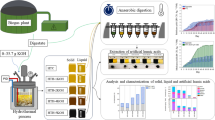
Abstract
The production of high-value products from microalgae, one of the preferred emerging biorefineries’ feedstocks, relies on the crucial step of biomass fractionation. In this work, the fractionation of Chlorella vulgaris and Scenedesmus obliquus biomass was tested for protein extraction using a wide range of physical, chemical, and enzymatic treatment combinations, including ultrasound, cell homogenizer, cellulase, and alcalase combinations in aqueous and alkali extraction conditions. The impact of these processes on biomass carbohydrates was also evaluated. Alkaline-assisted ultrasound treatments using alcalase presented the highest protein extraction yield, reaching 90 g/100 g protein on C. vulgaris, closely followed by the same treatment in aqueous conditions (85 g/100 g protein). The same aqueous treatment achieved the best performance on S. obliquus, reaching 82 g/100 g protein. All treatments on both microalgae partially solubilized the polysaccharide fraction with all alkaline treatments solubilizing over 50 g/100 g sugars for all conditions. Overall, all the treatments applied were effective methods for biomass fractionation, although they showed low selectivity regarding the individual extraction of protein or carbohydrates.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
1 Introduction
Microalgae are one of the most innovative and promising biomass sources that have been suggested for biorefinery applications, namely for the production of biofuels, e.g., biodiesel from microalgae oil [1]. However, microalgae potential is much wider, as they have also been reported as a viable source of bioproducts [2], namely of pigments [3,4,5], protein [6, 7], amino acids [6], and fatty acids [8] for food and feed applications. The microalgae sector has gained increasing relevance in the EU, as part of the European Green Deal that includes the use of microalgae as an essential facet of the Blue Bioeconomy in the European space [9].
Microalgae have a large range of potential high-value commercial applications, in sectors such as bioplastics and biomaterials [10] with nutraceutical applications, due to their high nutritional value [11]. Applications for the production of advanced biofuels have also gained increased interest, thereby granting microalgae an even more important role in climate change mitigation [12]. The first approaches to use microalgae in biorefineries were based on solvent extraction for isolation of their lipid fraction and subsequent conversion of bio-oil to biodiesel, while a protein and sugar-rich solid residue was left with little use. By itself, biofuel production from microalgae is not economically viable [13], and needs to be coupled with the production of other high-value products for the economic feasibility of the approach. Therefore, the most recent strategy is to target microalgae biomass towards the production of added-value products such as platform chemicals, biomaterials, and food and feed products, which may also be connected to biofuel production.
Microalgae have an incredible ability to accumulate different types of macromolecules, depending on their growth conditions, thereby presenting a versatility in composition that is virtually unparalleled elsewhere in the biosphere. Due to this versatility, growth conditions are very important as, for example, in nutrient-depleted microalgae, the protein content can drop significantly [14,15,16]. But this compositional diversity that grants microalgae a huge potential for the production of many bioproducts is not the only interesting aspect since they also have great wastewater treatment capabilities [17, 18]. Microalgae can be excellent bioremediators with high effectiveness in removing pollutants and heavy metals from aqueous biomes [19]. Also, as the cultivation of microalgae can be a very costly process, the use of low-cost culture media like wastewater can make microalgae more economically sustainable [20].
Microalgae are adequate to be used in feed, and chemical industries. When compared to the lignocellulosic biomasses commonly used in biorefineries, microalgae do not contain lignin, which is a technical advantage in the fractionation and handling of downstream processes. Also, they are an alternative protein source for food and feed as they can contain all essential amino acids [7, 21]. This amino acid profile is rarely found in terrestrial plants and can therefore become an integral part of non-meat diets that are increasingly popular in the Western world, providing a greener alternative to meat production, which is an important source of carbon emissions [13].
However, microalgae valorisation requires fractionation techniques to separate the most important components, mainly protein, and sugars, and the challenge is to find methodologies that are cheap and can provide selective fractionation. Simple, fast, and economical fractionation methods are required for microalgae that allow for the release and easy recovery of most protein [22], facilitate downstream processing [23], and also enable the separation and recovery of the sugar fraction.
