Abstract
Diabetic retinopathy (DR), a blinding disease, is one of the high-incidence chronic complications of diabetes. However, the current treatment for DR is mainly based on advanced pathological changes, which cannot reverse pre-existing retinal tissue damage and visual impairment. Signal transducer and activator of transcription (STAT) proteins are essential in DR through early and late stages. They participate in the early stage of DR through multiple mechanisms and have a strong proangiogenic effect in the late stage. Inhibiting STAT proteins activity has also achieved a significant effect in reversing the pathological changes of DR. Thus, STAT proteins are expected to be an effective therapeutic target in the early stage of DR and can make up for inadequate late treatment. This review introduces the structure, signal transduction mode, and biological functions of STAT proteins in detail and focuses on their role in the mechanism of DR. We also summarize the current research on STAT-related biological agents in DR, aiming to provide a theoretical basis for the treatment of DR.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
The current treatment of diabetic retinopathy (DR) is mainly aimed at the pathological changes in the advanced disease stage, resulting in limited curative effect and irreversible early neurodegeneration. Finding new effective targets to prevent and treat DR is an urgent problem. | |
Signal transducer and activator of transcription (STAT) proteins are involved in the early stage of DR through various mechanisms, including damage to the inner and outer blood retinal barrier, regulation of the activation of immune cells, increased expression of inflammatory mediators, and enhanced oxidative stress, resulting in retinal neurovascular unit damage. | |
STAT proteins have a strong proangiogenic effect in the late stage of DR. STAT inhibitors show significant therapeutic effects. | |
STAT proteins are expected to be an effective therapeutic target in the early stage of DR and can make up for inadequate late treatment. |
Introduction
Diabetic retinopathy (DR), including nonproliferative DR (NPDR) and proliferative DR (PDR), is a type of neurovascular complication of diabetes. NPDR occurs at an early stage and is characterized by increased vascular permeability, while the PDR is characterized by late angiogenesis. The neurovascular unit (NVU) of the retina consists of neurons (optical cells, bipolar cells, ganglion cells, horizontal cells, and amacrine cells), glial cells (Müller cells, astrocytes, and microglia), and vascular cells [endothelial cells (ECs) and pericytes] (Fig. 1). The anatomical and functional homeostasis of these cells is disrupted in DR, leading to disease progression and irreversible blindness [1]. Research indicated that nearly one billion people worldwide had diabetes in 2019, and this number is expected to increase by 25% by 2030 and by 51% by 2045 [2]. DR is the most common cause of new cases of blindness in adults aged 20–74. Nearly all patients with type 1 diabetes (T1D) and more than 60% of those with type 2 diabetes (T2D) have DR in the first 20 years of diabetes [3]. Currently, patients with DR receive various treatments, including systemic control of blood sugar and blood pressure, surgery, laser photocoagulation, and intravitreal injection of steroids or anti-vascular endothelial growth factor (VEGF) drugs [4, 5], but these treatments are mainly aimed at the pathological changes in advanced DR, resulting in irreversible structural abnormalities and visual impairment, with many adverse effects [5]. The diversity of molecules involved in the pathogenesis of DR leads to the unsatisfactory effect of single-molecule drug treatments, such as anti-VEGF therapy. Therefore, there are still many challenges in the treatment of DR, and finding new targets that are more comprehensively involved in the pathogenesis of DR and more effective is an urgent problem.
Drawing of the retinal neurovascular unit (NVU) and the blood retinal barrier. a Structure of the retinal NVU. The retinal NVU consists of neurons (rod, cone, bipolar cell, retinal ganglion cell, horizontal cell, and amacrine cell), glial cells (Müller cell and microglia), and blood vessels. NFL nerve fiber layer, GCL ganglion cell layer, IPL inner plexiform layer, INL inner nuclear layer, OPL outer plexiform layer, ONL outer nuclear layer, RPE retinal pigment epithelium. b Structure of the inner blood retinal barrier (iBRB). The iBRB is mainly composed of capillary endothelial cells. Pericytes and endothelial cells are in close contact. c Structure of the outer blood retinal barrier (oBRB). The oBRB mainly consists of tight junctions between RPE cells
Signal transducer and activator of transcription (STAT) proteins, including STAT1, STAT2, STAT3, STAT4, STAT5a, STAT5b, and STAT6, are a unique family of DNA-binding proteins. They are of great significance in cell proliferation, differentiation, apoptosis, inflammation induction, immune regulation, tumorigenesis, etc. In addition, the biological functions of each STAT protein are different [6].
In this review, we introduce the STAT proteins in detail from aspects of structure, signal transduction mode, and biological function and comprehensively summarize their mechanism in the occurrence and development of DR. Furthermore, we emphasize the potential role of STAT-related biologics in the specific therapy of DR, providing new insights into the treatment of DR.
Methods
Combining the keywords “signal transducer and activator of transcription proteins” and “diabetic retinopathy”, we searched related articles published in PubMed up to July 2022, obtaining a total of 78 articles (including seven reviews). Among them, 50 articles indicating that STAT proteins are correlated or insufficiently correlated with DR were included in this review. Searches were not limited by study design and only articles published in the English language were included.
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Structure, Signal Transduction Mode, and Biological Functions of STAT Proteins
Structures of STAT Proteins
STAT proteins are a transcription factor family composed of STAT1, STAT2, STAT3, STAT4, STAT5a, STAT5b, and STAT6. Among them, STAT1 and STAT3 both have two functionally distinct splicing isomers due to alternative splicing. STAT proteins have six distinct regions, and each of them carries out different important functions: (1) The N-terminal domain (NTD) promotes the formation of dimers and tetramers, which enable them to bind to transcription factors. Additionally, the NTD can regulate the nuclear import of STAT proteins [7]. (2) The coiled-coil domain (CCD) participates in nuclear translocation processes [8] and interacts with interferon regulatory factor-9 (IRF-9), N-myc-interactor (Nmi), and other regulatory proteins to enhance STAT protein-dependent transcription [9, 10]. (3) The DNA-binding domain (DBD) is used to identify and bind to specific DNA sequences of target genes. Similar to the NTD and CCD, the DBD also manages the nuclear import and export of STAT proteins [7, 11]. (4) The α-helical linker domain (LD) regulates the transcriptional activity of STAT proteins and is involved in nuclear export and DNA binding [12, 13]. (5) The Src homology 2 domain (SH2D) is a significant conserved domain of STAT proteins, mediating the interaction between STAT proteins and Janus kinase (JAK). The SH2D also mediates the formation of dimers after it is phosphorylated by receptor-related tyrosine kinases [14]. (6) The C-terminal transcriptional activation domain (TAD), which may be deleted as a result of alternative splicing, is the least conserved region. It contains specific residues phosphorylated during transcription activation and can recruit different coactivators. In addition, it is a target for protein ubiquitination, regulating the stability of proteins [15]. The number of amino acids, the phosphorylation site, and the size of each STAT protein are shown in Fig. 2.
Structure, the number of amino acids, size, and phosphorylation site of each STAT protein. STAT proteins consist of 700–900 amino acids. The size of STAT proteins ranges from 80 to 120 kDa. NTD N-terminal domain, CCD coiled-coil domain, DBD DNA-binding domain, LD linker domain, SH2D Src homology 2 domain, TAD transcriptional activation domain
Signal Transduction Mode of STAT Proteins
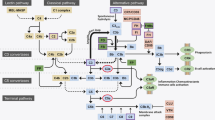
STAT proteins are important components of the JAK-STAT signalling pathway. In addition to STAT proteins, the JAK-STAT system includes ligand–receptor complexes and JAK. The JAK family includes JAK1, JAK2, JAK3, and tyrosine kinase 2 (TYK2) [16, 17]. The process of classic JAK-STAT signalling is as follows: first, interferons (IFNs) [18], interleukin-6 (IL-6) [19], somatotropin (GH) [20], etc. can bind to receptors outside the cell membrane to form ligand–receptor complexes to activate receptors. Then, receptors transmit signals to JAK, which phosphorylates JAK. Furthermore, phosphorylated JAK phosphorylates STAT proteins. Finally, homodimers or heterodimers are formed and transferred to the nucleus, controlling the transcription of genes such as interleukin-1β (IL-1β), IL-6, and tumor necrosis factor alpha (TNFα) [21] as a part of the transcription factor complex, thereby affecting the biological function of cells (Fig. 3).
