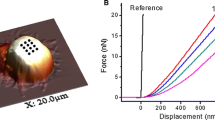
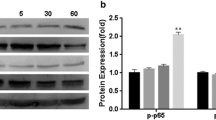
Abstract
Surface stiffness is a unique indicator of various cellular states and events and needs to be tightly controlled. α-Mangostin, a natural compound with numerous bioactivities, reduces the mechanical stiffness of various cells; however, the mechanism by which it affects the actin cytoskeleton remains unclear. We aimed to elucidate the mechanism underlying α-mangostin activity on the surface stiffness of leukocytes. We treated spherical non-adherent myelomonocytic KG-1 cells with α-mangostin; it clearly reduced their surface stiffness and disrupted their microvilli. The α-mangostin-induced reduction in surface stiffness was inhibited by calyculin A, a protein phosphatase inhibitor. α-Mangostin also induced KG-1 cell adhesion to a fibronectin-coated surface. In KG-1 cells, a decrease in surface stiffness and the induction of cell adhesion are largely attributed to the dephosphorylation of ezrin/radixin/moesin proteins (ERMs); α-mangostin reduced the levels of phosphorylated ERMs. It further increased protein kinase C (PKC) activity. α-Mangostin-induced KG-1 cell adhesion and cell surface softness were inhibited by the PKC inhibitor GF109203X. The results of the present study suggest that α-mangostin decreases stiffness and induces adhesion of KG-1 cells via PKC activation and ERM dephosphorylation.










Similar content being viewed by others
References
Jung H-A, Su B-N, Keller WJ, Mehta RG, Kinghorn AD. Antioxidant xanthones from the pericarp of Garcinia mangostana (Mangosteen). J Agric Food Chem. 2006;54:2077–82.
Zuo J, Yin Q, Wang L, Zhang W, Fan Y, Zhou Y-Y, et al. Mangosteen ethanol extract alleviated the severity of collagen-induced arthritis in rats and produced synergistic effects with methotrexate. Pharm Biol. 2018;56:455–64.
Akao Y, Nakagawa Y, Iinuma M, Nozawa Y. Anti-cancer effects of xanthones from pericarps of mangosteen. Int J Mol Sci. 2008;9:355–70.
Phan TKT, Shahbazzadeh F, Pham TTH, Kihara T. Alpha-mangostin inhibits the migration and invasion of A549 lung cancer cells. PeerJ. 2018;6:e5027.
Phan TKT, Shahbazzadeh F, Kihara T. Alpha-mangostin reduces mechanical stiffness of various cells. Hum Cell. 2020;33:347–55.
Kunda P, Pelling AE, Liu T, Baum B. Moesin controls cortical rigidity, cell rounding, and spindle morphogenesis during mitosis. Curr Biol England. 2008;18:91–101.
Matzke R, Jacobson K, Radmacher M. Direct, high-resolution measurement of furrow stiffening during division of adherent cells. Nat Cell Biol. 2001;3:607–10.
Shimizu Y, Haghparast SMA, Kihara T, Miyake J. Cortical rigidity of round cells in mitotic phase and suspended state. Micron. 2012;43:1246–51.
Eiraku M, Takata N, Ishibashi H, Kawada M, Sakakura E, Okuda S, et al. Self-organizing optic-cup morphogenesis in three-dimensional culture. Nature. 2011;472:51–6.
Eiraku M, Adachi T, Sasai Y. Relaxation-expansion model for self-driven retinal morphogenesis: a hypothesis from the perspective of biosystems dynamics at the multi-cellular level. BioEssays. 2012;34:17–25.
Tachibana K, Ohnishi H, Ali Haghparast SM, Kihara T, Miyake J. Activation of PKC induces leukocyte adhesion by the dephosphorylation of ERM. Biochem Biophys Res Commun. 2020;523:177–82.
Kihara T, Matsumoto T, Nakahashi Y, Tachibana K. Mechanical stiffness softening and cell adhesion are coordinately regulated by ERM dephosphorylation in KG-1 cells. Hum Cell. 2021;34:1709–16.
Collinsworth AM, Zhang S, Kraus WE, Truskey GA. Apparent elastic modulus and hysteresis of skeletal muscle cells throughout differentiation. Am J Physiol Cell Physiol. 2002;283:C1219–27.
Dai J, Sheetz MP. Mechanical properties of neuronal growth cone membranes studied by tether formation with laser optical tweezers. Biophys J. 1995;68:988–96.
