Abstract
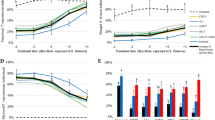
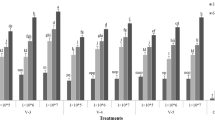
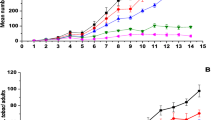
The susceptibility of 1st to 4th instars of Bemisia tabaci (Gennadius, 1989) (Hemiptera: Aleyrodidae) to three isolates of Cordyceps sp. (Hypocreales: Cordycipitaceae) was evaluated in screenhouse experiments under variable temperatures and moisture conditions. No differences in susceptibility to the Cordyceps sp. isolates were observed among 1st, 2nd, and 3rd instar nymphs with respect to median lethal time (LT50) values. Confirmed mortalities ranged from 63.7 to 87.8% when the isolates were tested at 5 × 107 conidia mL−1. The 4th instar was the least susceptible to the fungal isolates (≤ 36.6% mortality). However, 60.0 to 99.5% of the adults that emerged from 4th instar nymphs previously treated with the fungus succumbed to the infection. Temperature was more detrimental to Cordyceps sp. virulence towards B. tabaci nymphs than relative humidity (RH). At similar RH, median LT50 for 1st instar (9.4 days) was higher than for 3rd instar (5.3 days) when the fungus was tested at 5 × 107 conidia mL−1; minimal temperatures of ≥ 12.6°C compared to ≥ 17.0°C were registered for experiments with 1st and 3rd instars, respectively. However, temperatures ≥ 35°C for 4 to 6 h daily did not affect the efficacy of the fungus against nymphs. Cordyceps sp. showed high virulence to all life stages of B. tabaci at relatively low RH, and an ability to grow extensively over the leaf surface and to produce high amounts of conidia on infected hosts. These attributes certainly boost its potential as an important pest control component of B. tabaci biotype B, especially for management of populations resistant to synthetic insecticides.





Similar content being viewed by others
References
Basit M (2019) Status of insecticide resistance in Bemisia tabaci: resistance, cross resistance, stability of resistance, genetics and fitness costs. Phytoparasitica 47:207–225. https://doi.org/10.1007/s12600-019-00722-5
Borisade OA, Magan N (2014) Growth and sporulation of entomopathogenic Beauveria bassiana, Metarhizium anisopliae, Isaria farinosa and Isaria fumosorosea strains in relation to water activity and temperature interactions. Biocontrol Sci Tech 24:999–1011. https://doi.org/10.1080/09583157.2014.909007
Boulard T, Mermier M, Fargues J, Smits N, Rougier M, Roy JC (2002) Tomato leaf boundary layer climate: implications for microbiological whitefly control in greenhouses. Agr Forest Meterol 110:159–176. https://doi.org/10.1016/S0168-1923(01)00292-1
Cabanillas E, Jones WA (2009) Pathogenicity of Isaria sp. (Hypocreales: Clavicipitaceae) against the sweet potato whitefly B biotype, Bemisia tabaci (Hemiptera: Aleyrodidae). Crop Prot 28:333–337. https://doi.org/10.1016/j.cropro.2008.11.015
Cuthbertson AGS, Buxton JH, Blackburn LF, Mathers JJ, Robinson KA, Powell ME, Fleming DA, Bell HA (2012) Eradicating Bemisia tabaci Q biotype on poinsettia plants in the UK. Crop Prot 42:42–48. https://doi.org/10.1016/j.cropro.2012.08.009
Fargues J, Bon MC (2004) Influence of temperature preferences of two Paecilomyces fumosoroseus lineages on their co-infection pattern. J Invertebr Pathol 87:94–104. https://doi.org/10.1016/j.jip.2004.07.001
Fargues J, Maniania NK, Delmas JC, Smits N (1992) Influence de la température sur la croissance in vitro d’hyphomycétes entomopathogénes. Agronomie 12:557–564. https://doi.org/10.1051/agro:19920708
Fargues J, Goettel MS, Smits N, Ouedraogo A, Rougier M (1997) Effect of temperature on vegetative growth of Beauveria bassiana isolates from different origins. Mycologia 89:383–392. https://doi.org/10.1080/00275514.1997.12026797
Faria M, Wraight SP (2001) Biological control of Bemisia tabaci with fungi. Crop Prot 20:767–778. https://doi.org/10.1016/S0261-2194(01)00110-7