Signalling and regulation of the JAK-STAT pathway. Black arrows indicate the signalling process. (1) Interferon (IFN), cytokines (CK), or growth hormone (GH) bind to receptors to form receptor–ligand complexes that activate receptors and further phosphorylate Janus kinase (JAK). (2) STAT proteins are activated and form homodimers or heterodimers. (3) STAT homodimers transfer to the nucleus. (4) STAT proteins act as part of a transcription factor complex, controlling the transcription of interleukin-6 (IL-6), interleukin-10 (IL-10), tumor necrosis factor (TNF), transforming growth factor-β (TGFβ), vascular endothelial growth factor (VEGF), and so on. (5) Exerting biological effects. Suppressor of cytokine signalling (SOCS)-1/2/3 and cytokine-inducible SH2 proteins (CIS) can negatively regulate the JAK-STAT pathway by inhibiting activation of GH–receptor–JAK complex. Protein inhibitor of activated STAT (PIAS) negatively regulates the activation of the STAT proteins. The SH domain in protein tyrosine phosphatases (PTPs) can bind to signalling molecules, activated receptors, and JAK to dephosphorylate the substrates. CREB-binding protein (CBP) and P300 are essential coactivators of one of the STAT proteins
Biological Functions of STAT Proteins
STAT proteins have a variety of biological functions, and each STAT protein has different biological effects. We summarize the signalling molecules transduced by each STAT protein (Table 1) and classify their multiple biological functions into the following four aspects.
STAT Proteins can Regulate Cell Proliferation, Differentiation, and Apoptosis
STAT1 can respond to interferon-γ (IFNγ) and other cytokines to negatively regulate cell growth by inducing the cyclin-dependent kinase (CDK) inhibitor P21 [22]. In addition, by upregulating the expression of factor-related apoptosis (Fas) and factor-related apoptosis ligand (FasL), STAT1 can also participate in apoptosis mediated by IFNγ, which induces the inhibition of cell growth [23]. Research has shown that STAT5 can participate in cell growth and the progression of the cell cycle by reducing mitochondrial membrane potential and generating reactive oxygen species. In addition, STAT5b had a greater effect on cell apoptosis than STAT5a did [24]. STAT5b can also participate in cell growth and the progression of the cell cycle by regulating the expression of B cell lymphoma-sss2 (Bcl-2), p21, p27, and VEGF [25].
STAT Proteins are Involved in Tumorigenesis
Some studies have suggested that the expression of STAT1 can promote the invasive growth of tumors [26]. In addition, continuously activated STAT3α and STAT5 can increase the proliferation, survival, and invasion of tumor cells and inhibit antitumor immunity [27]. Interestingly, STAT3α was thought to be helpful in the mechanism of tumor immune escape, while STAT3β was thought to be a negative regulator of tumors [28].
STAT Proteins can Regulate Immune Function and the Inflammatory Response
STAT proteins affect the development and differentiation of T cells. By upregulating the levels of NLRC5 and major histocompatibility complex 1 (MHC-I), STAT1 can protect T cells from being eliminated by NK cells [29], which plays an important role in the survival of T cells. Studies have found that STAT3 promotes the differentiation of Th17 cells via the reversible S-palmitoylation pathway on cysteine [30]. In addition, STAT5a can promote the differentiation of CD4+ T cells into Th2 cells, regulate the development of CD4+CD25+ regulatory T cells [31], and inhibit the differentiation of Th17 cells [32]. In addition, research has shown that STAT6 regulates the expression of inflammatory cytokines and chemokines by preventing the activation of nuclear factor kappa-B (NF-κB) [33].
STAT Proteins can Regulate Lipid Metabolism
STAT proteins participate in lipid metabolism in adipocytes. Activated STAT1 can mediate the effect of IFNγ on reducing insulin sensitivity and inhibiting the differentiation of human adipocytes [34]. STAT5 plays an important role in maintaining lipid homeostasis in white adipose tissue. Loss of STAT5 in adipocytes leads to obesity, insulin resistance, and decreased gluconeogenesis [35]. In addition, some research has suggested that STAT3 also participates in the regulation of lipid metabolism, including blocking the expression of various lipid metabolism genes by inhibiting the JAK-STAT3 pathway [36].
STAT Proteins Are Upregulated and Activated in DR
STAT proteins [41], as important signal transduction molecules and transcriptional regulators of various cytokines, hormones, and growth factors, have been shown to be dysregulated in the serum and ocular tissues of patients with diabetes [48, 49]. Significant increases in STAT5 were found in ocular surface tissues from patients with T1D via conjunctival impression cytology specimens [50]. In addition, the evidence that STAT proteins are upregulated in DR has gradually emerged. Glucose variability (GV) is a contributing factor to diabetic complications, and increased GV predicts PDR and diabetic macular edema in patients with T2D. Notably, studies have shown that GV-related genes are significantly enriched in the JAK-STAT pathway. Gene prioritization analysis identified a high correlation between STAT3 and GV [51], which was carried out using the ToppGene system (BMI CCHMC, Cincinnati, OH, USA) [52] and was based on protein–protein interaction network analyses, demonstrating that STAT proteins are important players in GV. In addition, abnormally high expression levels of STAT1, STAT3, and STAT5 were detected in the serum of patients with DR, retinas of diabetic rats and mice, ECs, Müller cells, and retinal pigment epithelium (RPE) cells induced by high glucose [53,54,55,56,57,58]. After other systemic and ocular diseases were excluded, the serum level of STAT3 was found to be significantly higher in patients with DR (including 18 NPDR, 22 PDR, and 16 PrePDR) than in healthy controls and patients with diabetes but without DR. In addition, serum STAT3 protein levels were also significantly higher in patients with PDR and patients with PrePDR compared with healthy controls. The investigators defined PrePDR as patients with four quadrants of severe retinal hemorrhage, two quadrants of venous bleeding, and loss of capillary perfusion in fundus fluorescein angiography. The researchers did not compare patients with NPDR and healthy controls, thus it is unclear whether there is a significant difference between them [56]. A detailed summary of preclinical studies of STAT protein dysregulation in DR is provided in Table 2.
Mechanisms of STAT Proteins in the Early Stage of DR
STATs Cause Capillary Leakage by Damaging the Inner and Outer Blood Retinal Barrier
The blood retinal barrier (BRB) is divided into inner and outer layers (Fig. 1). The inner BRB (iBRB) is mainly composed of capillary ECs. Pericytes and ECs are in close contact and are surrounded by a common basement membrane. The outer BRB (oBRB) mainly consists of tight junctions between RPE cells. The integrity of the iBRB and oBRB ensures the normal physiological function of ions, proteins, and water entering and leaving the retina [59]. Microvascular changes such as damage to ECs in the iBRB, shedding and loss of pericytes, and thickening of the retinal capillary basement membrane have been shown to appear in the early stage of DR. In contrast, studies on the oBRB in DR are limited, but dysfunction of RPE cells has also been observed. The destruction of the iBRB and oBRB eventually leads to capillary leakage, causing various substances in the circulation to enter the retinal parenchyma and triggering inflammatory and neurotoxic reactions [60, 61]. It is worth noting that STAT proteins can ultimately lead to the destruction of the iBRB and oBRB by affecting the tight junctions and the proliferation, apoptosis, and pyroptosis of ECs, pericytes, and RPE cells.
