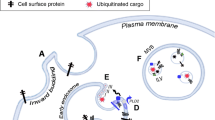
Abstract
Extracellular vesicles (EVs) are cell-released, membranous structures essential for intercellular communication. The biochemical compositions and physiological impacts of exosomes, lipid-bound, endosomal origin EVs, have been focused on, especially on the tumor-host interactions in a defined tumor microenvironment (TME). Despite recent progress in targeted therapy and cancer immunotherapy in colorectal cancer (CRC), cancer patients still suffer from distal metastasis and tumor relapse, suggesting unmet needs for biomarkers directing therapeutic interventions and predicting treatment responsiveness. As exosomes are indispensable for intercellular communication and high exosome abundance makes them feasible biomarker molecules, this review discusses exosome heterogeneity and how exosomes orchestrate the interplay among tumor cells, cancer stem cells (CSCs) and host cells, including stromal cells, endothelial cells and immunocytes, in the CRC TME. This review also discusses mechanisms for loading exosomal contents and potential exosomal DNA, RNA and protein biomarkers for early CRC detection. Finally, we summarize the diagnostic and therapeutic exosomes in clinical trials. We envision that detecting and targeting cancer-specific exosomes could provide therapeutic advances in developing personalized cancer medicine.


Similar content being viewed by others
Abbreviations
- CAFs:
-
Cancer-associated fibroblasts
- CEA:
-
Carcinoembryonic antigen
- ceRNA:
-
Endogenous RNA
- circRNAs:
-
Circular RNAs
- CRC:
-
Colorectal cancer
- CRCSCs:
-
Colorectal cancer stem cells
- CSCs:
-
Cancer stem cells
- CTC:
-
Circulating tumor cells
- DCs:
-
Dendritic cells
- ECM:
-
Extracellular matrix
- EMT:
-
Epithelial-mesenchymal transition
- ESCRT:
-
Endosomal sorting complexes required for transport
- EVs:
-
Extracellular vesicles
- FOBT:
-
Fecal occult blood test
- HDI:
-
High developing index
- HLECs:
-
Human lymphatic endothelial cells
- HMEC-1:
-
Human microvascular endothelial cells
- HUVECs:
-
Human umbilical vein endothelial cells
- ILVs:
-
Intraluminal vesicles
- LPS:
-
Lipopolysaccharide
- MDSCs:
-
Myeloid-derived suppressor cells
- MIIC:
-
Major histocompatibility complex class II-enriched component
- MISEVs:
-
Minimal information for studies of extracellular vesicles
- MN:
-
Micronuclei
- mtDNA:
-
Mitochondria DNA
- MVBs:
-
Multivesicular bodies
- nSMase:
-
Neutral sphingomyelinase
- SCD:
-
Symmetric cell division
- SEER:
-
Surveillance, Epidemiology, and End Results
- SLNs:
-
Sentinel lymph nodes
- TGF-β1:
-
Transforming growth factor-β1
- TME:
-
Tumor microenvironment
- TRAF3:
-
TNF receptor-associated Factor 3
- Tregs:
-
Regulatory T cells
- VEGF:
-
Vascular endothelial growth factor
- WHO:
-
World Health Organization
References
World Health Organization, Global Health Estimates 2020: Deaths by Cause, Age, Sex, by Country and by Region, 2000–2019 (World Health Organization, Geneva, 2020)
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249. doi: https://doi.org/10.3322/caac.21660.
