Abstract
Background:
Recruitment and homing cells into graft materials from host tissue is crucial for bone regeneration.
Methods:
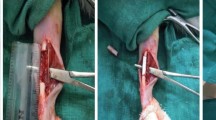
Highly porous, multi-level structural, hydroxyapatite bone void filler (HA-BVF) have been investigated to restore critical size bone defects. The aim was to investigate a feasibility of bone regeneration of synthetic HA-BVF compared to commercial xenograft (Bio-Oss). HA-BVF of 0.7 mm in average diameter was prepared via template coating method. Groups of animals (n = 6) were divided into two with normal (Sham) or induced osteoporotic conditions (Ovx). Subsequently, subdivided into three treated with HA-BVF as an experiment or Bio-Oss as a positive control or no treatment as a negative control (defect). The new bone formation was analyzed by micro-CT and histology.
Results:
At 4 weeks post-surgery, new bone formation was initiated from all groups. At 8 weeks post-surgery, new bone formation in the HA-BVF groups was greater than Bio-Oss groups. Extraordinarily greater bone regeneration within the Ovx-HA group than Sham–Bio-Oss or Ovx–Bio-Oss group (p < 0.05).
Conclusion:
This study suggests that the immediate wicking property of HA-BVF from host tissue activates a natural healing cascade without the addition of exogeneous factors or progenitor cells. HA-BVF may be an effective alternative for repairing bone defects under both normal and osteoporotic bone conditions.





Similar content being viewed by others
References
Petrie Aronin CE, Sadik KW, Lay AL, Rion DB, Tholpady SS, Ogle RC, et al. Comparative effects of scaffold pore size, pore volume, and total void volume on cranial bone healing patterns using microsphere-based scaffolds. J Biomed Mater Res A. 2009;89:632–41.
Guzmán R, Nardecchia S, Gutiérrez MC, Ferrer ML, Ramos V, del Monte F, et al. Chitosan scaffolds containing calcium phosphate salts and rhBMP-2: in vitro and in vivo testing for bone tissue regeneration. PLoS One. 2014;9:e87149.
Jabbarzadeh E, Starnes T, Khan YM, Jiang T, Wirtel AJ, Deng M, et al. Induction of angiogenesis in tissue-engineered scaffolds designed for bone repair: a combined gene therapy-cell transplantation approach. Proc Natl Acad Sci U S A. 2018;105:11099–104.
Cha JK, Lee JS, Kim MS, Choi SH, Cho KS, Jung UW. Sinus augmentation using BMP-2 in a bovine hydroxyapatite/collagen carrier in dogs. J Clin Periodontol. 2014;41:86–93.
Zhao J, Zhang Z, Wang S, Sun X, Zhang X, Chen J, et al. Apatite-coated silk fibroin scaffolds to healing mandibular border defects in canines. Bone. 2009;45:517–27.
Kim HJ, Kim UJ, Kim HS, Li C, Wada M, Leisk GG, et al. Bone tissue engineering with premineralized silk scaffolds. Bone. 2008;42:1226–34.
Karageorgiou V, Kaplan D. Porosity of 3D biomaterial scaffolds and osteogenesis. Biomaterials. 2005;26:5474–91.
Vats A, Tolley NS, Polak JM, Gough JE. Scaffolds and biomaterials for tissue engineering: a review of clinical applications. Clin Otolaryngol Allied Sci. 2003;28:165–72.
Woodard JR, Hilldore AJ, Lan SK, Park CJ, Morgan AW, Eurell JA, et al. The mechanical properties and osteoconductivity of hydroxyapatite bone scaffolds with multi-scale porosity. Biomaterials. 2007;28:45–54.
Zong C, Qian X, Tang Z, Hu Q, Chen J, Gao C, et al. Biocompatibility and bone-repairing effects: comparison between porous poly-lactic-co-glycolic acid and nano-hydroxyapatite/poly(lactic acid) scaffolds. J Biomed Nanotechnol. 2014;10:1091–104.
Unger RE, Sartoris A, Peters K, Motta A, Migliaresi C, Kunkel M, et al. Tissue-like self-assembly in cocultures of endothelial cells and osteoblasts and the formation of microcapillary like structures on three-dimensional porous biomaterials. Biomaterials. 2007;28:3965–76.
Zhang J, Zhou H, Yang K, Yuan Y, Liu C. RhBMP-2-loaded calcium silicate/calcium phosphate cement scaffold with hierarchically porous structure for enhanced bone tissue regeneration. Biomaterials. 2013;34:9381–92.
Hofmann S, Hagenmüller H, Koch AM, Müller R, Vunjak-Novakovic G, Kaplan DL, et al. Control of in vitro tissue-engineered bone-like structures using human mesenchymal stem cells and porous silk scaffolds. Biomaterials. 2007;28:1152–62.
Correia C, Bhumiratana S, Yan LP, Oliveira AL, Gimble JM, Rockwood D, et al. Development of silk-based scaffolds for tissue engineering of bone from human adipose-derived stem cells. Acta Biomater. 2012;8:2483–92.