Mechanisms by Which STAT Proteins Impair the Outer Blood Retinal Barrier
STAT proteins can directly regulate the expression of tight junction proteins in RPE cells. Yang et al. found that angiopoietin-related protein 4 (ANGPTL4) mediated the permeability and migration ability of RPE cells in vitro. Blockade of ANGPTL4 or the use of the STAT3 inhibitor WP1066 inhibited phosphorylation of STAT3 in hypoxia-induced RPE cells, thereby increasing the expression levels of the tight junction proteins ZO-1 and occludin [62]. STAT3 was originally discovered as a component of the acute phase response factor complex upon IL-6 activation [63]. In the streptozocin (STZ) mouse model of diabetes, IL-6-affected RPE cells recruited microglia by increasing the expression of vascular endothelial growth factor A (VEGFA). Microglia, which were also affected by IL-6, upregulated multiple chemokines [monocyte chemotactic protein 1 (MCP-1), macrophage inflammatory protein 1α (MIP-1α), and macrophage inflammatory protein 1β (MIP-1β)] to amplify their own recruitment and produced TNFα, which reduced the expression level of ZO-1 in RPE cells. STAT3 acts as a signal transduction molecule when IL-6 is activated. Its inhibitor WP1066 reversed the effects of IL-6 treatment on microglia and RPE cells [61]. In addition, high-glucose-treated RPE cells could also reduce the expression of occludin and E-cadherin through the miR-132/JAK/STAT3 axis, affecting the mobility and permeability of RPE cells [64]. Moreover, STAT3 can also affect the survival of RPE cells. For example, miR-20b-5p enhanced the proliferation of RPE cells and reduced their apoptosis and pyroptosis by inhibiting the function of STAT3. However, this beneficial marker was downregulated in high-glucose-induced RPE cells and the serum of patients with DR [56]. In conclusion, the ANGPTL4-STAT3, IL-6-STAT3, miR-132-JAK-STAT3, and miR-20b-5p-STAT3 axes have important regulatory roles in the disruption of the oBRB and capillary leakage.
Mechanisms by Which STAT Proteins Impair the Inner Blood Retinal Barrier
STAT proteins can lead to vascular leakage by damaging retinal ECs. Studies have found that high-glucose-induced microglia produce IL-6 to induce the activation of STAT3 in retinal ECs. Activated STAT3 binds to the VEGF promoter and enhances the transcription and expression of VEGF, resulting in decreased expression levels of ZO-1 and occludin and increased EC permeability [65]. Angiopoietin-1 (ANG-1) reverses this effect because it induces SH2-containing protein tyrosine phosphatase-1 (SHP-1) to dissociate from tyrosine-protein kinase receptor (TIE-2) and increases SHP-1 binding to STAT3, thereby downregulating STAT3 activation, resulting in decreased VEGF transcription and restoration of tight junction factor expression [66]. Similarly, miR-146a can reverse the effect of IL-6 on ECs. Its mechanism is to reduce the expression levels of STAT3 and VEGF in high-glucose-induced ECs, thus leading to a reduction in apoptosis [67]. miR-216a can also protect ECs in DR from damage by inhibiting the JAK-STAT pathway [68]. In contrast, miR-19b can promote the proliferation, apoptosis, and inflammation of retinal ECs under high glucose by inhibiting the expression of SOCS6 and thus activating the JAK2-STAT3 pathway, while maternally expressed gene 3 (MEG3), an important long noncoding RNA (lncRNA), can inhibit this effect of miR-19b [69]. In addition, STAT proteins can disrupt the function of pericytes through STAT1. Activation of STAT1 in high-glucose-induced pericytes leads to increased expression of Bcl-2-interacting mediator of cell death (Bim), which further induces oxidative stress and apoptosis in pericytes [70].
STAT Proteins Regulate the Activation of Microglia and Circulating Immune Cells
Retinal microglia are a special class of macrophages that originate from the yolk sac precursor. In DR, microglia participate in early neuronal damage, microvascular lesions, and other pathological processes by mediating inflammatory responses. There are two polarization states of activated microglia in the retina, namely, proinflammatory (M1) and anti-inflammatory (M2), and the imbalance between proinflammatory and anti-inflammatory mechanisms leads to the occurrence or exacerbation of many neurodegenerative changes [71]. In the oxygen-induced retinopathy (OIR) model, which is widely used in the study of large numbers of ocular diseases characterized by retinal angiogenesis, including DR, activation of NF-κB and STAT3 induces M1-type polarization of microglia, leading to upregulation of proinflammatory factors. STAT6 was originally found to be a transcription factor of IL-4 [72]. After stimulation by IL-4, STAT6 is phosphorylated on T641 to exert transcriptional activity. It has also been demonstrated in this model that microglia undergo M2-type polarization and inhibit angiogenesis through the IL-4/STAT6/peroxisome proliferator-activated receptor γ (PPAR-γ) pathway [73]. In DR, the high expression of IFNγ and IL-6 can activate STAT3 in microglia, induce M1-type activation, and produce TNFα, which can inhibit the protein kinase Akt/p70 ribosomal protein S6 kinase (p70S6K) signalling pathway of pericytes, leading to pericyte apoptosis [74]. In addition, the phosphoinositide 3-kinase (PI3K)/AKT/STAT3/NF-κB axis has been shown to lead to excessive proinflammatory activation of microglia in DR [75].
STAT proteins can also induce the activation of circulating immune cells in patients with DR, which are essential for leukocyte adhesion and aggregation in retinal capillaries. Leukocyte arrest is considered a low-grade intravascular inflammatory response that may lead to capillary occlusion and retinal ischemia and ultimately to the destruction of the BRB [59, 76]. Phosphorylated STAT3 in circulating immune cells, including myeloid cells, was upregulated in patients with T1D and mild NPDR, suggesting that STAT3 activation occurred in early DR. Activation of the IL-6/STAT3/suppressor of cytokine signalling 3 (SOCS3) pathway in circulating immune cells of STZ diabetic mice was also detected. Moreover, mice with knockout of SOCS3, a STAT3 inhibitor, exhibited significantly increased leukocyte activation, retinal leukocyte arrest, number of acellular capillaries, and uncontrolled activation of STAT3 in myeloid cells, leading to more severe retinal vasculopathy [77].
STAT Proteins Amplify Inflammation by Regulating the Expression of Proinflammatory Factors
DR is characterized by long duration and chronic tissue destruction of chronic inflammation. When the proinflammatory effect is too strong, neurotoxicity and microvascular pathology can be increased. The occurrence of inflammation has been shown to precede the appearance of microvascular pathological changes [78]. STAT proteins can not only transduce the signals of various inflammatory factors but also affect their transcription. Animal experiments have shown that under the stimulation of interleukin-23 (IL-23), STAT3 can bind to the promoter regions of interleukin-17A (IL-17A) and interleukin-17F (IL-17F) to regulate their transcription [48]. In addition, hyperactivated STAT proteins are also associated with enhanced expression of proinflammatory mediators such as IL-1β, TNFα, and IL-6 [49]. The proinflammatory molecules IL-6, TNFα, transforming growth factor-β1 (TGFβ-1), monocyte chemoattractant protein 1 (MCP-1), and intracellular adhesion molecule 1 (ICAM-1) were upregulated in STZ-induced diabetic rat retinas, whereas CLT-005, an inhibitor of STAT3, restored their expression to nondiabetic levels and decreased the expression levels of keratinocyte chemokine (KC), IL-1β, and interleukin-2 (IL-2) [79]. Moreover, miR-17-3p can inhibit the activation of STAT1 by targeting STAT1, thereby reducing the expression of inflammatory factors and retinal damage [57]. Chen et al. demonstrated that enhanced endoplasmic reticulum stress or overexpression of activating transcription factor 4 (ATF4) induced inflammation of ECs by activating STAT3, whereas knockdown or inhibition of STAT3 reversed these effects [80]. The expression of inflammatory factors and adhesion molecules in ECs can also be induced by IL-6/STAT3 signalling [81].