Surveillance E, and End Results (SEER) Program Populations (1969-2019) National Cancer Institute, DCCPS, Surveillance Research Program, released February 2021. (Cancer Stat Facts: Colorectal Cancer, Maryland, 2021)
L. Guittet, V. Bouvier, N. Mariotte, J.P. Vallee, D. Arsene, S. Boutreux et al., Comparison of a guaiac based and an immunochemical faecal occult blood test in screening for colorectal cancer in a general average risk population. Gut 56, 210–214 (2007). https://doi.org/10.1136/gut.2006.101428
D.J. Robertson, T.F. Imperiale, Stool testing for colorectal cancer screening. Gastroenterology 149, 1286–1293 (2015). https://doi.org/10.1053/j.gastro.2015.05.045
R.H. Fletcher, Carcinoembryonic antigen. Ann. Intern. Med. 104, 66–73 (1986). https://doi.org/10.7326/0003-4819-104-1-66
J.S. Moore, T.H. Aulet, Colorectal cancer screening. Surg. Clin. North Am. 97, 487–502 (2017). https://doi.org/10.1016/j.suc.2017.01.001
C. Harding, J. Heuser, P. Stahl, Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J. Cell. Biol. 97, 329–339 (1983). https://doi.org/10.1083/jcb.97.2.329
B.T. Pan, R.M. Johnstone, Fate of the transferrin receptor during maturation of sheep reticulocytes in vitro: selective externalization of the receptor. Cell 33, 967–978 (1983). https://doi.org/10.1016/0092-8674(83)90040-5
R.M. Johnstone, M. Adam, J.R. Hammond, L. Orr, C. Turbide, Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J. Biol. Chem. 262, 9412–9420 (1987)
M.P. Bebelman, M.J. Smit, D.M. Pegtel, S.R. Baglio, Biogenesis and function of extracellular vesicles in cancer. Pharmacol. Ther. 188, 1–11 (2018). https://doi.org/10.1016/j.pharmthera.2018.02.013
M. Catalano, L. O’Driscoll, Inhibiting extracellular vesicles formation and release: a review of EV inhibitors. J. Extracell. Vesicles 9, 1703244 (2020). https://doi.org/10.1080/20013078.2019.1703244
M. Colombo, C. Moita, G. van Niel, J. Kowal, J. Vigneron et al., Analysis of ESCRT functions in exosome biogenesis, composition and secretion highlights the heterogeneity of extracellular vesicles. J. Cell Sci. 126, 5553–5565 (2013). https://doi.org/10.1242/jcs.128868
T. Wollert, J.H. Hurley, Molecular mechanism of multivesicular body biogenesis by ESCRT complexes. Nature 464, 864–869 (2010). https://doi.org/10.1038/nature08849
R.L. Williams, S. Urbe, The emerging shape of the ESCRT machinery. Nat. Rev. Mol. Cell Biol. 8, 355–368 (2007). https://doi.org/10.1038/nrm2162
K. Trajkovic, C. Hsu, S. Chiantia, L. Rajendran, D. Wenzel et al., Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science 319, 1244–1247 (2008). https://doi.org/10.1126/science.1153124
M. Chivet, F. Hemming, K. Pernet-Gallay, S. Fraboulet, R. Sadoul, Emerging role of neuronal exosomes in the central nervous system. Front Physiol. 3, 145 (2012). https://doi.org/10.3389/fphys.2012.00145
G. Raposo, H.W. Nijman, W. Stoorvogel, R. Liejendekker, C.V. Harding et al., B lymphocytes secrete antigen-presenting vesicles. J. Exp. Med. 183, 1161–1172 (1996). https://doi.org/10.1084/jem.183.3.1161
L. Zitvogel, A. Regnault, A. Lozier, J. Wolfers, C. Flament et al., Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat. Med. 4, 594–600 (1998). https://doi.org/10.1038/nm0598-594
D. Koppers-Lalic, M. Hackenberg, I.V. Bijnsdorp, M.A.J. van Eijndhoven, P. Sadek et al., Nontemplated nucleotide additions distinguish the small RNA composition in cells from exosomes. Cell Rep. 8, 1649–1658 (2014). https://doi.org/10.1016/j.celrep.2014.08.027
M.F. Bolukbasi, A. Mizrak, G.B. Ozdener, S. Madlener, T. Strobel et al., miR-1289 and “zipcode”-like sequence enrich mRNAs in microvesicles. Mol. Ther. Nucleic Acids 1, e10 (2012). https://doi.org/10.1038/mtna.2011.2