In contrast to other molecules, such as starch and lipids that can be found in cell storage organelles, protein is present in almost every part of the cell and therefore can be easily removed by disrupting the cells and extraction with solvents. Microalgae commonly have rigid cell walls consisting of polysaccharides, mainly cellulose [24], and other polysaccharides containing xylose, mannose, and uronic acids, or trilaminar layers with pectin and glycoproteins in the middle layer. Algeanan is also a biopolymer that forms an outer layer in several microalgae species, conferring protection against degradation, hydrolysis, and mechanical disruption of the cells [25] which will make protein extraction more challenging. This compound is present in some Chlorophyta species such as Scenedesmus and Chlorella [25] that were studied in this work. In addition to algeanan, some species of the Scenedesmus genus also present a crystalline glycoprotein in the cell wall, which can further affect cell disruption and product recovery, and increase the difficulty in protein extraction. However, some proteins can be found freely in the cytosol like ribosomes, free enzymes, and other protein complexes, and are easily accessible through cell disruption. Although some advances have been made in the past decade, the study of algal proteins and their extraction remain a poorly researched topic, when compared to proteins from terrestrial crops [26]. Protein extraction can be a challenging endeavor, due to the complexity of the cell walls, which are in many cases interlinked with protein [27]. Therefore, protein extraction is mostly achieved in two steps, cell disruption and protein recovery [28]. This extraction may be enhanced by the use of some pre-treatments that cause cell wall degradation, and improve accessibility [29]; i.e., cell disruption techniques can be used for the extraction of intracellular components [7, 30, 31]. Many processes have been tested such as cell homogenization [32], ultrasonication [33, 34], microwave radiation [35], high-pressure cell disruption [36], and enzymatic lysis kits [37]. Some of these methods, such as high-pressure homogenization and enzymatic treatment, have been reported to achieve extraction yields of around 80–90% [38], although their commercial application for algal protein extraction remains limited, due to issues with up-scaling and operational costs [26]. Furthermore, the effectiveness of these techniques is highly dependent on the algae species, and therefore individual studies are vital to assess their application suitability for each case [26].
Although effective for protein extraction, the impact of these technologies on the sugar fraction is usually not mentioned, and it is still largely unknown, despite its importance. Also, studies focusing on carbohydrate fractionation are few and only achieve sugar recovery rates above 50% when using very high concentrations of acid catalysts and severe and potentially uneconomic conditions [39].
In this work, a new approach to microalgae valorisation was studied to attempt an effective protein and carbohydrate separation. For this purpose, multiple physical, chemical, and enzymatic methods were tested, both per se and in combination, including ultrasonication, cell homogenization, alkaline treatments, and cellulase and alcalase treatments for protein extraction from commercial Chlorella vulgaris. The most effective methods were selected and further validated on the microalgae Scenedesmus obliquus that was previously grown on secondary wastewater brewery’s effluent. The impact of protein extraction methods on the carbohydrate fraction in both algae was also evaluated, thereby providing a first insight into the effect of these pre-treatments in the sugar extraction from algae.
2 Materials and methods
2.1 Microalgae cultivation and harvesting
Chlorella vulgaris biomass was obtained from Allmicroalgae (Pataias, Portugal), in freeze-dried form. Scenedesmus obliquus (ACOI 204/07, Coimbra University Culture Collection, Portugal), obtained from the LNEG culture bank, was cultivated in 70-L vertical acrylic column photobioreactors. This microalga was grown in brewery secondary effluent (Table 1) provided by Central de Cervejas, SA (Vialonga, Portugal) in batch mode, as described [39]. Microalgae were harvested in the exponential growth phase and concentrated using an in-house developed centrifugation methodology with a continuous dairy centrifuge (electric cream separator, Alfa Laval, Sweden) [39]. The centrifuged biomass was washed with distilled water and centrifuged again to obtain a concentrated paste with 90% moisture content. This paste was frozen (−18°C) and then freeze-dried for further use (Thermo Scientific Heto PowerDry LL3000, USA).
2.2 Pre-treatment processes
All pre-treatments and their combinations were carried out in duplicate.