STAT Proteins Induce Retinal Damage by Mediating Oxidative Stress
Oxidative stress, which can be caused by the protein kinase C (PKC), polyol, hexosamine, and advanced glycation end-products (AGEs) pathways, plays a key role in the pathogenesis of DR and can appear at an early stage. In DR, there is a significant increase in reactive oxygen species (ROS), which is a general term for a class of molecules or ions with high oxidative activity, mainly including superoxide anion, hydrogen peroxide, and hydroxyl radicals. Excessive accumulation of ROS can damage retinal organization and accelerate the progression of DR [82]. Studies have shown that STAT3 can be activated by nicotinamide adenine dinucleotide phosphate (NADPH) oxidase under hyperglycemic conditions, resulting in the induction of ROS [83]. In rats with DR, the inhibitor of STAT3, CLT-005, can reduce the content of malondialdehyde (MDA), one of the important products of membrane lipid peroxidation, and conversely can increase the expression of superoxide dismutase (SOD), an important antioxidant enzyme in vivo. CLT-005 can also improve retinal edema and capillary dilatation, indicating the mediating role of STAT3 in oxidative stress in DR, which is at least partially mediated through the N-myc downstream-regulated gene 2 protein (NDRG2)/IL-6/STAT3 axis [84]. A miR-375 inhibitor can reverse this effect [85].
STAT Proteins Play a Pathogenic Role in Advanced DR by Promoting Angiogenesis
The aforementioned mechanism of STAT proteins involved in DR occurs in the early stage. It is worth noting that STAT proteins also promote angiogenesis in the late stage of DR, the activation of which may run through the whole process of DR. Retinal angiogenesis refers to being under the influence of proangiogenic factors, the growth of abnormally new blood vessels and fibrous tissue on the retina or optic disc along the posterior surface of the vitreous, which occurs during PDR (Fig. 4). When fibrovascular tissue contracts, it stretches the retina, leading to retinal detachment and vision loss in severe cases [86]. Studies have shown that transthyretin (TTR) can upregulate F-box/WD repeat-containing protein 7 (FBXW7) by reducing the expression of STAT4 and miR223-3p and ultimately inhibit the migration and proliferation of ECs and the formation of neovascularization by reducing the expression levels of neurogenic locus notch homologue protein 1 (Notch1) and Akt [87].
Drawing of pathological manifestations and molecular mechanisms of angiogenesis in proliferative diabetic retinopathy (PDR). a Schematic representation of the pathological changes in the PDR. PDR occurs in the late stage of DR and is characterized by abnormal retinal neovascularization. b Schematic diagram of the molecular mechanism of angiogenesis during PDR. Under the influence of proangiogenic molecules, retinal endothelial cells migrate, proliferate, and then develop vascular tube formation and maturation
Fibroblast growth factor 2 (FGF2), TGFβ-1, and ANG-1 have been shown to be essential in angiogenesis during DR. Upregulation of mitsugumin-53 (MG53) in high-glucose retinal ECs decreases their expression by inhibiting the early growth response protein 1 (EGR1)-STAT3 axis, and STAT3 activation reverses this protective effect of MG53 [88]. STAT proteins also have a mutual regulatory relationship with VEGF. STAT3 is activated by the VEGF/VEGF receptor 2 axis, which triggers EC proliferation . VEGF knockdown in retinal ECs cultured with high glucose inhibited the phosphorylation of STAT1 and cell death [58]. Conversely, phosphorylation and nuclear translocation of STAT3 can induce the expression of VEGFA in retinal ECs, stimulating EC migration and angiogenesis under high glucose. This effect can be induced by leptin [89] and NADPH oxidase [83], while being inhibited by lncRNA TPTE pseudogene 1 (TPTEP1) [90]. Interestingly, knockdown of STAT3 in retinal ECs was found to reduce pathological intravitreal neovascularization (IVNV) and maintain retinal thickness in the OIR model but had no effect on physiological retinal vascular development. However, knockdown of VEGFR2 in retinal ECs not only reduced pathological IVNV and increased retinal thickness but also prolonged physiological retinal vascular development [91], indicating the important value of targeting STAT3 in preventing and treating pathological angiogenesis.
Potential Role of STAT-Related Biologics in DR
Methods to inhibit the biological effects of STAT proteins include the use of JAK inhibitors, upstream cytokine/receptor antibodies, and STAT inhibitors. They are widely used in the preclinical and clinical trial stages or in the treatment of various diseases, such as cancer, rheumatoid arthritis, inflammatory bowel disease, and even coronavirus disease 2019 (COVID-19), which is a threat to global public health [92, 93]. Among them, research on JAK inhibitor drugs is rapid, and a variety of drugs have been developed, some of which have been approved for marketing, such as tofacitinib and ruxolitinib. As for the application of JAK inhibitors in ophthalmology, current studies have shown that tofacitinib can be used to treat ulcerative keratitis in patients with rheumatoid arthritis [94], reduce the damage of BRB in patients with T2D [95], and also be used to treat diabetic macular edema. In addition, local infusion of tofacitinib may be helpful in the treatment of corneal inflammation [96]
JAK inhibitors and cytokine/receptor antibodies block signalling to STAT proteins by inhibiting molecules upstream of STAT proteins, thereby preventing STAT protein phosphorylation and activation [97]. Their disadvantages include poor specificity and many side effects. In contrast, STAT inhibitors are more specific and have fewer adverse effects due to inhibition of upstream tyrosine kinases. Drug development of STAT inhibitors is challenging because STAT proteins do not have intrinsic catalytic activity. Most relevant drug research is based on preclinical studies, and few drugs are in clinical trials or approved for marketing. Exogenous STAT inhibitors include peptides and peptidomimetics (such as PY*LKTK, ISS 610, PM-73G, and CJ-1383), small-molecule inhibitors (such as STA-21, S3I-201.1066, and WP1066), natural products and derivatives (such as curcumin, resveratrol, and emodin), and nucleotide-based agents (such as AZD9150) [98, 99].
Research Progress of STAT3-Targeting Inhibitors in DR
Many endogenous molecules can inhibit STAT3 activity in vivo. As described above, microRNA (such as miR-20B-5p, miR-146a, and miR-216a), lncRNA (such as MEG3), and some other factors such as ANG-1 and MG53 could attenuate the pathological changes in DR significantly (Table 3).
In addition, exogenous small-molecule inhibitors of STAT3 (such as WP1006 and CLT-005) (Table 3) were also proved to delay the progress of DR. WP1066 increased the expression levels of ZO-1 and occludin in hypoxia-induced RPE cells or inhibited the inflammatory response of microglia in diabetic mice to reduce damage to RPE cells [61, 62]. CLT-005 binds to SH2D of STAT3 with high affinity, inhibiting its dimerization, nuclear translocation, and binding to target genes. CLT-005 can reduce the oxidative stress level, retinal vascular permeability, and the expression levels of proinflammatory (IL-6, IL-1β, IL-2, TNFα, TGFβ-1, MCP-1, ICAM-1) and proangiogenic molecules [low-density lipoprotein receptor-related protein 5 (LRP-5) and low-density lipoprotein receptor-related protein 6 (LRP-6)] in the retina of diabetic rats [79, 84]. Among them, LRP-5 and LRP-6 are components of the WNT-Frizzled-LRP5/6 complex and are essential molecules in the transduction of the WNT signalling pathway, which coordinates the behavior of ECs. The activation of this pathway in DR leads to vascular leakage and neovascularization [100].