E.N. Nolte-’t Hoen, H.P. Buermans, M. Waasdorp, W. Stoorvogel, M.H. Wauben et al., Deep sequencing of RNA from immune cell-derived vesicles uncovers the selective incorporation of small non-coding RNA biotypes with potential regulatory functions. Nucleic Acids Res. 40, 9272–9285 (2012). https://doi.org/10.1093/nar/gks658
A. Yokoi, A. Villar-Prados, P.A. Oliphint, J. Zhang, X. Song et al., Mechanisms of nuclear content loading to exosomes. Sci. Adv. 5, eaax8849 (2019). https://doi.org/10.1126/sciadv.aax8849
A. Takahashi, R. Okada, K. Nagao, Y. Kawamata, A. Hanyu et al., Exosomes maintain cellular homeostasis by excreting harmful DNA from cells. Nat. Commun. 8, 15287 (2017). https://doi.org/10.1038/ncomms15287
D. Torralba, F. Baixauli, C. Villarroya-Beltri, I. Fernandez-Delgado, A. Latorre-Pellicer et al., Priming of dendritic cells by DNA-containing extracellular vesicles from activated T cells through antigen-driven contacts. Nat. Commun. 9, 2658 (2018). https://doi.org/10.1038/s41467-018-05077-9
P. Sansone, C. Savini, I. Kurelac, Q. Chang, L.B. Amato et al., Packaging and transfer of mitochondrial DNA via exosomes regulate escape from dormancy in hormonal therapy-resistant breast cancer. Proc. Natl. Acad. Sci. U. S. A. 114, E9066–E9075 (2017). https://doi.org/10.1073/pnas.1704862114
J. Guinney, R. Dienstmann, X. Wang, A. de Reynies, A. Schlicker et al., The consensus molecular subtypes of colorectal cancer. Nat. Med. 21, 1350–1356 (2015). https://doi.org/10.1038/nm.3967
Z. Huang, M. Yang, Y. Li, F. Yang, Y. Feng, Exosomes derived from hypoxic colorectal cancer cells transfer Wnt4 to normoxic cells to elicit a prometastatic phenotype. Int. J. Biol. Sci. 14, 2094–2102 (2018). https://doi.org/10.7150/ijbs.28288
X. Hu, Y. Mu, J. Liu, X. Mu, F. Gao et al., Exosomes derived from hypoxic colorectal cancer cells transfer miR-410-3p to regulate tumor progression. J. Cancer 11, 4724–4735 (2020). https://doi.org/10.7150/jca.33232
J. Li, P. Yang, F. Chen, Y. Tan, C. Huang et al., Hypoxic colorectal cancer-derived extracellular vesicles deliver microRNA-361-3p to facilitate cell proliferation by targeting TRAF3 via the noncanonical NF-kappaB pathways. Clin. Transl. Med. 11, e349 (2021). https://doi.org/10.1002/ctm2.349
X. Zhang, J. Bai, H. Yin, L. Long, Z. Zheng et al., Exosomal miR-1255b-5p targets human telomerase reverse transcriptase in colorectal cancer cells to suppress epithelial-to-mesenchymal transition. Mol. Oncol. 14, 2589–2608 (2020). https://doi.org/10.1002/1878-0261.12765
H. Kalra, L. Gangoda, P. Fonseka, S.V. Chitti, M. Liem et al., Extracellular vesicles containing oncogenic mutant beta-catenin activate Wnt signalling pathway in the recipient cells. J. Extracell. Vesicles 8, 1690217 (2019). https://doi.org/10.1080/20013078.2019.1690217
R. Domenis, A. Cifu, C. Mio, M. Fabris, F. Curcio, Pro-inflammatory microenvironment modulates the transfer of mutated TP53 mediated by tumor exosomes. Int. J. Mol. Sci. 22(2021). https://doi.org/10.3390/ijms22126258
A. Shang, C. Gu, W. Wang, X. Wang, J. Sun et al., Exosomal circPACRGL promotes progression of colorectal cancer via the miR-142-3p/miR-506-3p- TGF-beta1 axis. Mol. Cancer 19, 117 (2020). https://doi.org/10.1186/s12943-020-01235-0
Z. Zeng, Y. Li, Y. Pan, X. Lan, F. Song et al., Cancer-derived exosomal miR-25-3p promotes pre-metastatic niche formation by inducing vascular permeability and angiogenesis. Nat. Commun. 9, 5395 (2018). https://doi.org/10.1038/s41467-018-07810-w
