with relapsed/refractory B-precursor acute lymphoblastic leukemia (r/r ALL). J Clin Oncol. 2015;33(15 Suppl):Abstract 2561.
Huang SM, Zhao H, Lee JI, Reynolds K, Zhang L, Temple R, et al. Therapeutic protein-drug interactions and implications for drug development. Clin Pharmacol Ther. 2010;87:497–503.
Sato AK, Viswanathan M, Kent RB, Wood CR. Therapeutic peptides: technological advances driving peptides into development. Curr Opin Biotechnol. 2006;17:638–42.
Roferon® (Interferon alfa-2a) prescribing information. Basel, Switzerland: F. Hoffmann-La Roche Ltd.; 2001.
Neupogen® (filgrastim) prescribing information. Thousand Oaks, CA, USA: Amgen; 2015.
Cerezyme® (imiglucerase for injection) prescribing information. Cambridge, MA, USA: Genzyme Corporation; 2011.
Wu B, Johnson J, Soto M, Ponce M, Calamba D, Sun YN. Investigation of the mechanism of clearance of AMG 386, a selective angiopoietin-1/2 neutralizing peptibody, in splenectomized, nephrectomized, and FcRn knockout rodent models. Pharm Res. 2012;29:1057–65.
Leader B, Baca QJ, Golan DE. Protein therapeutics: a summary and pharmacological classification. Nat Rev Drug Discov. 2008;7:21–39.
Zhao L, Ren TH, Wang DD. Clinical pharmacology considerations in biologics development. Acta Pharmacol Sin. 2012;33:1339–47.
Meibohm B, Zhou H. Characterizing the impact of renal impairment on the clinical pharmacology of biologics. J Clin Pharmacol. 2012;52:54S–62S.
US Food and Drug Administration. Guidance for industry: pharmacokinetics in patients with impaired renal function: study design, data analysis, and impact on dosing and labeling. Draft Guidance. 2010.
Meijer DKF, Ziegler K. Mechanisms for the hepatic clearance of oligopeptides and proteins: implications for rate of elimination, bioavailability, and cell-specific drug delivery to the liver. In: KL Audus TJRE, editor. Biological barriers to protein delivery. New York: Plenum Press; 1993. p. 339–407.
Hoffmann P, Hofmeister R, Brischwein K, Brandl C, Crommer S, Bargou R, et al. Serial killing of tumor cells by cytotoxic T cells redirected with a CD19-/CD3-bispecific single-chain antibody construct. Int J Cancer. 2005;115:98–104.
Lameris R, de Bruin RC, Schneiders FL, van Bergen en Henegouwen PM, Verheul HM, de Gruijl TD, et al. Bispecific antibody platforms for cancer immunotherapy. Crit Rev Oncol Hematol. 2014;92:153–65.
Klinger M, Kufer P, Kirchinger P, Lutterbuese P, Leo E, Reinhardt C, et al. T cell responses during long-term continuous infusion of MT103 (MEDI-538; anti-CD19 BiTE) in patients with relapsed B-NHL: data from dose-escalation study MT103-104. Blood (ASH Annual Meeting Abstracts). 2006;108:Abstract 2725.
Gökbuget N, Dombret H, Bonifacio M, Reichle A, Graux C, Havelange V, et al. BLAST: a confirmatory, single-arm, phase 2 study of blinatumomab, a bispecific T-cell engager (BiTE®) antibody construct, in patients with minimal residual disease B-precursor acute lymphoblastic leukemia (ALL). Blood (ASH Annual Meeting Abstracts). 2014;124:Abstract 379.
Schub A, Nagele V, Zugmaier G, Brandl C, Hijazi Y, Topp MS, et al. Immunopharmacodynamic response to blinatumomab in patients with relapsed/refractory B-precursor acute lymphoblastic leukemia (ALL). J Clin Oncol. 2013;31(15 suppl):Abstract 7020.
Viardot A, Goebeler M, Hess G, Neumann S, Pfreundschuh M, Adrian N, et al. Treatment of relapsed/refractory diffuse large B-cell lymphoma with the bispecific T-cell engager (BiTE®) antibody construct blinatumomab: primary analysis results from an open-label, phase 2 study. Blood (ASH Annual Meeting Abstracts). 2014;124:Abstract 4460.
Acknowledgments
The authors acknowledge Yu-Nien (Tom) Sun and Juan Jose Perez Ruixo for helpful comments during analyses. Micah Robinson (Amgen Inc.) provided medical writing support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was funded by Amgen Inc.
Clinical studies
The clinical studies included in this manuscript have the following ClinicalTrials.gov identifiers: NCT00274742, NCT00560794, NCT01207388, NCT01209286, NCT01741792, and NCT01466179.
Author disclosures
Min Zhu, Benjamin Wu, Christian Brandl, Jessica Johnson, Andreas Wolf, Andrew Chow, and Sameer Doshi are employees of and shareholders in Amgen Inc.
Ethical approval
All procedures performed in the studies involving human participants described here were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the studies described here. Additional informed consent was obtained from all individual participants for whom identifying information is included in this article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhu, M., Wu, B., Brandl, C. et al. Blinatumomab, a Bispecific T-cell Engager (BiTE®) for CD-19 Targeted Cancer Immunotherapy: Clinical Pharmacology and Its Implications. Clin Pharmacokinet 55, 1271–1288 (2016). https://doi.org/10.1007/s40262-016-0405-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40262-016-0405-4