2.2.1 Aqueous extraction
For aqueous protein extraction, 0.5 g of freeze-dried microalgae (C. vulgaris and S. obliquus) were weighted to 50-mL closed flasks and suspended in 20 mL of distilled water (physical extraction) or buffer (enzymatic extraction) and incubated in an orbital shaker (Comecta; Spain) at 30 °C, 250 rpm for 2 h. After incubation, the obtained suspension was subjected to further treatments of ultrasonication or cell homogenization, as listed below.
2.2.2 Ultrasonication
Sample ultrasonication was carried out using an ultrasound probe (Sonics vibracell, USA) with a treatment time of 2 min in cycles of 5-s pulse and 15-s resting time. These conditions were chosen after an optimization of ultrasonication conditions determined by chlorophyll extraction yield. During the treatment, an ice bath was used to prevent sample overheating and degradation that can normally occur in this process [37].
2.2.3 Cell homogenization
Cell homogenization was carried out using a Rotor Stator disperser homogenizer (Turrax-type homogenizer, Atomixmill, Germany) for 30 s.
2.2.4 Enzymatic treatment
Sample preparation for enzymatic treatment was similar to the aqueous process, except that water was replaced by an adequate buffer. For samples treated with cellulase mixture (i), sample hydrolysis was carried out using 0.1M citrate buffer (pH 5.0). A Cellic® CTec2 (Novozymes, Denmark) enzyme cocktail (199 FPU/ml) was used in a ratio of 0.5 mg/g biomass, and samples were incubated at 50°C and 250 rpm for 16 h.
For samples tested with alcalase (ii), the treatment was carried out using 0.1 M phosphate buffer (pH 8.0). Alcalase 2.4L (Sigma-Aldrich, Germany) was used in a ratio of 0.5 mg/g biomass, and samples were incubated at 60°C and 150 rpm for 16 h. These enzymatic processes were tested both before (for C. vulgaris) and after physical treatment (for C. vulgaris and S. obliquus).
2.2.5 Alkaline pre-treatment
The samples that were treated as above described were subjected to chemical protein extraction. For this, sodium hydroxide (2 M) was added until pH 12 was reached. Samples were then incubated in similar conditions as described in Section 2.2.1, except that temperature was 40°C.
2.2.6 Pre-treated biomass separation and protein precipitation
Pre-treated samples were centrifuged (Ortoalresa digicen 21 R, Spain) at 5300 g, and 15°C for 10 min to separate the pre-treated biomass biomass pellet from the supernatant. The supernatant was treated with an HCl 0.1 M solution to decrease pH to 2.5. The protein precipitate obtained was separated by centrifugation, in the same conditions as described above. The biomass pellets and protein precipitates were freeze-dried for further analysis.
2.3 Analytical methods
Throughout the work, for all experiments, deviations were typically lower than 5%.
2.3.1 Quantification of extractives
Extractives content of microalgae (C. vulgaris and S. obliquus) was determined by sequential solvent extraction in Soxhlet using dichloromethane, ethanol, and water, using a modified method based on TAPPI-T204 cm-97 [40] and NREL/TP-510-42619 protocols for lignocellulosic biomass [41]. For this determination, 2 g of sample was weighted into extraction cartridges, inserted into a 125-mL Soxhlet apparatus, and extracted with 190 mL of solvent. The extraction times were 6 h for dichloromethane and 18 h for both ethanol and water. Extractives solubilized by each solvent were calculated by solvent evaporation in relation to the total dry mass.
2.3.2 Moisture and ash content
The moisture content was determined by oven-drying at 105°C to constant weight and the ash content was determined by incineration at 550°C for 16 h using NREL/TP-510-42622 protocol [42].
2.3.3 Protein quantification
Nitrogen content of microalgae biomass and of both supernatant and pellet obtained after protein extraction was determined using the Kjeldahl method [43] and a semiautomatic protein analyzer (Tecator, Sweden). Protein content was calculated using a conversion factor of N × 4.78, as indicated for algae [44].
2.3.4 Carbohydrate analysis
Extracted and non-extracted biomass samples were subjected to quantitative acid hydrolysis using 72% (w/w) H2SO4 followed by hydrolysis with 4% (w/w) H2SO4 at 121°C in an autoclave for 1 h, according to NREL protocol for algae [45]. The acid-insoluble residue was determined by filtration using 1.22 μm glass fiber filters (VWR, USA), after correction for ash (incineration at 550°C for 16 h).