Fernandes EKK, Rangel DEN, Braga GUL, Roberts DW (2015) Tolerance of entomopathogenic fungi to ultraviolet radiation: a review on screening of strains and their formulation. Curr Genet 61:427–440. https://doi.org/10.1007/s00294-015-0492-z
Ferron P (1977) Influence of relative humidity on the development of fungal infection caused by Beauveria bassiana (Fungi Imperfecti, Moniliales) in imagines of Acanthoscelides obtectus (Col.: Bruchidae). Entomophaga 22:393–396. https://doi.org/10.1007/BF02373264
Gilbertson RL, Batuman O, Webster CG, Adkins S (2015) Role of the insect supervectors Bemisia tabaci and Frankliniella occidentalis in the emergence and global spread of plant viruses. Annu Rev Virol 2:67–93. https://doi.org/10.1146/annurev-virology-031413-085410
Hesketh H, Roy HE, Eilenberg J, Pell JK, Hails RS (2010) Challenges in modelling complexity of fungal entomopathogens in semi-natural populations of insects. BioControl 55:55–73. https://doi.org/10.1007/s10526-009-9249-2
Horowitz AR, Ishaaya I (2014) Dynamics of biotypes B and Q of the whitefly Bemisia tabaci and its impact on insecticide resistance. Pest Manag Sci 70:1568–1572. https://doi.org/10.1002/ps.3752
Huang Z, Sahar F, Ren S, Ali S (2010) Effect of Isaria fumosorosea on Eretmocerus sp. nr. furuhaskii (Hymenoptera: Aphelinidae), a parasitoid of Bemisia tabaci (Hemiptera: Aleyrodidae). Pakistan J Zool 42:121–127
Ihara F, Toyama M, Mishiro K, Yaginuma K (2008) Laboratory studies on the infection of stink bugs with Metarhizium anisopliae strain FRM515. Appl Entomol Zool 43:503–509. https://doi.org/10.1303/aez.2008.503
Instituto Brasileiro de Geografia e Estatística (IBGE) (2020) Available in https://sidra.ibge.gov.br/home/lspa/brasil>. Accessed in june 29 2020, https://doi.org/10.3310/hsdr08390
Instituto Nacional de Meteorologia. Estação Meteorológica de observação de Superfície Automática (INMET) (2020) Available in http://www.inmet.gov.br/portal/index.php?r=estacoes/estacoesAutomaticas. Accessed in August 1, 2020, https://doi.org/10.3310/hsdr08390
James RR, Buckner JS, Freeman TP (2003) Cuticular lipids and silverleaf whitefly stage affect conidial germination of Beauveria bassiana and Paecilomyces fumosoroseus. J Invertebr Pathol 84:67–74. https://doi.org/10.1016/j.jip.2003.08.006
Jaronski ST (2010) Ecological factors in the inundative use of fungal entomopathogens. BioControl 55:159–185. https://doi.org/10.1007/s10526-009-9248-3
Kepler RM, Luangsa-ard JJ, Hywel-Jones NL, Quandt CA, Sung GH, Rehner SA, Aime MC, Henkel TW, Sanjuan T, Zare R, Chen M, Li Z, Rossman AY, Spatafora JW, Shrestha B (2017) A phylogenetically-based nomenclature for Cordycipitaceae (Hypocreales). IMA Fungus 8:335–353. https://doi.org/10.5598/imafungus.2017.08.02.08
Lacey LA, Fransen JJ, Carruthers RI (1996) Global distribution of naturally occurring fungi of Bemisia, their biologies and use as biological control agentes. In: Gerling D, Mayer RT (eds) Bemisia 1995: taxonomy, biology, damage. Control and Management. Intercept, Andover, pp 356–456
Lacey LA, Wraight SP, Kirk AA (2008) Entomopathogenic fungi for control of Bemisia tabaci biotype B: foreign exploration, research and implementation. In: Gould J, Hoelmer K, Goolsby J (eds) Classical biological control of Bemisia tabaci in the United States - a review of interagency research and implementation. Springer, Netherlands, pp 33–69
Lapidot M, Legg JP, Wintermantel WM, Polston JE (2014) Management of whitefly-transmitted viruses in open-field production systems. Advances in Virus Research. Elsevier, In, pp 147–206. https://doi.org/10.1016/B978-0-12-801246-8.00003-2
Marcandier S, Khachatourians GG (1987) Susceptibility of the migratory grasshopper, Melanoplus sanguinipes (Fab.) (Orthoptera: Acrididae), to Beauveria bassiana (Bals.) Vuillemin (Hyphomycete): influence of relative humidity. Can Entomol 119:901–907. https://doi.org/10.4039/Ent119901-10
