Abstract
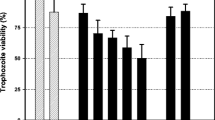
Trichomoniasis, a common cause of vaginitis, is the most common sexually transmitted infection in humans, worldwide. It is caused by infection with the single-celled protozoan parasite, Trichomonas vaginalis, which disrupts normal vaginal flora. The administration of metronidazole, approved by the WHO, is currently being used for trichomoniasis treatment. However, there is a great need for development of safe alternative drugs from natural products because of metronidazole’s adverse effects. In this investigation, bee venom effectively inhibited Trichomonas vaginalis growth in a concentration-dependent manner.
Similar content being viewed by others
References
Pertrin, D., Delgaty, K., Bhatt, R. & Garber, G. Clinical and microbiological aspects of Trichomonas vaginalis. Clin. Microbiol Rev. 11, 300–317 (1998).
Ryu, J. S. & Min, D. Y. Trichomonas vaginalis and trichomoniasis in the Republic of Korea. Korean J. Parasitol. 44, 101–116 (2006).
Tiwari, P., Sihgh, D. & Singh, M. M. Anti-Trichomonas activity of Sapindus saponins, a candidate for development as microbicidal contraceptive. J. Antimicrob. Chemother. 62, 526–534 (2008).
Meingassner, J. G., Havelec, L. & Mieth, H. Studies on strain sensitivity of Trichomonas vaginalis to metronidazole. Br. J. Vener. Dis. 54, 72–76 (1978).
Wolner-Hanssen, P. et al. Clinical manifestations of vaginal trichomoniasis. JAMA 261, 571–576 (1989).
Dunne, R. L., Dunn, L. A., O’Donoqhue, P. J. & Upcroft, J. A. Drug resistance in the sexually transmitted protozoan Trichomonas vaginalis. Cell Res. 13, 239–249 (2003).
De Jesus, J. B. et al. A further proteomic study on the effect of iron in the human pathogen Trichomonas vaginalis. Proteomics 12, 1961–1972 (2007).
Lenker, M. W., Arroyo, R. & Alderete, J. F. The regulation of iron of the synthesis of adhesions and cytoadherence levels in the protozoan. Trichomonas vaginalis. J. Exp. Med. 174, 311–318 (1991).
Ribeiro, K. C., Mariante, R. M., Coutinho, L. L. & Benchimol, M. Nucleus behavior during the closed mitosis of Tritrichomonas foetus. Biol. Cell. 94, 289–301 (2002).
Lindmark, D. G. & Muller, M. Hydrogenosome, a cytoplasmic organelle of the anaerobic flagellate Tritrichomonas foetus, and its role in pyruvate metabolism. J. Biol. Chem. 248, 7724–7728 (1973).
ter Kuile, B. H. Metabolic adaptation of Trichomonas vagianlis to growth rate and glucose availability. Microbiology 142, 3337–3345 (1996).
Bouma, M. J. et al. Acitivity of disulfiram(bis(diethylthiocarbamoyl) disulphide) and ditiocarb (diethyldothiocarbamate) against metronidazole-sensitibe and — resistant Trichomonas vaginalis and Tritrichomonas foetus. J. Antimicrob. Chemother. 42, 817–820 (1998).
Kirkcaldy, R. D. et al. Trichomonas vaginalis antimicrobial drug resistance in 6 US cities, STD surveillance network, 2009–2010. Emerg. Infect. Dis. 18, 939–943 (2012).
Ryang, Y. S. et al. Antiparasitic effects of a herb extract from Gentiana scabra var. buergeri on Trichomonas vaginalis. Korean J. Biomed. Lab Sci. 7, 53–58 (2001).
EI-Sherbiny, G. M. & EI-Sherbiny, E. T. The effect of Commiphora molmol (Myrrh) in treatment of Trichomoniasis vaginalis infection. Iran Red Crescent Med. J. 13, 480–486 (2011).
Lee, Y. J. et al. Cytotoxicity of honeybee (Apis mellifera L.) venom in normal human lymphocytes and HL-60 cells. Chem. Biol. Interact. 169, 189–197 (2007).
Huh, J. E. et al. Bee venom inhibits tumor angiogenesis and metastasis by inhibiting tyrosine phophorylation of VEGFR-2 in LLC-tumor-bearing mice. Cancer Lett. 292, 98–110 (2010).