Guilak F, Erickson GR, Ting-Beall HP. The effects of osmotic stress on the viscoelastic and physical properties of articular chondrocytes. Biophys J. 2002;82:720–7.
Trickey WR, Vail TP, Guilak F. The role of the cytoskeleton in the viscoelastic properties of human articular chondrocytes. J Orthop Res. 2004;22:131–9.
Niggli V, Rossy J. Ezrin/radixin/moesin: versatile controllers of signaling molecules and of the cortical cytoskeleton. Int J Biochem Cell Biol. 2008;40:344–9.
Dustin ML, Springer TA. T-cell receptor cross-linking transiently stimulates adhesiveness through LFA-1. Nature. 1989;341:619–24.
Shimizu Y, Newman W, Gopal TV, Horgan KJ, Graber N, Beall LD, et al. Four molecular pathways of T cell adhesion to endothelial cells: roles of LFA-1, VCAM-1, and ELAM-1 and changes in pathway hierarchy under different activation conditions. J Cell Biol. 1991;113:1203–12.
van Kooyk Y, van de Wiel-van KP, Weder P, Kuijpers TW, Figdor CG. Enhancement of LFA-1-mediated cell adhesion by triggering through CD2 or CD3 on T lymphocytes. Nature. 1989;342:811–3.
Fiévet B, Louvard D, Arpin M. ERM proteins in epithelial cell organization and functions. Biochim Biophys Acta. 2007;1773:653–60.
Tachibana K, Haghparast SM, Miyake J. Inhibition of cell adhesion by phosphorylated Ezrin/Radixin/Moesin. Cell Adh Migr. 2015;9:502–12.
Tachibana K, Yamane J, Haghparast SMA, Miyake J. Sialomucin and phosphorylated-ERM are inhibitors for cadherin-mediated aggregate formation. Biochem Biophys Res Commun. 2019;520:159–65.
Brown MJ, Nijhara R, Hallam JA, Gignac M, Yamada KM, Erlandsen SL, et al. Chemokine stimulation of human peripheral blood T lymphocytes induces rapid dephosphorylation of ERM proteins, which facilitates loss of microvilli and polarization. Blood. 2003;102:3890–9.
Haghparast SMA, Kihara T, Shimizu Y, Yuba S, Miyake J. Actin-based biomechanical features of suspended normal and cancer cells. J Biosci Bioeng. 2013;116:380–5.
Kato K, Umezawa K, Funeriu DP, Miyake M, Miyake J, Nagamune T. Immobilized culture of nonadherent cells on an oleyl poly(ethylene glycol) ether-modified surface. Biotechniques. 2003;35:1014–21.
Ohnishi H, Sasaki H, Nakamura Y, Kato S, Ando K, Narimatsu H, et al. Regulation of cell shape and adhesion by CD34. Cell Adh Migr. 2013;7:426–33.
Charras GT, Hu C-K, Coughlin M, Mitchison TJ. Reassembly of contractile actin cortex in cell blebs. J Cell Biol. 2006;175:477–90.
Yamane J, Ohnishi H, Sasaki H, Narimatsu H, Ohgushi H, Tachibana K. Formation of microvilli and phosphorylation of ERM family proteins by CD43, a potent inhibitor for cell adhesion: cell detachment is a potential cue for ERM phosphorylation and organization of cell morphology. Cell Adh Migr. 2011;5:119–32.
Fang Y, Su T, Qiu X, Mao P, Xu Y, Hu Z, et al. Protective effect of alpha-mangostin against oxidative stress induced-retinal cell death. Sci Rep. 2016;6:21018.
Faure S, Salazar-Fontana LI, Semichon M, Tybulewicz VLJ, Bismuth G, Trautmann A, et al. ERM proteins regulate cytoskeleton relaxation promoting T cell–APC conjugation. Nat Immunol. 2004;5:272–9.
Allenspach EJ, Cullinan P, Tong J, Tang Q, Tesciuba AG, Cannon JL, et al. ERM-dependent movement of CD43 defines a novel protein complex distal to the immunological synapse. Immunity. 2001;15:739–50.
Delon J, Kaibuchi K, Germain RN. Exclusion of CD43 from the immunological synapse is mediated by phosphorylation-regulated relocation of the cytoskeletal adaptor moesin. Immunity. 2001;15:691–701.
Roumier A, Olivo-Marin JC, Arpin M, Michel F, Martin M, Mangeat P, et al. The membrane-microfilament linker ezrin is involved in the formation of the immunological synapse and in T cell activation. Immunity. 2001;15:715–28.