Mascarin GM, Kobori NN, Quintela ED, Delalibera I Jr (2013) The virulence of entomopathogenic fungi against Bemisia tabaci biotype B (Hemiptera: Aleyrodidae) and their conidial production using solid substrate fermentation. Biol Control 66:209–218. https://doi.org/10.1016/j.biocontrol.2013.05.001
Mascarin GM, Kobori NN, Quintela ED, Arthurs SP, Delalibera I Jr (2014) Toxicity of non-ionic surfactants and interactions with fungal entomopathogens toward Bemisia tabaci biotype B. BioControl 59:111–123. https://doi.org/10.1007/s10526-013-9543-x
Mascarin GM, Jackson MA, Kobori NN, Behle RW, Delalibera I Jr (2015) Liquid culture fermentation for rapid production of desiccation tolerant blastospores of Beauveria bassiana and Isaria fumosorosea strains. J Invertebr Pathol 127:11–20. https://doi.org/10.1016/j.jip.2014.12.001
Mascarin GM, Pereira-Junior RA, Fernandes EKK, Quintela ED, Dunlap CA, Arthurs SP (2018) Phenotype responses to abiotic stresses, asexual reproduction and virulence among isolates of the entomopathogenic fungus Cordyceps javanica (Hypocreales: Cordycipitaceae). Microbiol Res 216:12–22. https://doi.org/10.1016/j.micres.2018.08.002
McCoy C, Quintela ED, Faria M (2002) Environmental persistence of entomopathogenic fungi. In: Baur ME, Fuxa JR (eds) Factors affecting the survival of entomopathogens. Louisiana State University Agricultural Center, Southern Cooperative Series Bulletin 400:1–9. https://www.researchgate.net/publication/280668546_Factors_Affecting_the_Survival_of_Entomopathogens
Mietkiewski R, Tkaczuk C, Zurek M, Van der Geest LPS (1994) Temperature requirement of four entomopathogenic fungi. Acta Mycol 29:109–120. https://doi.org/10.5586/am.1994.012
Mongkolsamrit S, Noisripoom W, Thanakitpipattana D, Wutikhun T, Spatafora JW, Luangsa-ard J (2018) Disentangling cryptic species with isaria-like morphs in Cordycipitaceae. Mycologia 110:230–257. https://doi.org/10.1080/00275514.2018.1446651
Navas-Castillo J, Fiallo-Olive E, Sanchez-Campos S (2011) Emerging virus diseases transmitted by whiteflies. Annu Rev Phytopathol 49:219–248. https://doi.org/10.1146/annurev-phyto-072910-095235
Osborne LS (1990) Biological control of whiteflies and other pests with a fungal pathogen. IOBC WPRS Bull 13:153–160. https://doi.org/10.2307/3496127
Quintela ED (2004) Manejo integrado de insetos e outros invertebrados pragas do feijoeiro. Inf Agropec 25:113–136
Quintela ED, Abreu AG, Lima JFS, Mascarin GM, Santos JB, Brown JK (2016) Reproduction of the whitefly Bemisia tabaci (Hemiptera: Aleyrodidae) B biotype in maize fields (Zea mays L.) in Brazil. Pest Manag Sci 72:2181–2187. https://doi.org/10.1002/ps.4259
R Core Team (2016) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/
Ramoska WA (1984) The influence of relative humidity on Beauveria bassiana infectivity and replication in the chinch bug, Blissus leucopterus. J Invertebr Pathol 43:389–394. https://doi.org/10.1016/0022-2011(84)90085-5
Riba G, Marcandier S (1984) Influence de l’humidité relative sur l’agressivite et la viabilité des souches de Beauveria bassiana (Bals.) Vuillemin et de Metarhizium anisopliae (Metsch.) Sorokin, hyphomycétes pathogenes de la pyrale du maı¨s, Ostrinia nubilalis Hubn. Agronomie 4:189–194. https://doi.org/10.1051/agro:19840210
Ritz C, Baty F, Streibig JC, Gerhard D (2015) Dose-response analysis using R. PLoS One 10(12):e0146021. https://doi.org/10.1371/journal.pone.0146021
Santos TTM, Quintela ED, Mascarin GM, Santana MV (2017) Enhanced mortality of Bemisia tabaci nymphs by Isaria javanica combined with sublethal doses of chemical insecticides. J Appl Entomol 142:598–609. https://doi.org/10.1111/jen.12504