H.Y. Hu, C.H. Yu, H.H. Zhang, S.Z. Zhang, W.Y. Yu et al., Exosomal miR-1229 derived from colorectal cancer cells promotes angiogenesis by targeting HIPK2. Int. J. Biol. Macromol. 132, 470–477 (2019). https://doi.org/10.1016/j.ijbiomac.2019.03.221
Q. He, A. Ye, W. Ye, X. Liao, G. Qin et al., Cancer-secreted exosomal miR-21-5p induces angiogenesis and vascular permeability by targeting KRIT1. Cell Death Dis. 12, 576 (2021). https://doi.org/10.1038/s41419-021-03803-8
A. Shang, X. Wang, C. Gu, W. Liu, J. Sun et al., Exosomal miR-183-5p promotes angiogenesis in colorectal cancer by regulation of FOXO1. Aging (Albany NY) 12, 8352–8371 (2020). https://doi.org/10.18632/aging.103145
J. Zhi, X.J. Jia, J. Yan, H.C. Wang, B. Feng et al., BRAF(V600E) mutant colorectal cancer cells mediate local immunosuppressive microenvironment through exosomal long noncoding RNAs. World J. Gastrointest. Oncol. 13, 2129–2148 (2021). https://doi.org/10.4251/wjgo.v13.i12.2129
X. Zheng, J. Liu, X. Li, R. Tian, K. Shang et al., Angiogenesis is promoted by exosomal DPP4 derived from 5-fluorouracil-resistant colon cancer cells. Cancer Lett. 497, 190–201 (2021). https://doi.org/10.1016/j.canlet.2020.10.009
P.S. Steeg, Targeting metastasis. Nat. Rev. Cancer 16, 201–218 (2016). https://doi.org/10.1038/nrc.2016.25
T. Ning, J. Li, Y. He, H. Zhang, X. Wang et al., Exosomal miR-208b related with oxaliplatin resistance promotes Treg expansion in colorectal cancer. Mol. Ther. 29, 2723–2736 (2021). https://doi.org/10.1016/j.ymthe.2021.04.028
F. Chalmin, S. Ladoire, G. Mignot, J. Vincent, M. Bruchard et al., Membrane-associated Hsp72 from tumor-derived exosomes mediates STAT3-dependent immunosuppressive function of mouse and human myeloid-derived suppressor cells. J. Clin. Invest. 120, 457–471 (2010). https://doi.org/10.1172/JCI40483
B. Sun, Y. Zhou, Y. Fang, Z. Li, X. Gu, J. Xiang, Colorectal cancer exosomes induce lymphatic network remodeling in lymph nodes. Int. J. Cancer 145, 1648–1659 (2019). https://doi.org/10.1002/ijc.32196
S. Zhao, Y. Mi, B. Guan, B. Zheng, P. Wei et al., Tumor-derived exosomal miR-934 induces macrophage M2 polarization to promote liver metastasis of colorectal cancer. J. Hematol. Oncol. 13, 156 (2020). https://doi.org/10.1186/s13045-020-00991-2
J. Lan, L. Sun, F. Xu, L. Liu, F. Hu et al., M2 Macrophage-derived exosomes promote cell migration and invasion in colon cancer. Cancer Res. 79, 146–158 (2019). https://doi.org/10.1158/0008-5472.CAN-18-0014
T. Liu, L. Zhou, D. Li, T. Andl, Y. Zhang, Cancer-associated fibroblasts build and secure the tumor microenvironment. Front. Cell Dev. Biol. 7, 60 (2019). https://doi.org/10.3389/fcell.2019.00060
A. Rai, D.W. Greening, M. Chen, R. Xu, H. Ji et al., Exosomes derived from human primary and metastatic colorectal cancer cells contribute to functional heterogeneity of activated fibroblasts by reprogramming their proteome. Proteomics 19, e1800148 (2019). https://doi.org/10.1002/pmic.201800148
A. Rai, D.W. Greening, R. Xu, W. Suwakulsiri, R.J. Simpson, Exosomes derived from the human primary colorectal cancer cell line SW480 orchestrate fibroblast-led cancer invasion. Proteomics 20, e2000016 (2020). https://doi.org/10.1002/pmic.202000016
S.P. Clerici, M. Peppelenbosch, G. Fuhler, S.R. Consonni, C.V. Ferreira-Halder, Colorectal cancer cell-derived small extracellular vesicles educate human fibroblasts to stimulate migratory capacity. Front. Cell. Dev. Biol. 9, 696373 (2021). https://doi.org/10.3389/fcell.2021.696373
S. Yoshii, Y. Hayashi, H. Iijima, T. Inoue, K. Kimura et al., Exosomal microRNAs derived from colon cancer cells promote tumor progression by suppressing fibroblast TP53 expression. Cancer Sci. 110, 2396–2407 (2019). https://doi.org/10.1111/cas.14084