Jadhav S, Eggleton CD, Konstantopoulos K. A 3-D computational model predicts that cell deformation affects selectin-mediated leukocyte rolling. Biophys J. 2005;88:96–104.
Wojcikiewicz EP, Zhang X, Chen A, Moy VT. Contributions of molecular binding events and cellular compliance to the modulation of leukocyte adhesion. J Cell Sci. 2003;116:2531–9.
García-Ortiz A, Serrador JM. ERM proteins at the crossroad of leukocyte polarization, migration and intercellular adhesion. Int J Mol Sci. 2020;21:1502.
Jinsart W, Ternai B, Buddhasukh D, Polya GM. Inhibition of wheat embryo calcium-dependent protein kinase and other kinases by mangostin and gamma-mangostin. Phytochemistry. 1992;31:3711–3.
Itoh T, Ohguchi K, Iinuma M, Nozawa Y, Akao Y. Inhibitory effect of xanthones isolated from the pericarp of Garcinia mangostana L. on rat basophilic leukemia RBL-2H3 cell degranulation. Bioorg Med Chem. 2008;16:4500–8.
Clucas J, Valderrama F. ERM proteins in cancer progression. J Cell Sci. 2014;127:267–75.
Ilmonen S, Vaheri A, Asko-Seljavaara S, Carpen O. Ezrin in primary cutaneous melanoma. Mod Pathol. 2005;18:503–10.
Khanna C, Wan X, Bose S, Cassaday R, Olomu O, Mendoza A, et al. The membrane-cytoskeleton linker ezrin is necessary for osteosarcoma metastasis. Nat Med. 2004;10:182–6.
Pang S-T, Fang X, Valdman A, Norstedt G, Pousette A, Egevad L, et al. Expression of ezrin in prostatic intraepithelial neoplasia. Urology. 2004;63:609–12.
Hung SH, Shen KH, Wu CH, Liu CL, Shih YW. Alpha-mangostin suppresses PC-3 human prostate carcinoma cell metastasis by inhibiting matrix metalloproteinase-2/9 and urokinase-plasminogen expression through the JNK signaling pathway. J Agric Food Chem. 2009;57:1291–8.
Lee YB, Ko KC, Shi MD, Liao YC, Chiang TA, Wu PF, et al. alpha-Mangostin, a novel dietary xanthone, suppresses TPA-mediated MMP-2 and MMP-9 expressions through the ERK signaling pathway in MCF-7 human breast adenocarcinoma cells. J Food Sci. 2010;75:H13-23.
Shibata MA, Iinuma M, Morimoto J, Kurose H, Akamatsu K, Okuno Y, et al. alpha-Mangostin extracted from the pericarp of the mangosteen (Garcinia mangostana Linn) reduces tumor growth and lymph node metastasis in an immunocompetent xenograft model of metastatic mammary cancer carrying a p53 mutation. BMC Med. 2011;9:69.
Shih YW, Chien ST, Chen PS, Lee JH, Wu SH, Yin LT. Alpha-mangostin suppresses phorbol 12-myristate 13-acetate-induced MMP-2/MMP-9 expressions via alphavbeta3 integrin/FAK/ERK and NF-kappaB signaling pathway in human lung adenocarcinoma A549 cells. Cell Biochem Biophys. 2010;58:31–44.
Acknowledgements
This work was partly conducted at the Instrumentation Center, The University of Kitakyushu.
Funding
This work was supported by JSPS KAKENHI Grant Number JP21K04797, MEXT Promotion of Distinctive Joint Research Center Program Grant Number JPMXP0621467946, and grant for Young Scientists, Institute of Environmental Science and Technology, The University of Kitakyushu.
Author information
Authors and Affiliations
Contributions
TKTP, KT, and TK conceived and designed the work. TKTP, TLD, and TK performed the experiments. TKTP, TLD, KT, and TK analyzed and interpreted the data. TKTP, TLD, and TK drafted the manuscript. KT revised the manuscript. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
This work does not involve human participants or their data. In this study, we used KG-1 cell line RRID:CVCL_0374 and U937 DE-4 cell line RRID:CVCL_8765, which obtained from RIKEN BioResource Center (Ibaraki, Japan).
Informed consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Phan, T.K.T., Do, T.L., Tachibana, K. et al. Alpha-mangostin dephosphorylates ERM to induce adhesion and decrease surface stiffness in KG-1 cells. Human Cell 35, 189–198 (2022). https://doi.org/10.1007/s13577-021-00651-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13577-021-00651-8