Scorsetti AC, Humber RA, De Gregório C, Lastra CL (2008) New records of entomopathogenic fungi infecting Bemisia tabaci and Trialeurodes vaporariorum pests of horticultural crops in Argentina. BioControl 53:787–796. https://doi.org/10.1007/s10526-007-9118-9
Shapiro-Ilan DI, Brucky DJ, Lacey LA (2012) Principles of epizootiology and microbial Control. In: Vega FE, Harry KK (eds) Insect Pathology, 2nd edn. Elsevier, Amsterdam, pp 29–72. https://doi.org/10.1016/B978-0-12-384984-7.00003-8
Stansly PA, Naranjo SE (2010) Bemisia: bionomics and management of a global pest. Springer, Amsterdam
Stansly PA, Natwick ET (2010) Integrated systems for managing Bemisia tabaci in protected on open field agriculture. In: Stansly PA, Naranjo SE (eds) Bemisia: bionomics and management of a global pest. Springer, Amsterdam, pp 467–489
Tian J, Diao H, Liang L, Hao C, Arthurs S, Ma S (2015) Pathogenicity of Isaria fumosorosea to Bemisia tabaci, with some observations on the fungal infection process and host immune response. J Invertebr Pathol 130:147–153. https://doi.org/10.1016/j.jip.2015.08.003
Tian J, Diao H, Liang L, Arthurs S, Hao C, Mascarin G, Ma R (2016) Host plants influence susceptibility of whitefly Bemisia tabaci (Hemiptera: Aleyrodidae) to the entomopathogenic fungus Isaria fumosorosea (Hypocreales: Cordycipitaceae). Biocontrol Sci Tech 26:528–538. https://doi.org/10.1080/09583157.2015.1129393
Vidal C, Fargues J, Lacey LA (1997) Intraspecific variability of Paecilomyces fumosoroseus: effect of temperature on vegetative growth. J Invertebr Pathol 70:18–26. https://doi.org/10.1006/jipa.1997.4658
Wraight SP, Carruthers RI, Bradley CA, Jaronski ST, Lacey LA, Wood P, Galaini-Wraight S (1998) Pathogenicity of the entomopathogenic fungi Paecilomyces spp. and Beauveria bassiana against the silverleaf whitefly, Bemisia argentifolii. J Invertebr Pathol 71:217–226. https://doi.org/10.1006/jipa.1997.4734
Wraight SP, Carruthers RI, Jaronski ST, Bradley CA, Garza CJ, Galaini-Wraight S (2000) Evaluation of the entomopathogenic fungi Beauveria bassiana and Paecilomyces fumosoroseus for microbial control of the silverleaf whitefly, Bemisia argentifolii. Biol Control 17:203–217. https://doi.org/10.1006/bcon.1999.0799
Zhang C, Shao ZF, Han YY, Wang XM, Wang ZQ, Musa PD, Ali S (2018) Effects of Aschersonia aleyrodis on the life table and demographic parameters of Bemisia tabaci. J Integr Agric 17:389–396. https://doi.org/10.1016/S2095-3119(17)61773-8
Acknowledgments
The authors thank Edmar Cardoso de Moura (in memoriam) and Edson Djalma for technical support. We also thank Ana Lúcia Delilabera Faria for assistance with the cited references.
Funding
This work was possible through financial support from Lallemand grant no. 0978009 and Embrapa grant no. 021107005, and to the National Council for Scientific and Technological Development (CNPq) for support with the scholarship for the first author no. 132678/2017-9.
Author information
Authors and Affiliations
Contributions
HB and EQ designed the studies. HB and ES performed the bioassays. HB and JFS analyzed the data. HB, EQ and RH wrote the manuscript. All authors reviewed and approved the manuscript before submission.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Edited by Marcos R de Faria – Embrapa
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(PDF 147 KB)
Rights and permissions
About this article
Cite this article
Boaventura, H.A., Quintela, E.D., Santos, E.N. et al. Susceptibility of All Nymphal Stages of Bemisia tabaci Biotype B (Hemiptera: Aleyrodidae) to Three Brazilian Isolates of Cordyceps sp. (Hypocreales: Cordycipitaceae) in a Screenhouse Under Variable Temperature and Moisture Conditions. Neotrop Entomol 50, 100–113 (2021). https://doi.org/10.1007/s13744-020-00823-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13744-020-00823-5