Y. Zhang, S. Wang, Q. Lai, Y. Fang, C. Wu, Y. Liu, Q. Li, X. Wang, C. Gu, J. Chen, J. Cai, A. Li, S. Liu, Cancer-associated fibroblasts-derived exosomal miR-17-5p promotes colorectal cancer aggressive phenotype by initiating a RUNX3/MYC/TGF-beta1 positive feedback loop. Cancer Lett. 491, 22–35 (2020). https://doi.org/10.1016/j.canlet.2020.07.023
X. Chen, J. Liu, Q. Zhang, B. Liu, Y. Cheng, Y. Zhang et al., Exosome-mediated transfer of miR-93-5p from cancer-associated fibroblasts confer radioresistance in colorectal cancer cells by downregulating FOXA1 and upregulating TGFB3. J. Exp. Clin. Cancer Res. 39, 65 (2020). https://doi.org/10.1186/s13046-019-1507-2
C. Gu, H. Lu, Z. Qian, Matrine reduces the secretion of exosomal circSLC7A6 from cancer-associated fibroblast to inhibit tumorigenesis of colorectal cancer by regulating CXCR5. Biochem. Biophys. Res. Commun. 527, 638–645 (2020). https://doi.org/10.1016/j.bbrc.2020.04.142
M.H. Frank, B.J. Wilson, J.S. Gold, N.Y. Frank, Clinical implications of colorectal cancer stem cells in the age of single-cell omics and targeted therapies. Gastroenterology 160, 1947–1960 (2021). https://doi.org/10.1053/j.gastro.2020.12.080
H. Zhao, S. Chen, Q. Fu, Exosomes from CD133(+) cells carrying circ-ABCC1 mediate cell stemness and metastasis in colorectal cancer. J. Cell. Biochem. 121, 3286–3297 (2020). https://doi.org/10.1002/jcb.29600
D. Kyuno, K. Zhao, M. Schnolzer, J. Provaznik, T. Hackert et al., Claudin7-dependent exosome-promoted reprogramming of nonmetastasizing tumor cells. Int. J. Cancer 145, 2182–2200 (2019). https://doi.org/10.1002/ijc.32312
W.L. Hwang, J.K. Jiang, S.H. Yang, T.S. Huang, H.Y. Lan et al., MicroRNA-146a directs the symmetric division of Snail-dominant colorectal cancer stem cells. Nat. Cell. Biol. 16, 268–280 (2014). https://doi.org/10.1038/ncb2910
W.C. Cheng, T.T. Liao, C.C. Lin, L.E. Yuan, H.Y. Lan et al., RAB27B-activated secretion of stem-like tumor exosomes delivers the biomarker microRNA-146a-5p, which promotes tumorigenesis and associates with an immunosuppressive tumor microenvironment in colorectal cancer. Int. J. Cancer. 145, 2209–2224 (2019). https://doi.org/10.1002/ijc.32338
W.L. Hwang, H.Y. Lan, W.C. Cheng, S.C. Huang, M.H. Yang, Tumor stem-like cell-derived exosomal RNAs prime neutrophils for facilitating tumorigenesis of colon cancer. J. Hematol. Oncol. 12, 10 (2019). https://doi.org/10.1186/s13045-019-0699-4
Y.J. Huang, T.H. Huang, V.K. Yadav, M.R. Sumitra, D.T. Tzeng et al., Preclinical investigation of ovatodiolide as a potential inhibitor of colon cancer stem cells via downregulating sphere-derived exosomal beta-catenin/STAT3/miR-1246 cargoes. Am. J. Cancer Res. 10, 2337–2354 (2020)
X. Deng, H. Ruan, X. Zhang, X. Xu, Y. Zhu et al., Long noncoding RNA CCAL transferred from fibroblasts by exosomes promotes chemoresistance of colorectal cancer cells. Int. J. Cancer 146, 1700–1716 (2020). https://doi.org/10.1002/ijc.32608
J. Ren, L. Ding, D. Zhang, G. Shi, Q. Xu et al., Carcinoma-associated fibroblasts promote the stemness and chemoresistance of colorectal cancer by transferring exosomal lncRNA H19. Theranostics 8, 3932–3948 (2018). https://doi.org/10.7150/thno.25541
J.L. Hu, W. Wang, X.L. Lan, Z.C. Zeng, Y.S. Liang et al., CAFs secreted exosomes promote metastasis and chemotherapy resistance by enhancing cell stemness and epithelial-mesenchymal transition in colorectal cancer. Mol Cancer 18, 91 (2019). https://doi.org/10.1186/s12943-019-1019-x
B. Zhou, K. Xu, X. Zheng, T. Chen, J. Wang et al., Application of exosomes as liquid biopsy in clinical diagnosis. Signal Transduct. Target Ther. 5, 144 (2020). https://doi.org/10.1038/s41392-020-00258-9