Moreover, the natural products lutein [101], curcumin [102, 103], puerarin [104], ginseng [105], decursin [106], decursinol [106], HuoXueJieDu formula (HXJD) [107], and astragaloside IV [108] are considered ideal candidates for the treatment of retinal diseases, and one of the mechanisms is to target STAT3. Among them, puerarin [104] and HXJD [107] have been shown to improve DR by inhibiting STAT3 in diabetic rat models (Table 3). After administration of HXJD through intragastric gavage in STZ diabetic rats, the oxidative stress and the expression of inflammatory mediators were inhibited by affecting SOCS3-STAT3 and metalloproteinases 1 (TIMP1)-alpha 2 macroglobulin (A2M) pathways. Puerarin treatment can increase the level of SOD and decrease the level of MDA in serum and retinal tissue of STZ diabetic rats. The mechanism is related to the inhibition of STAT3 expression in the retina, indicating that puerarin can alleviate retinal oxidative stress injury by inhibiting STAT3. However, natural products may have broad-spectrum inhibitory effects on biological processes, complex pharmacokinetic problems, unstable efficacy reproducibility, and strong liver and kidney toxicity, leading to most of them being in only the preclinical stage of research.
Utilizing nanoparticles is expected to overcome the deficiencies of natural product inhibitors. Graphene quantum dots (GQDs) are small-scale two-dimensional nanomaterials. In recent years, they have attracted great attention in cancer therapy and other fields owing to their advantages of stability, biocompatibility, and fluorescence tunability. A recent study showed that GQDs could inhibit pathological retinal neovascularization in an OIR model by disrupting the STAT3/periostin/extracellular regulated protein kinase (ERK) pathway and thus regulating the expression levels of cell cycle-related proteins [109] (Table 3).
Several other drugs have also been shown to treat DR by inhibiting STAT3 activity (Table 3). Studies have shown that statin treatment can block NADPH oxidase-mediated activation of STAT3 and ensure the integrity of the BRB in diabetic rats [110]. Rosiglitazone, a thiazolidinedione hypoglycemic agent, attenuates diabetes-induced retinal neuronal apoptosis by inducing SOCS3 and inhibiting STAT3 activation [111]. Tangwang Mingmu granules (TWMM), Qi Ming granules, and calcium dobesilate capsules may inhibit retinal neovascularization by inhibiting the activity of STAT3 [112]. In conclusion, evidence suggests that STAT proteins, especially STAT3, are ideal targets for the treatment of DR, but the potential application of other STAT proteins in the treatment of DR has rarely been reported.
Research Progress of STAT1 and STAT4 Inhibitors in DR
It is a pity that the current research on exogenous inhibitors in DR is focused on STAT3, and there is no related research in other proteins. Only a few endogenous inhibitors such as miR-17-3p and TTR have been shown to target STAT1 and STAT4, respectively, reversing the development of DR in vivo (Table 3).
The results of western blot and real-time quantitative polymerase chain reaction (RT-qPCR) showed that miR-17-3p decreased and STAT1 increased in retinal tissues of mice with DR. After mice with DR were injected with exosomes containing miR-17-3p, the expression levels of TNFα, IL-1β, IL-6, MDA, VEGF, and ROS decreased and the expression levels of SOD and glutathione peroxidase increased by targeting STAT1. miR-17-3p can inhibit the inflammatory response, oxidative damage, and apoptosis of retinal cells in mice with DR [57].
In addition, studies have shown that STAT4 can promote the transcription of miR-223-3p. Compared with healthy controls, miR-223-3p was significantly increased in serum and aqueous humor of patients with NPDR and patients with PDR, and the increase was more obvious in patients with PDR. In human retinal endothelial cells induced by high glucose, TTR downregulates the level of miR-223-3p by inhibiting the expression of STAT4, and finally inhibits angiogenesis [87].
Discussion
STAT proteins are abnormally elevated in DR and are involved in the mechanisms of DR throughout the early and late stages during which they are associated with multiple factors, showing the unique, comprehensive, and powerful effects of STAT proteins in DR. In particular, recently researchers have shown great interest in the link between lncRNAs, microRNAs, and STAT proteins. MEG3, miR-17-3p, miR-132, miR-20b-5p, miR-146a, miR-216a, and miR-19b have been shown to regulate STAT proteins. In DR, the expression levels of STAT proteins are always increased, while those of beneficial miRNAs are decreased. Moreover, STAT proteins are also related to the well-known PI3K-AKT pathway, NF-κB, EGR1, etc., and the interaction of these pathways or factors with STAT proteins may lead to an inflammatory cascade or promote angiogenesis. In the early stage, STAT proteins contribute to DR development through many mechanisms, including damage to the iBRB and oBRB, regulation of the activation of microglia and circulating immune cells, increased expression of inflammatory mediators, and enhanced oxidative stress, leading to damage of retinal NVU by inducing BRB leakage and neurodegeneration. While in the late stage, STAT proteins have a strong angiogenesis effect. They can promote the proliferation and migration of ECs in DR, which leads to the generation of blood vessels, and can also induce the transcription of VEGF. In addition to antagonizing the aforementioned functions, inhibition of STAT proteins activity also reduced the expression of various proangiogenic molecules (FGF2, TGFβ-1, ANG-1, LRP-5, and LRP-6), which helps to compensate for the limitation of anti-VEGF therapy in advanced treatments in DR.
In fact, compared with the research on STAT proteins in inflammation-related and autoimmune diseases, less research has been done on DR. Most of the studies on STAT proteins and DR are based on the preclinical stage, and clinical studies are extremely rare. To date, the research on DR has mainly focused on STAT3 and less on the other STAT proteins. Therefore, more importance should be attached to the other STAT proteins. In addition, the relationship between STAT proteins and advanced DR is relatively mature compared to the early DR. Although STAT proteins are involved in the occurrence of the early DR through multiple mechanisms, there is still a lot of research space for each mechanism. Moreover, research on the role of STAT in DR mostly focuses on ECs, RPE cells, and microglia, while there have been few studies on other cells. Even research on some cells, such as neuronal cells and Müller cells, is still lacking.
Conclusion
STAT proteins are involved in the mechanisms of DR throughout the early and late stages. Inhibiting STAT protein activity has achieved a significant effect in reducing the pathological changes of DR in both early and late stages. Clearly, STAT proteins are an effective potential target for the treatment of DR. Targeting STAT proteins can not only play a role in the early treatment of DR but also make up for some deficiencies in advanced treatment. However, there is still a great space for research on STAT proteins in DR, and more mechanisms of reference significance for drug therapy need to be further explored.
References
Nian S, Lo A, Mi Y, Ren K, Yang D. Neurovascular unit in diabetic retinopathy: pathophysiological roles and potential therapeutical targets. Eye Vis (Lond). 2021;8:15.
Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res Clin Pract. 2019;157:107843.
Bloomgarden ZT. Diabetic retinopathy. Diabetes Care. 2008;31:1080–3.
Antonetti DA, Klein R, Gardner TW. Diabetic retinopathy. N Engl J Med. 2012;366:1227–39.
Jo DH, Kim JH, Kim JH. How to overcome retinal neuropathy: the fight against angiogenesis-related blindness. Arch Pharm Res. 2010;33:1557–65.
Levy DE, Darnell JJ. Stats: transcriptional control and biological impact. Nat Rev Mol Cell Biol. 2002;3:651–62.
Lim CP, Cao X. Structure, function, and regulation of STAT proteins. Mol Biosyst. 2006;2:536–50.
Begitt A, Meyer T, van Rossum M, Vinkemeier U. Nucleocytoplasmic translocation of Stat1 is regulated by a leucine-rich export signal in the coiled-coil domain. Proc Natl Acad Sci USA. 2000;97:10418–23.