The monosaccharides in the hydrolysis liquor (glucose, mannose, xylose, galactose, and arabinose) were analyzed in an HPLC system (Agilent 1100 Series, Waldbronn, Germany), equipped with a refractive index (RI) detector and a diode array detector (DAD). An Aminex HPX-87P column (Bio-Rad, Hercules, USA) in combination with a cation Pb2+-guard column (Bio-Rad) was used. The column temperature was 80°C, and water was used as eluent at a flow rate of 0.6 mL/min [46]. Samples were previously neutralized, when needed, using barium hydroxide or a combination of Amberlite® MB-20 resin (Sigma-Aldrich, USA) and calcium carbonate [39]. All samples were filtered through 0.22 nylon membrane filters (VWR, USA) before HPLC analysis. The percentage of polymeric sugars was calculated according to the methodology described in [39].
Sugars’ solubilization was calculated based on the composition of the residual solid obtained after the protein extraction treatments, according to the equation:
where SI corresponds to the biomass initial sugars, Ssol to the sugars in treated solids, and SY to the solid yield obtained with the treatment.
3 Results and discussion
3.1 Biomass composition
Two microalgae were used to evaluate the effects of different treatments on the protein and carbohydrate fractions of the biomass. Chlorella vulgaris was used as benchmarking due to its commercial relevance and availability, and for being a widely studied species. Therefore, it was used as a model in a more extensive set of test conditions, allowing screening to provide optimization to be further applied to Scenedesmus obliquus.
Table 2 shows the chemical composition of the microalgae used in this study. The protein content found on C. vulgaris is high, similar to those previously reported for this species [34, 47, 48], and in agreement with the commercial analysis certificate (provided by Allmicroalgae). The protein content found is only slightly lower than that of other species like the marine microalgae Dunaliella salina (57%) and the cyanobacteria Spirulina platensis (55.8%) [38, 48].
The high protein content of C. vulgaris suggests that this species is a highly viable source of this nutrient that can be potentially fractionated and further upgraded. Therefore, it is a suitable biomass for an extended battery of protein extraction tests.
S. obliquus protein content was 25.4%, a lower value than that previously reported for this [38] and other microalgal genera, e.g., Chlorella, Dunalliella, and Porphyridium [14, 21, 34, 48]. This depends on the microalgae species but can also be attributed to the low nitrogen content of the culture medium [49]. In contrast to C. vulgaris, which has a quite low sugar content (6.9%), S. obliquus contains a much higher sugar content (16.0%), making it an interesting source of both protein and sugars.
The lipid fraction as given by the dichloromethane extractives [50, 51] was 2.8% and 5.3% for C. vulgaris and S. obliquus, respectively. For protein production and to facilitate downstream purification, this lipid content could be lowered with a small nitrogen supplementation, since nitrogen-limited environments boosted the production and accumulation of lipids in other microalgal species [15].
Although ash content is not often reported for microalgae, the values found for both biomasses were slightly higher than the desired values. However, the ash content of S. obliquus was obtained after washing with distilled water during the harvest process which led to a 10% reduction in ash content. This value could be further lowered by using additional washing steps or by the use of acid buffer solutions (around pH 4), such as citrate buffer.
3.2 Protein extraction
Different fractionation methods mainly targeted for protein extraction including ultrasound, homogenization (dispersion), alkali, and enzymatic (cellulase and alcalase) treatments were used to break cell walls and release protein. Some combinations of these treatments were also tested to evaluate their effect on protein extraction.
Figure 1 shows the effect of the extraction process or combination of processes on the protein extraction yield from C. vulgaris, under aqueous conditions only (panel A), and in aqueous conditions followed by alkali treatment (panel B). Data are shown as the percentage of protein in relation to the initial biomass protein obtained in the precipitate and in the corresponding liquid fraction.
Overall, alkaline-assisted treatments had a better performance on protein extraction, as protein recovery yield can be enhanced in alkaline conditions [47].