J. Silva, V. Garcia, M. Rodriguez, M. Compte, E. Cisneros et al., Analysis of exosome release and its prognostic value in human colorectal cancer. Genes Chromosomes Cancer 51, 409–418 (2012). https://doi.org/10.1002/gcc.21926
L. Dong, W. Lin, P. Qi, M.D. Xu, X. Wu et al., Circulating long RNAs in serum extracellular vesicles: their characterization and potential application as biomarkers for diagnosis of Colorectal Cancer. Cancer Epidemiol. Biomarkers Prev. 25, 1158–1166 (2016). https://doi.org/10.1158/1055-9965.EPI-16-0006
D. Hu, Y. Zhan, K. Zhu, M. Bai, J. Han et al., Plasma exosomal long non-coding RNAs serve as biomarkers for early detection of colorectal cancer. Cell Physiol. Biochem. 51, 2704–2715 (2018). https://doi.org/10.1159/000495961
B.K. Thakur, H. Zhang, A. Becker, I. Matei, Y. Huang et al., Double-stranded DNA in exosomes: a novel biomarker in cancer detection. Cell Res. 24, 766–769 (2014). https://doi.org/10.1038/cr.2014.44
D. Lucchetti, I.V. Zurlo, F. Colella, C. Ricciardi-Tenore, M. Di Salvatore et al., Mutational status of plasma exosomal KRAS predicts outcome in patients with metastatic colorectal cancer. Sci. Rep. 11, 22686 (2021). https://doi.org/10.1038/s41598-021-01668-7
F. Aqil, R. Munagala, J. Jeyabalan, A.K. Agrawal, R. Gupta, Exosomes for the enhanced tissue bioavailability and efficacy of curcumin. AAPS J. 19, 1691–1702 (2017). https://doi.org/10.1208/s12248-017-0154-9
M. Fujii, M. Shimokawa, S. Date, A. Takano, M. Matano et al., A colorectal tumor organoid library demonstrates progressive loss of niche factor requirements during tumorigenesis. Cell Stem Cell 18, 827–838 (2016). https://doi.org/10.1016/j.stem.2016.04.003
C. Thery, K.W. Witwer, E. Aikawa, M.J. Alcaraz, J.D. Anderson et al., Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 7, 1535750 (2018). https://doi.org/10.1080/20013078.2018.1535750
Acknowledgements
This work was supported by the Cancer Progression Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) and YEN TJING LAING MEDICAL FOUNDATION in Taiwan.
Funding
Ministry of Science and Technology (109–2320-B-010–021 and 111–2628-B-A49-017 to W-L.H., 109–2314-B-075–081-MY3 to H-W. T., 109–2314-B-075–026 to C–C.L.); a grant from the Yen Tjing Ling Medical Foundation (CI-111–15 to W-L.H.), a grant from the Ministry of Health and Welfare, Center of Excellence for Cancer Research (MOHW107-TDU-B-211–114019, 111 W31208 to W-L.H.) and a grant from the Higher Education Sprout Project by the Ministry of Education (MOE) (110AC-D303 to W-L.H.). Taipei Veterans General Hospital (V111C-069 and V110C-189 to H-W.T.). The Innovative Research Grant of the National Health Research Institutes of Taiwan (NHRI-EX110-11010BI to W–H.Y.) and Ying Tsai Young Scholar Award (CMU108-YTY-04 to W–H.Y.).
Author information
Authors and Affiliations
Contributions
W–C.L., C–C.L. Y-Y.L., H-W.T. and W-L.H. wrote and revised the manuscript with assistance from W–H.Y. and Y-C.T.
Corresponding authors
Ethics declarations
Ethical approval and consent to participate
Not applicable.
Consent for publication
All authors have agreed to publish this manuscript.
Availability of supporting data
Not applicable.
Competing interests
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, WC., Lin, CC., Lin, YY. et al. Molecular actions of exosomes and their theragnostics in colorectal cancer: current findings and limitations. Cell Oncol. 45, 1043–1052 (2022). https://doi.org/10.1007/s13402-022-00711-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13402-022-00711-7