Bohner M, Loosli Y, Baroud G, Lacroix D. Commentary: deciphering the link between architecture and biological response of a bone graft substitute. Acta Biomater. 2011;7:478–84.
Hing KA, Annaz B, Saeed S, Revell PA, Buckland T. Microporosity enhances bioactivity of synthetic bone graft substitutes. J Mater Sci Mater Med. 2005;16:467–75.
Lan Levengood SK, Polak SJ, Wheeler MB, Maki AJ, Clark SG, Jamison RD, et al. Multiscale osteointegration as a new paradigm for the design of calcium phosphate scaffolds for bone regeneration. Biomaterials. 2010;31:3552–63.
Poole KE, Treece GM, Ridgway GR, Mayhew PM, Borggrefe J, Gee AH. Targeted regeneration of bone in the osteoporotic human femur. PLoS One. 2011;6:e16190.
Leppänen OV, Sievänen H, Jokihaara J, Pajamäki I, Kannus P, Järvinen TL. Pathogenesis of age-related osteoporosis: impaired mechano-responsiveness of bone is not the culprit. PLoS One. 2008;3:e2540.
Khosla S, Westendorf JJ, Oursler MJ. Building bone to reverse osteoporosis and repair fractures. J Clin Invest. 2008;118:421–8.
Dominguez LJ, Scalisi R, Barbagallo M. Therapeutic options in osteoporosis. Acta Biomed. 2010;81 Suppl 1:55–65.
Teófilo JM, Brentegani LG, Lamano-Carvalho TL. Bone healing in osteoporotic female rats following intra-alveolar grafting of bioactive glass. Arch Oral Biol. 2004;49:755–62.
Okazaki A, Koshino T, Saito T, Takagi T. Osseous tissue reaction around hydroxyapatite block implanted into proximal metaphysis of tibia of rat with collagen-induced arthritis. Biomaterials. 2000;21:483–7.
Tami AE, Leitner MM, Baucke MG, Mueller TL, van Lenthe GH, Müller R, et al. Hydroxyapatite particles maintain peri-implant bone mantle during osseointegration in osteoporotic bone. Bone. 2009;45:1117–24.
Xuan F, Lee CU, Son JS, Jeong SM, Choi BH. A comparative study of the regenerative effect of sinus bone grafting with platelet-rich fibrin-mixed Bio-Oss® and commercial fibrin-mixed Bio-Oss®: an experimental study. J Craniomaxillofac Surg. 2014;42:e47–50.
Oh DS, Koch A, Eisig S, Kim SG, Kim YH, Kim DG, et al. Distinctive capillary action by micro-channels in bone-like templates can enhance recruitment of cells for restoration of large bony defect. J Vis Exp. 2015. https://doi.org/10.3791/52947.
Starý V, Douděrová M, Bačáková L. Influence of surface roughness of carbon materials on human osteoblast-like cell growth. J Biomed Mater Res A. 2014;102:1868–79.
Zhou H, Wu X, Wei J, Lu X, Zhang S, Shi J, et al. Stimulated osteoblastic proliferation by mesoporous silica xerogel with high specific surface area. J Mater Sci Mater Med. 2011;22:731–9.
Ito H, Sasaki H, Saito K, Honma S, Yajima Y, Yoshinari M. Response of osteoblast-like cells to zirconia with different surface topography. Dent Mater J. 2013;32:122–9.
Hollinger JO, Kleinschmidt JC. The critical size defect as an experimental model to test bone repair materials. J Craniofac Surg. 1990;1:60–8.
Gomes PS, Fernandes MH. Rodent models in bone-related research: the relevance of calvarial defects in the assessment of bone regeneration strategies. Lab Anim. 2011;45:14–24.
Bosch C, Melsen B, Vargervik K. Importance of the critical size bone defect in testing bone-regenerating materials. J Craniofac Surg. 1998;9:310–6.
Oh DS, Kim YH, Ganbat D, Han MH, Lim P, Back JH, et al. Bone marrow absorption and retention properties of engineered scaffolds with micro-channels and nano-pores for tissue engineering: a proof of concept. Ceram Int. 2013;39:8401–10.
Acknowledgements
This study was partially supported by Chonnam National University, 2016 and Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by Ministry of Education (2017R1D1A1B03034829).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical statement
Animal experimental procedures were approved by the Institutional Animal Care and Use Committee of Chonnam National University (CNU IACUC-YB-R-2014-37) and the animals were cared for in accordance with the Guidelines for Animal Experiments of Chonnam National University.
Rights and permissions
About this article
Cite this article
Kim, S., Ahn, T., Han, MH. et al. Wicking Property of Graft Material Enhanced Bone Regeneration in the Ovariectomized Rat Model. Tissue Eng Regen Med 15, 503–510 (2018). https://doi.org/10.1007/s13770-018-0142-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13770-018-0142-x