In aqueous conditions (panel A), the treatments were compared with an aqueous control. It has been reported that in microalgae, a significant fraction of the protein can be found free inside the cell walls [14]. Moreover, it has been documented that water can be an excellent solvent for protein extraction when compared with other solvents such as ethanol and methanol [23, 52], which can cause protein denaturation [53], and facilitate its downstream processing. Therefore, the aqueous treatment only (control) can be a good benchmark for comparing with other treatments. It amounted to 34%, slightly higher than the water-extractable fraction determined in the chemical characterization (Table 2). The extraction yield above this value suggests the impact of each treatment on cell disruption and in the desegregation of protein from the cell structure and other macromolecules, making the protein available for recovery.
This protein out-diffusion from the cells by aqueous extraction was higher than that previously reported for this microalga [34]. This difference could be attributed to cell fragility derived from the manufacturing process, since the sample was from an industrial source, and commonly employed processes like freeze-drying have been reported to have some positive effect on protein extraction [54].
Overall, when comparing the aqueous treatments, only 5 of the assays had yields close to the control, with a maximum increase of 15%. All the others amounted to yield increases ranging from 34 to 52%. Regarding the physical non-combined treatments, the turrax-type homogenizer yielded the lowest protein recovery, only 10% higher than the aqueous control. This physical method is based on the rotor-stator principle and is commonly used in disrupting microalgae for lipid extraction [55] or carotenoids [56]. In contrast, the application of ultrasound treatment resulted in one of the highest aqueous protein recoveries, with 71% of total protein. This process does not rely on mechanical aspects but on the application of ultrasonic waves that causes cavitation of the cell walls. As such, it can easily affect a wider number of cells and effectively disrupt cell walls and cell membranes, causing the release of intracellular components [57]. This method has been reported as effective for cell disruption and protein recovery [58].
Enzymatic treatments are highly dependent on the composition and complexity of the cell wall [59]. Therefore, two different enzyme cocktails were tested, with cellulase affecting cell integrity by breaking down cellulose, and alcalase disrupting by targeting the cell wall proteins. Compared to cellulase, alcalase was much more effective, extracting 80% of the initial protein, mostly in the non-acid precipitable form. Furthermore, due to the proteolytic capabilities of alcalase, these will most likely be in peptide form [59]. These peptides can have interesting high-value applications as protein supplements and in nutraceutical applications [59], making this process not only an effective method for protein extraction but also for simultaneous protein processing. On the other hand, if the functional properties of the protein are important for its further application, this method is not adequate [59]. The use of cellulase resulted in only 15% higher extraction than the aqueous control. This can be explained by the presence of carbohydrate polymers like algeanan, which provide cell wall stability and protection [25], making them more resilient against non-specific enzymes [60].
Several combinations of these treatments were also tested. In cellulase-assisted treatments, the protein yields obtained were quite similar compared to their non-enzymatic counterparts. In alcalase-assisted treatments, the enzyme impact was much higher, leading to an increase of 11–15% in ultrasound treatments and 27–35% in homogenizer treatments. It has been suggested that combining ultrasound with other treatments makes a strong boost in protein extraction for some microalgal species [61]. However, the boosting effect in ultrasound-based treatments was low, suggesting that adding enzymes to the ultrasound treatment might not be cost-effective. This is even more pronounced when comparing the ultrasound combined with the pure alcalase treatment, where the yield increase was just 2–6%.
The application of the enzyme cocktails either before or after the physical treatment was also tested. It only had a slight boosting effect (5%) when the enzyme was applied before the physical treatment. Overall, the highest recovery yield was 86 g/100 g total protein when using alcalase followed by ultrasound.
After all the treatments under aqueous conditions, alkaline conditions were also tested (Fig. 1(B)). By itself, the alkaline extraction resulted in a 24% boosting effect, when compared to the aqueous control, with a protein extraction of 58.5% of the total protein. This was higher than previously reported for this microalga [34]. In the case of alkali treatments, six were similar to the alkali control, with a maximum yield increase of 18%. All the other conditions yielded a 23 to 35% higher protein increase. Although the alkaline treatment only led to better results than the aqueous treatments using only homogenizer and cellulase, it still did not outperform the use of alcalase, which was suggested as a cheaper method [62].