Han, S. M., Lee, K. G., Yeo, J. H., Baek, H. J. & Park, K. K. Antibacterial and anti-inflammatory effects of honeybee (Apis mellifera L.) venom against acne-inducing bacteria. J. Med. Plants Res. 4, 459–464 (2010).
Ahn, B. J. & Song, H. S. Effect of bee venom death receptor dependent apoptosis and JAK2/STAT3 pathway in the ovarian cancer. The Acupuncture 29, 47–59 (2012).
Ryu, J. S., Min, D. Y., Kim, M. C., Kim, N. S. & Shin, M. H. In vitro activities of 2,2′-dipyridyl against Trichomonas vaginalis, Candida albicans, and Gardnerella vaginalis. J. Microbiol. Biotechnol. 11, 124–130 (2001).
Ertabaklar, H., Kivcak, B., Mert, T. & Ozensoy Töz, S. In vitro activity of Arvutus unedo leaf extracts against Trichomonas vaginalis triphozoites. Turkiye Parazitol. Derg. 33, 263–265 (2009).
Yu, A. R. et al. The antifungal activity of bee venom against dermatophytes. J. Appl. Biol. Chem. 55, 7–11 (2012).
Stocker, J. F. & Traynor, J. R. The action of various venoms on Escherichia coli. J. Appl. Bacteriol. 61, 383–388 (1986).
Perumal, S. R. et al. Antibacterial activity of snake, scorpion and bee venoms: A comparison with purified venom phopholipase A2 enzymes. J. Appl. Microbiol. 102, 650–659 (2007).
Nakatuji, T. et al. Antimicrobial property of lauric acid against Propionibacterium acnes: Its therapeutic potential for inflammatory acne vulgaris. J. Invest Dermatol. 129, 2480–2488 (2009).
Hoe, S. T. & Crabbe, M. J. Kinetic effects of metal ion chelating reagents and their analogues on bovine lens aldehyde dehydrogenase. Exp. Eye Res. 44, 663–675 (1987).
Horky, L. L., Pluta, R. M., Boock, R. J. & Oldifield, E. H. Role of ferrous iron chelator 2,2′-dipyridyl in preventing delayed vasospasm in a primate model of subarachnoid hemorrhage. J. Neurosurg. 88, 298–303 (1998).
Kerbs, S., Hutton, R. & Lancaster, M. Effects of deferoxamine methanesulfonate on Trichophyton mentagrophytes. Sabouraudia. 17, 241–250 (1979).
Park, S. K., Jin, E. S., Lee, C. G. & Lee, M. Y. High light-induced changes in the activities of antioxidant enzymes and the accumulation of astaxanthin in the green alga Haematococcus pluvialis. Mol. Cell Toxicol. 4, 300–306 (2008).
Choi, G. M. & Lee, M. Y. Differential protein expression associated with heat stress in antarctic microalga. BioChip J. 6, 271–279 (2012).
Ellis, J. E., Yarlett, N., Cole, D., Humphreys, M. J. & Lloyd, D. Antioxidant defences in the microaerophilic protozoan Trichomonas vaginalis: comparison of metronidazole-resistant and sensitive strains. Microbiology 140, 2489–2494 (1994).
Voncken, F. et al. Multiple origins of hydrogenosomes: finctional and phylogenetic evidence from the ADP/ ATP carrier of the anaerobic chytrid Neocallimastix sp. Mol. Microbiol. 44, 1441–1454 (2002).
Gelbart, S. M. et al. Growth of Trichomonas vaginalis in commercial culture media. J. Clin. Microbiol. 28, 962–964 (1990).
Ryu, J. S., Chio, R., Park, S. Y., Park, H. & Min, D. Y. Biological and biochemical modulation of Trichomonas vaginalis KT9 isolate after shifting of culture medium from TPS-1 into TYM. Korean J. Parasitol. 36, 255–260 (1998).
Smith, R. F. Viability of Trichomonas vaginalis in vitro at four temperatures. J. Clin. Microbiol. 18, 834–836 (1983).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, JH., Ryu, JS. & Lee, MY. Inhibitory effect of bee venom on the growth of Trichomonas vaginalis . Toxicol. Environ. Health Sci. 6, 48–53 (2014). https://doi.org/10.1007/s13530-014-0187-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13530-014-0187-8