Rengachari S, Groiss S, Devos JM, Caron E, Grandvaux N, Panne D. Structural basis of STAT2 recognition by IRF9 reveals molecular insights into ISGF3 function. Proc Natl Acad Sci USA. 2018;115(4):E601–E609.
Zhu M, John S, Berg M, Leonard WJ. Functional association of Nmi with Stat5 and Stat1 in IL-2- and IFNgamma-mediated signaling. Cell. 1999;96:121–30.
Horvath CM, Wen Z, Darnell JE. A STAT protein domain that determines DNA sequence recognition suggests a novel DNA-binding domain. Genes Dev. 1995;9:984–94.
Hüntelmann B, Staab J, Herrmann-Lingen C, Meyer T. A conserved motif in the linker domain of STAT1 transcription factor is required for both recognition and release from high-affinity DNA-binding sites. PLOS One. 2014;9:e97633.
Yang E, Wen Z, Haspel RL, Zhang JJ, Darnell JE. The linker domain of Stat1 is required for gamma interferon-driven transcription. Mol Cell Biol Am Soc Microbiol. 1999;19:5106–12.
Fu XY. A direct signaling pathway through tyrosine kinase activation of SH2 domain-containing transcription factors. J Leukoc Biol. 1995;57:529–35.
Wang D, Moriggl R, Stravopodis D, et al. A small amphipathic alpha-helical region is required for transcriptional activities and proteasome-dependent turnover of the tyrosine-phosphorylated Stat5. EMBO J. 2000;19:392–9.
Leonard WJ, O’Shea JJ. Jaks and STATs: biological implications. Annu Rev Immunol. 1998;16:293–322.
Verbsky JW, Bach EA, Fang YF, Yang L, Randolph DA, Fields LE. Expression of Janus kinase 3 in human endothelial and other non-lymphoid and non-myeloid cells. J Biol Chem. 1996;271:13976–80.
Darnell JE, Kerr IM, Stark GR. Jak-STAT pathways and transcriptional activation in response to IFNs and other extracellular signaling proteins. Science. 1994;264:1415–21.
Johnson DE, O’Keefe RA, Grandis JR. Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev Clin Oncol. 2018;15:234–48.
Carter-Su C, Rui L, Herrington J. Role of the tyrosine kinase JAK2 in signal transduction by growth hormone. Pediatr Nephrol. 2000;14:550–7.
Qin H, Holdbrooks AT, Liu Y, Reynolds SL, Yanagisawa LL, Benveniste EN. SOCS3 deficiency promotes M1 macrophage polarization and inflammation. J Immunol. 2012;189:3439–48.
Chin YE, Kitagawa M, Su WC, You ZH, Iwamoto Y, Fu XY. Cell growth arrest and induction of cyclin-dependent kinase inhibitor p21 WAF1/CIP1 mediated by STAT1. Science. 1996;272:719–22.
Xu X, Fu XY, Plate J, Chong AS. IFN-gamma induces cell growth inhibition by Fas-mediated apoptosis: requirement of STAT1 protein for up-regulation of Fas and FasL expression. Cancer Res. 1998;58:2832–7.
Liang Q-C, Xiong H, Zhao Z-W, et al. Inhibition of transcription factor STAT5b suppresses proliferation, induces G1 cell cycle arrest and reduces tumor cell invasion in human glioblastoma multiforme cells. Cancer Lett. 2009;273:164–71.
Du W, Wang Y-C, Hong J, et al. STAT5 isoforms regulate colorectal cancer cell apoptosis via reduction of mitochondrial membrane potential and generation of reactive oxygen species. J Cell Physiol. 2012;227:2421–9.
Khodarev NN, Roizman B, Weichselbaum RR. Molecular pathways: interferon/stat1 pathway: role in the tumor resistance to genotoxic stress and aggressive growth. Clin Cancer Res. 2012;18:3015–21.
Yu H, Pardoll D, Jove R. STATs in cancer inflammation and immunity: a leading role for STAT3. Nat Rev Cancer. 2009;9:798–809.
Zhang H-X, Yang P-L, Li E-M, Xu L-Y. STAT3beta, a distinct isoform from STAT3. Int J Biochem Cell Biol. 2019;110:130–9.
Kang YH, Biswas A, Field M, Snapper SB. STAT1 signaling shields T cells from NK cell-mediated cytotoxicity. Nat Commun. 2019;10:912.
Zhang M, Zhou L, Xu Y, et al. A STAT3 palmitoylation cycle promotes TH17 differentiation and colitis. Nature. 2020;586:434–9.
Kagami S, Nakajima H, Suto A, et al. Stat5a regulates T helper cell differentiation by several distinct mechanisms. Blood. 2001;97:2358–65.
Laurence A, Tato CM, Davidson TS, et al. Interleukin-2 signaling via STAT5 constrains T helper 17 cell generation. Immunity. 2007;26:371–81.
Lentsch AB, Kato A, Davis B, Wang W, Chao C, Edwards MJ. STAT4 and STAT6 regulate systemic inflammation and protect against lethal endotoxemia. J Clin Invest. 2001;108:1475–82.
McGillicuddy FC, Chiquoine EH, Hinkle CC, et al. Interferon gamma attenuates insulin signaling, lipid storage, and differentiation in human adipocytes via activation of the JAK/STAT pathway. J Biol Chem. 2009;284:31936–44.
Kaltenecker D, Mueller KM, Benedikt P, et al. Adipocyte STAT5 deficiency promotes adiposity and impairs lipid mobilisation in mice. Diabetologia. 2017;60:296–305.
Wang T, Fahrmann JF, Lee H, Li Y-J, Tripathi SC, Yue C, et al. JAK/STAT3-regulated fatty acid β-oxidation is critical for breast cancer stem cell self-renewal and chemoresistance. Cell Metab. 2018;27:1357.
Li J, Williams MJ, Park HJ, et al. STAT1 is essential for HSC function and maintains MHCIIhi stem cells that resists myeloablation and neoplastic expansion. Blood. 2022. https://doi.org/10.1182/blood.2021014009.
Mikhak Z, Fleming CM, Medoff BD, et al. STAT1 in peripheral tissue differentially regulates homing of antigen-specific Th1 and Th2 cells. J Immunol. 2006;176:4959–67.
Lee C-J, An H-J, Kim S-M, et al. FBXW7-mediated stability regulation of signal transducer and activator of transcription 2 in melanoma formation. Proc Natl Acad Sci U S A. 2020;117:584–94.
Yu W, Wang X, Zhao J, et al. Stat2-Drp1 mediated mitochondrial mass increase is necessary for pro-inflammatory differentiation of macrophages. Redox Biol. 2020;37: 101761.
Damasceno LEA, Prado DS, Veras FP, et al. PKM2 promotes Th17 cell differentiation and autoimmune inflammation by fine-tuning STAT3 activation. J Exp Med. 2020;217:e20190613.
Meier JA, Larner AC. Toward a new STATe: the role of STATs in mitochondrial function. Semin Immunol. 2014;26:20–8.
Yang C, Mai H, Peng J, Zhou B, Hou J, Jiang D. STAT4: an immunoregulator contributing to diverse human diseases. Int J Biol Sci. 2020;16:1575–85.
Jacobson NG, Szabo SJ, Weber-Nordt RM, et al. Interleukin 12 signaling in T helper type 1 (Th1) cells involves tyrosine phosphorylation of signal transducer and activator of transcription (Stat)3 and Stat4. J Exp Med. 1995;181(5):1755–62.
Liu X, Robinson GW, Wagner KU, Garrett L, Wynshaw-Boris A, Hennighausen L. Stat5a is mandatory for adult mammary gland development and lactogenesis. Genes Dev. 1997;11:179–86.
Kuperman DA, Huang X, Koth LL, et al. Direct effects of interleukin-13 on epithelial cells cause airway hyperreactivity and mucus overproduction in asthma. Nat Med. 2002;8(8):885–9.