When the alkali process is applied after other treatments, the protein yield was increased in all of the conditions, between 12 and 30%, and more notorious for the least effective aqueous treatments (cellulase and homogenizer). This boosting effect was milder in the other treatments. Overall, the application of alkaline extraction decreased the difference between the extraction yields obtained (59% in the lowest and 92% in the highest), compared with the yields verified in the aqueous extractions (between 34% and 86%). Overall, the highest protein recovery was obtained for the ultrasound treatments using alcalase, followed by an alkali treatment, by which a recovery of 93 g/100 g total protein was obtained. These results are in agreement with previous findings stating that this chemical treatment is effective for protein recovery [34, 54].
Most of the protein recovered was obtained in solubilized form. The highest yield of protein recovered as precipitate was 33.3% of the total protein obtained for the aqueous ultrasound treatment, higher than the 29% obtained with its alkali counterpart. The applied acid precipitation method was selected as it is a cheap and simple method for protein recovery [20], although it was not very effective in this case.
A better option for protein recovery could be membrane separation avoiding the precipitation step, which would increase the protein extract purity [63]. Nevertheless, acid precipitation has several advantages as compared to other methods such as salts or ethanol, as these could bring difficulties in the downstream process or contamination by undesired compounds in the precipitate [64]. The purity of the precipitates was also determined (% of protein in the total precipitate) and found to be up to 60% for most conditions. On average, the purities were 10% higher on aqueous treatments than on their alkali counterparts. These values indicate protein fragmentation into small peptides, inhibiting their precipitation by acid. Also, the isoelectric point of the protein from this particular case can differ from the pH 2.5 applied, which has been suggested for similar species [65].
The most effective treatments for protein extraction from C. vulgaris were then applied to S. obliquus. These included both enzymatic and ultrasound treatments. The results obtained are shown in Fig. 2. As alkaline treatments in C. vulgaris had a masking effect on the impact of the physical treatments when used in combinations, only ultrasound was tested for comparison. The control aqueous extraction yielded 11% of total protein extraction, a lower value than that obtained for C. vulgaris. This might indicate a higher resistance of this microalga, not reaching the 32% water-extractable protein found in the chemical characterization (Table 2). This could be attributed to the duration of the treatment (2 h) when compared to the 16 h of water extraction for chemical characterization. Alkaline extraction yielded an increase of 30% in relation to the aqueous counterpart, higher than for C. vulgaris but still only reaching 42% of total protein recovery. Ultrasound treatment provided a yield of 38.6 g/100 g total protein, with the precipitated fraction representing 58% of the total extracted protein, the highest value found for all treatments. However, this result was nearly half the reported in other studies [37], for protein-richer Scenedesmus strains. The alkaline version of this treatment provided a very mild yield increase of 7%, but the precipitated fraction dropped significantly to 22.6%, indicating a severe impact of the alkaline conditions on the structure of protein and its acid precipitation capabilities.
When using enzymes, the combination of ultrasound treatment and cellulase is virtually ineffective, providing a similar extraction yield as the ultrasound treatment only. This does not happen for alcalase, which is highly effective both by itself and in combination with ultrasound, reaching yields of 78% and 82%, respectively. These results suggest that contrary to C. vulgaris, S. obliquus is more resistant to physical treatments, but it is highly vulnerable to proteolytic enzymes which can be a powerful tool in the fractionation of this biomass.
3.3 Carbohydrate extraction
The effect of protein extraction processes on carbohydrate fraction is generally neglected in studies dealing with protein extraction from algal biomass [29, 34]. In this work, the effect of the different protein extraction processes on sugar solubilization was also evaluated. Figure 3 shows the data obtained regarding the potential total sugar extraction and the corresponding monomeric sugar composition is given in Figs. 4 and 5. Aqueous control resulted in a sugar extraction of 45% of the total algae carbohydrates, 12% more than the 33% total water-soluble sugars measured by Soxhlet extraction, in the biomass characterization (Table 2). In general, aqueous treatments had a lower effect on sugar solubilization than treatments followed by alkaline conditions. These showed a very pronounced effect, with a minimum sugar solubilization of 59.8% for alkali treatment only, and a maximum of 91.4% for the ultrasound and alcalase treatment with the same alkaline step. This was expected as alkaline treatments are known to have an important impact on hemicellulose solubilization in lignocellulosic materials [66].
