Abstract
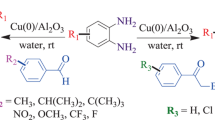
The efficient synthesis of tetrahydro-2,6-dioxopyrimidin-4-yl)-2,3-dihydrophthalazine-1,4-dione derivatives via four-component reaction of hydrazine hydrate, phthalic anhydride, aromatic aldehydes and barbituric acid using vitamin B1 supported on alumina (VB1-Al2O3) as a heterogeneous catalyst under thermal solvent-free conditions in excellent yields is described.






Similar content being viewed by others
References
J. Zhu, H. Bienaym, Multicomponent Reactions (Wiley-VCH, Weinheim, 2005)
R.V.A. Orru, Synthesis of HeterocyclesVia Multicomponent Reactions II (Springer, Berlin Hidelberg, 2010)
S. Grasso, G. DeSarro, N. Micale, M. Zappala, G. Puia, M. Baraldi, C. Demicheli, J. Med. Chem. 43, 2851 (2000)
Y. Nomoto, H. Obase, H. Takai, M. Teranishi, J. Nakamura, K. Kubo, Studies. Chem. Pharm. Bull. 38, 2179 (1990)
N. Watanabe, Y. Kabasawa, Y. Takase, M. Matsukura, K. Miyazaki, H. Ishihara, K. Kodama, H. Adachi, J. Med. Chem. 41, 3367 (1998)
M. Sayyafi, M. Seyyedhamzeh, H.R. Khavasi, A. Bazgir, Tetrahedron 64, 2375 (2008)
G. Sabitha, C. Srinivas, A. Raghavendar, J.S. Yadav, Helv. Chim. Acta 93, 1375 (2010)
M.V. Reddy, C.R. Rani, Y.T. Jeonga, Tetrahedron 70, 3762 (2014)
H.R. Shaterian, M. Mohammadnia, J. Mol. Liquid 177, 353 (2013)
H.R. Shaterian, M. Aghakhanizadeh, Catal. Sci. Technol. 3, 425 (2013)
M. Lei, L. Ma, L. Hua, Tetrahedron Lett. 51, 4746 (2010)
T. Costa, M.R. Gallas, E.V. Benvenutti, J.A.H. Jornada, J. Phys. Chem. 103, 4278 (1999)
H. Kno¨zinger, P. Ratnasamy, Catal. ReV. sSci. Eng. 17, 31 (1978)
Acknowledgments
We are thankful to the University of Sistan and Baluchestan Research Council for the partial support of this research.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Biabangard, A., Shaterian, H.R. Vitamin B1 supported on alumina as an efficient heterogeneous catalyst for synthesis of tetrahydro-2,6-dioxopyrimidin-4-yl)-2,3-dihydrophthalazine-1,4-dione derivatives. J IRAN CHEM SOC 12, 1529–1534 (2015). https://doi.org/10.1007/s13738-015-0623-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13738-015-0623-y