Shimoda K, van Deursen J, Sangster MY, et al. Lack of IL-4-induced Th2 response and IgE class switching in mice with disrupted Stat6 gene. Nature. 1996;380:630–3.
Gurzov EN, Stanley WJ, Pappas EG, Thomas HE, Gough DJ. The JAK/STAT pathway in obesity and diabetes. FEBS J. 2016;283:3002–15.
Dodington DW, Desai HR, Woo M. JAK/STAT—emerging players in metabolism. Trends Endocrinol Metab. 2018;29:55–65.
Di Zazzo A, Coassin M, Micera A, et al. Ocular surface diabetic disease: a neurogenic condition? Ocul Surf. 2021;19:218–23.
Saik OV, Klimontov VV. Bioinformatic reconstruction and analysis of gene networks related to glucose variability in diabetes and its complications. Int J Mol Sci. 2020;21(22):8691.
Chen J, Aronow BJ, Jegga AG. Disease candidate gene identification and prioritization using protein interaction networks. BMC Bioinform. 2009;10:73.
Kim HW, Kim JL, Lee HK, Hur DY, Yun IH, Kim SD. Enalapril alters expression of key growth factors in experimental diabetic retinopathy. Curr Eye Res. 2009;34:976–87.
Wang LL, Chen H, Huang K, Zheng L. Elevated histone acetylations in Müller cells contribute to inflammation: a novel inhibitory effect of minocycline. Glia. 2012;60:1896–905.
Cho CH, Roh KH, Lim NY, Park SJ, Park S, Kim HW. Role of the JAK/STAT pathway in a streptozotocin-induced diabetic retinopathy mouse model. Graefes Arch Clin Exp Ophthalmol. 2022. https://doi.org/10.1007/s00417-022-05694-7.
Liang GH, Luo YN, Wei RZ, et al. CircZNF532 knockdown protects retinal pigment epithelial cells against high glucose-induced apoptosis and pyroptosis by regulating the miR-20b-5p/STAT3 axis. J Diabetes Investig. 2022;13:781–95.
Li W, Jin LY, Cui YB, Xie N. Human umbilical cord mesenchymal stem cells-derived exosomal microRNA-17-3p ameliorates inflammatory reaction and antioxidant injury of mice with diabetic retinopathy via targeting STAT1. Int Immunopharmacol. 2021;90: 107010.
Jiao W, Ji JF, Xu W, et al. Distinct downstream signaling and the roles of VEGF and PlGF in high glucose-mediated injuries of human retinal endothelial cells in culture. Sci Rep. 2019;9:15339.
Cunha-Vaz J, Bernardes R, Lobo C. Blood-retinal barrier. Eur J Ophthalmol. 2011;21(Suppl 6):S3-9.
Altmann C, Schmidt MHH. The role of microglia in diabetic retinopathy: inflammation, microvasculature defects and neurodegeneration. Int J Mol Sci. 2018;19:E110.
Jo DH, Yun JH, Cho CS, Kim JH, Kim JH, Cho CH. Interaction between microglia and retinal pigment epithelial cells determines the integrity of outer blood-retinal barrier in diabetic retinopathy. Glia. 2019;67:321–31.
Yang X, Cao J, Y D, Gong Q, Cheng Y, Su G. Angiopoietin-like protein 4 (ANGPTL4) induces retinal pigment epithelial barrier breakdown by activating signal transducer and activator of transcription 3 (STAT3): evidence from ARPE-19 cells under hypoxic condition and diabetic rats. Med Sci Monit. 2019;25:6742–54.
Zhong Z, Wen Z, Darnell JE. Stat3: a STAT family member activated by tyrosine phosphorylation in response to epidermal growth factor and interleukin-6. Science. 1994;264:95–8.
Wang SS, Liao X, Liu F, Zhang Q, Qiu JJ, Fu SH. miR-132 mediates cell permeability and migration by targeting occludin in high-glucose -induced ARPE-19 cells. Endocr J. 2021;68:531–41.
Yun JH, Park SW, Kim KJ, et al. Endothelial STAT3 activation increases vascular leakage through downregulating tight junction proteins: implications for diabetic retinopathy. J Cell Physiol. 2017;232:1123–34.
Yun JH, Han MH, Jeong HS, Lee DH, Cho CH. Angiopoietin 1 attenuates interleukin-6-induced endothelial cell permeability through SHP-1. Biochem Biophys Res Commun. 2019;518:286–93.
Ye EA, Steinle JJ. miR-146a suppresses STAT3/VEGF pathways and reduces apoptosis through IL-6 signaling in primary human retinal microvascular endothelial cells in high glucose conditions. Vision Res. 2017;139:15–22.
Liu Y, Xiao J, Zhao Y, et al. microRNA-216a protects against human retinal microvascular endothelial cell injury in diabetic retinopathy by suppressing the NOS2/JAK/STAT axis. Exp Mol Pathol. 2020;115:104445.
Xiao F, Li L, Fu JS, Hu YX, Luo R. Regulation of the miR-19b-mediated SOCS6-JAK2/STAT3 pathway by lncRNA MEG3 is involved in high glucose-induced apoptosis in hRMECs. Biosci Rep. 2020;40(7):BSR20194370.
Shin ES, Huang Q, Gurel Z, et al. STAT1-mediated Bim expression promotes the apoptosis of retinal pericytes under high glucose conditions. Cell Death Dis. 2014;5:e986.
Pan WW, Lin F, Fort PE. The innate immune system in diabetic retinopathy. Prog Retin Eye Res. 2021;84: 100940.
Hou J, Schindler U, Henzel WJ, Ho TC, Brasseur M, McKnight SL. An interleukin-4-induced transcription factor: IL-4 Stat. Science. 1994;265:1701–6.
Li J, Yu S, Lu X, et al. The phase changes of M1/M2 phenotype of microglia/macrophage following oxygen-induced retinopathy in mice. Inflamm Res. 2021;70:183–92.
Yun JH, Lee DH, Jeong HS, Kim SH, Ye SK, Cho CH. STAT3 activation in microglia increases pericyte apoptosis in diabetic retinas through TNF-α/AKT/p70S6 kinase signaling. Biochem Biophys Res Commun. 2022;613:133–9.
Tang L, Zhang C, Lu L, et al. Melatonin maintains inner blood-retinal barrier by regulating microglia via inhibition of PI3K/Akt/Stat3/NF-κB signaling pathways in experimental diabetic retinopathy. Front Immunol. 2022;13: 831660.
Kaur C, Foulds WS, Ling EA. Blood-retinal barrier in hypoxic ischaemic conditions: basic concepts, clinical features and management. Prog Retin Eye Res. 2008;27:622–47.
Chen M, Obasanmi G, Armstrong D, et al. STAT3 activation in circulating myeloid-derived cells contributes to retinal microvascular dysfunction in diabetes. J Neuroinflammation. 2019;16:138.
Rübsam A, Parikh S, Fort PE. Role of inflammation in diabetic retinopathy. Int J Mol Sci. 2018;19:E942.
Vanlandingham PA, Nuno DJ, Quiambao AB, et al. Inhibition of Stat3 by a small molecule inhibitor slows vision loss in a rat model of diabetic retinopathy. Invest Ophthalmol Vis Sci. 2017;58:2095–105.
Chen Y, Wang JJ, Li J, et al. Activating transcription factor 4 mediates hyperglycaemia-induced endothelial inflammation and retinal vascular leakage through activation of STAT3 in a mouse model of type 1 diabetes. Diabetologia. 2012;55:2533–45.
Valle ML, Dworshak J, Sharma A, Ibrahim AS, Al-Shabrawey M, Sharma S. Inhibition of interleukin-6 trans-signaling prevents inflammation and endothelial barrier disruption in retinal endothelial cells. Exp Eye Res. 2019;178:27–36.