The sugar yields obtained for alkali and aqueous extractions exceed both hydrothermal and alkaline pre-treatment results previously reported for this microalgae species [67]. Also, the sugar solubilization obtained in these mild conditions was similar [68] or much higher [69] than those reported for dilute acid hydrolysis of macroalgae in much more severe conditions. In microalgae, carbohydrates can be found mainly in starch grains, as glycolipids in intracellular membranes, and in cell walls as part of the structural matrix [70]. As both species had very low starch content (Table 2), most of the solubilized carbohydrates must derive from cell wall structural components. Therefore, it shows that treatments like ultrasound, rarely used in more conventional lignocellulosic biomasses [71], can be extremely effective for sugar solubilization in microalgae, opening up the possibility for alternative treatments to provide an innovative valorization of microalgae sugars.
Treatments using cellulase did not significantly increase sugar solubilization. This is likely due to the great variability of polysaccharides that compose the microalgae cell walls [72] and the other long-chain protective polymers [60] that may not be susceptible to enzymes that specifically target the glycosidic linkages in cellulose.
Figures 4 and 5 show the impact of the tested treatments on individual sugar removal. Glucose removal was above 40% of the total glucose in all treatments, both alkali and aqueous, although alkali treatments showed a higher impact on other sugars. Mannose solubilization could reach 100% removal in the majority of treatments that were followed by the alkaline step. The same solubilization boosting effect was verified for galactose, but to a lesser extent. Arabinose solubilization was also higher in alkali treatments.
Figure 6 illustrates the effects of the tested treatments on the sugar fraction of S. obliquus. All treatments strongly impacted the sugar fraction of S. obliquus, with the lowest solubilization of 56.6% obtained for the aqueous control. This contrasts with previous reports where the recovered carbohydrates were significantly less [67]. Solubilization exceeded 75% for all the other treatments and achieved higher or similar recovery yields for sugar and protein. As verified on C. vulgaris, these types of processes showed low selectivity for extraction of S. obliquus protein. The impact of the extraction methods on the sugar fraction is more significant for S. obliquus since it has a higher amount of sugar than C. vulgaris. Regarding the individual sugars (Fig. 7), the results obtained were quite similar to those found for C. vulgaris, with mannose being also the most affected sugar.
Overall, all the treatments tested for protein extraction also had an important impact on the sugar fraction, making these treatments a viable tool for further carbohydrate valorization. Their low selectivity towards the removal of only the protein fraction of the microalgae is a limitation that can be intrinsic to algae biomass. However, they can be a cheap, mild, and effective alternative for biomass fractionation since they require low temperatures (between 30°C and 60°C), and can prevent the protein and sugar degradation that can occur in other hydrothermal pre-treatments at higher temperatures [73]. Therefore, these processes can be effective for microalgae biomass fractionation if an efficient process for protein recovery, e.g., membrane separation, can be implemented.
4 Conclusions
Overall, all the treatments applied to C. vulgaris and S. obliquus were effective for the extraction of both protein and carbohydrates. Aqueous ultrasound treatment seems to be the most effective among the physical methods tested. Furthermore, all treatments resulted in higher extraction yields than those of the control, indicating their effectiveness in disrupting cell wall integrity and enhancing protein and sugar availability for solubilization and extraction.
Regarding the enzymatic treatments, only alcalase produced high protein extraction yields while also resulting in high sugar solubilizations, indicating that it can be viable, although non-selective alternative to physical methods. The aqueous ultrasound treatment was one of the best for protein extraction of both microalgae, with a lower impact on the carbohydrates, which prevents some potential downstream issues when compared to alkaline treatments, also being a simpler and cheaper alternative to enzymatic treatments.
These types of processes may be applied to microalgae for the production of low-purification supplements of protein or carbohydrates that can be used in food and feed applications, or as a complete growth medium or supplement for fermentation purposes for further valorization. Furthermore, the residual solid fractions obtained after extractions still present a relevant potential for further applications, e.g., anaerobic co-digestion to produce biogas or as fertilizer.
Data availability
The authors authorize the publication of the data and results shown in the paper.