Kang Q, Yang C. Oxidative stress and diabetic retinopathy: molecular mechanisms, pathogenetic role and therapeutic implications. Redox Biol. 2020;37:101799.
Li J, Wang JJ, Yu Q, Chen K, Mahadev K, Zhang SX. Inhibition of reactive oxygen species by lovastatin downregulates vascular endothelial growth factor expression and ameliorates blood-retinal barrier breakdown in db/db mice: role of NADPH oxidase 4. Diabetes. 2010;59:1528–38.
Wang Y, Zhai WL, Yang YW. Association between NDRG2/IL-6/STAT3 signaling pathway and diabetic retinopathy in rats. Eur Rev Med Pharmacol Sci. 2020;24:3476–84.
Pu LJ, Chen W, Liu QH, Huang AP, Zhao Q, Gu HH. Relationship between miR-375 regulating Ndrg2/IL-6/STAT3 signaling pathway and diabetic retinopathy in rats. Eur Rev Med Pharmacol Sci. 2020;24:2189–95.
Capitão M, Soares R. Angiogenesis and inflammation crosstalk in diabetic retinopathy. J Cell Biochem. 2016;117:2443–53.
Shao J, Fan G, Yin X, et al. A novel transthyretin/STAT4/miR-223-3p/FBXW7 signaling pathway affects neovascularization in diabetic retinopathy. Mol Cell Endocrinol. 2019;498:110541.
Cui KM, Hu ZP, Wang YL. MG53 represses high glucose-induced inflammation and angiogenesis in human retinal endothelial cells by repressing the EGR1/STAT3 axis. Immunopharmacol Immunotoxicol. 2022;(4):484–91.
Suganami E, Takagi H, Ohashi H, et al. Leptin stimulates ischemia-induced retinal neovascularization: possible role of vascular endothelial growth factor expressed in retinal endothelial cells. Diabetes. 2004;53:2443–8.
Sun X, Lu Y, Lei T. TPTEP1 suppresses high glucose-induced dysfunction in retinal vascular endothelial cells by interacting with STAT3 and targeting VEGFA. Acta Diabetol. 2021;58:759–69.
Simmons AB, Bretz CA, Wang H, et al. Gene therapy knockdown of VEGFR2 in retinal endothelial cells to treat retinopathy. Angiogenesis. 2018;21:751–64.
Goker BB, Biray AC. The potential of JAK/STAT pathway inhibition by ruxolitinib in the treatment of COVID-19. Cytokine Growth Factor Rev. 2020;54:51–62.
Cafferkey C, Chau I. Novel STAT 3 inhibitors for treating gastric cancer. Expert Opin Investig Drugs. 2016;25:1023–31.
Meadow PB, Nguyen J, Kesavarapu K. Tofacitinib citrate for ulcerative keratitis in a patient with rheumatoid arthritis. Case Rep Rheumatol. 2014;2014: 403452.
Byrne EM, Llorián-Salvador M, Lyons TJ, Chen M, Xu H. Tofacitinib ameliorates retinal vascular leakage in a murine model of diabetic retinopathy with type 2 diabetes. Int J Mol Sci. 2021;22:11876.
Sakimoto T, Ishimori A. Anti-inflammatory effect of topical administration of tofacitinib on corneal inflammation. Exp Eye Res. 2016;145:110–7.
Xin P, Xu X, Deng C, et al. The role of JAK/STAT signaling pathway and its inhibitors in diseases. Int Immunopharmacol. 2020;80:106210.
Miklossy G, Hilliard TS, Turkson J. Therapeutic modulators of STAT signalling for human diseases. Nat Rev Drug Discov. 2013;12:611–29.
Lai PS, Rosa DA, Magdy AA, et al. A STAT inhibitor patent review: progress since 2011. Expert Opin Ther Pat. 2015;25:1397–421.
Wang Z, Liu CH, Huang S, Chen J. WntSsignaling in vascular eye diseases. Prog Retin Eye Res. 2019;70:110–33.
Ahn YJ, Kim H. Lutein as a modulator of oxidative stress-mediated inflammatory diseases. Antioxidants (Basel). 2021;10(9):1448.
Şahin İO. How curcumin affects hyperglycemia-induced optic nerve damage: a short review. J Chem Neuroanat. 2021;113: 101932.
Chandrasekaran PR, Madanagopalan VG. Role of curcumin in retinal diseases—a review. Graefes Arch Clin Exp Ophthalmol. 2022;260:1457–73.
Cai Y, Zhang X, Xu X, Yu Y. Effects of puerarin on the retina and STAT3 expression in diabetic rats. Exp Ther Med. 2017;14:5480–4.
Liu Y, Zhang H, Dai X, et al. A comprehensive review on the phytochemistry, pharmacokinetics, and antidiabetic effect of ginseng. Phytomedicine. 2021;92: 153717.
Shehzad A, Parveen S, Qureshi M, Subhan F, Lee YS. Decursin and decursinol angelate: molecular mechanism and therapeutic potential in inflammatory diseases. Inflamm Res. 2018;67:209–18.
Wang H, Xing W, Tang S, et al. HuoXueJieDu formula alleviates diabetic retinopathy in rats by inhibiting SOCS3-STAT3 and TIMP1-A2M pathways. Int J Genomics. 2017;2017:4832125.
Zhao ZH, Xu M, Fu C, et al. A Mechanistic exploratory study on the therapeutic efficacy of astragaloside IV against diabetic retinopathy revealed by network pharmacology. Front Pharmacol. 2022;13: 903485.
Zhao N, Gui X, Fang Q, et al. Graphene quantum dots rescue angiogenic retinopathy via blocking STAT3/Periostin/ERK signaling. J Nanobiotechnology. 2022;20:174.
Al-Shabrawey M, Bartoli M, El-Remessy AB, et al. Role of NADPH oxidase and Stat3 in statin-mediated protection against diabetic retinopathy. Invest Ophthalmol Vis Sci. 2008;49:3231–8.
Li P, Xu X, Zheng Z, Zhu B, Shi Y, Liu K. Protective effects of rosiglitazone on retinal neuronal damage in diabetic rats. Curr Eye Res. 2011;36:673–9.
Chen M, Lv H, Gan J, et al. Granule attenuates diabetic retinopathy in type 2 diabetes rats. Front Physiol. 2017;8:1065.
Acknowledgements
Funding
No funding or sponsorship was received for this article. The journal’s Rapid Service Fee was funded by the authors.
Editorial Assistance
This article was edited for proper English language, grammar, punctuation, spelling, and overall style by the highly qualified native English speaking editors at American Journal Experts. Support for this assistance was funded by the authors.
Authorship
All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Author Contributions
Libing Hong conceived of and developed the structural design of the article and wrote the manuscript. Yongqi Li organized the relevant literature, wrote the manuscript, and made important revisions. Guoguo Yi and Min Fu improved the layout and structure of the paper and provided suggestions for important revisions. All the authors reviewed and edited the manuscript, and agreed to the published version of the manuscript.
Disclosures
Libing Hong, Yongqi Lin, Xiongyi Yang, Tong Wu, Yuxi Zhang, Zhuohang Xie, Jieli Yu, Hejia Zhao, Guoguo Yi, and Min Fu declare that they have no competing interests.
Compliance with Ethics Guidelines
This article is based on previously conducted studies and does not contain any new studies with human participants or animals performed by any of the authors.
Data Availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License, which permits any non-commercial use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc/4.0/.
About this article
Cite this article
Hong, L., Lin, Y., Yang, X. et al. A Narrative Review of STAT Proteins in Diabetic Retinopathy: From Mechanisms to Therapeutic Prospects. Ophthalmol Ther 11, 2005–2026 (2022). https://doi.org/10.1007/s40123-022-00581-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40123-022-00581-0