References
Zhang S, Zhang L, Xu G, Li F, Li X (2022) A review on biodiesel production from microalgae: influencing parameters and recent advanced technologies. Front Microbiol 13:970028. https://doi.org/10.3389/fmicb.2022.970028
Dolganyuk V, Belova D, Babich O, Prosekov A, Ivanova S, Katserov D, Patyukov N, Sukhikh S (2020) Microalgae: a promising source of valuable bioproducts. Biomolecules 10(8). https://doi.org/10.3390/biom10081153
Sirotiya V, Ahirwar A, Mourya M, Khan MJ, Rai A, Kwatra R, Sharma AK, Harish Schoefs B, Marchand J et al (2022) Astaxanthin bioaccumulation in microalgae under environmental stress simulated in industrial effluents highlighting prospects of Haematococcus pluvialis: knowledge gaps and prospective approaches. Phytochem Rev 22(4):1041–1066. https://doi.org/10.1007/s11101-022-09807-2
Nair A, Ahirwar A, Singh S, Lodhi R, Lodhi A, Rai A, Jadhav DA, Harish Varjani S, Singh G et al (2023) Astaxanthin as a king of ketocarotenoids: structure, synthesis, accumulation, bioavailability and antioxidant properties. Mar Drugs 21(3). https://doi.org/10.3390/md21030176
Sarkar S, Manna MS, Bhowmick TK, Gayen K (2020) Extraction of chlorophylls and carotenoids from dry and wet biomass of isolated Chlorella Thermophila: optimization of process parameters and modelling by artificial neural network. Process Biochem 96:58–72. https://doi.org/10.1016/j.procbio.2020.05.025
Wang Y, Tibbetts SM, McGinn PJ (2021) Microalgae as sources of high-quality protein for human food and protein supplements. Foods 10(12). https://doi.org/10.3390/foods10123002
Geada P, Moreira C, Silva M, Nunes R, Madureira L, Rocha CMR, Pereira RN, Vicente AA, Teixeira JA (2021) Algal proteins: production strategies and nutritional and functional properties. Bioresour Technol 332:125125. https://doi.org/10.1016/j.biortech.2021.125125
Ramesh Kumar B, Deviram G, Mathimani T, Duc PA, Pugazhendhi A (2019) Microalgae as rich source of polyunsaturated fatty acids. Biocatal Agric Biotechnol 17:583–588. https://doi.org/10.1016/j.bcab.2019.01.017
Towards a Strong and Sustainable EU Algae Sector (2022) Opinion of the European Economic and Social Committee on the communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee, and the Committee of the Regions, COM/2022/592, Official Journal of the European Union, C 228/126
Onen Cinar S, Chong ZK, Kucuker MA, Wieczorek N, Cengiz U, Kuchta K (2020) Bioplastic production from microalgae: a review. Int J Environ Res Public Health 17(11). https://doi.org/10.3390/ijerph17113842
Koyande AK, Chew KW, Rambabu K, Tao Y, Chu DT, Show PL (2019) Microalgae: a potential alternative to health supplementation for humans. Food Sci Hum Wellness 8(1):16–24. https://doi.org/10.1016/j.fshw.2019.03.001
Onyeaka H, Miri T, Obileke K, Hart A, Anumudu C, Al-Sharify ZT (2021) Minimizing carbon footprint via microalgae as a biological capture. Carbon Capture Sci Technol 1:100007. https://doi.org/10.1016/j.ccst.2021.100007
Kuech A, Breuer M, Popescu I (2023) Research for PECH Committee – The future of the EU algae sector, European Parliament, Policy Department for Structural and Cohesion Policies, Brussels. https://bit.ly/3ZszAt2
Foley PM, Beach ES, Zimmerman JB (2011) Algae as a source of renewable chemicals: opportunities and challenges. Green Chem 13(6):1399. https://doi.org/10.1039/c1gc00015b
Yang L, Chen J, Qin S, Zeng M, Jiang Y, Hu L, Xiao P, Hao W, Hu Z, Lei A et al (2018) Growth and lipid accumulation by different nutrients in the microalga Chlamydomonas reinhardtii. Biotechnol Biofuels 11:40. https://doi.org/10.1186/s13068-018-1041-z
Mourya M, Khan MJ, Ahirwar A, Schoefs B, Marchand J, Rai A, Varjani S, Rajendran K, Banu JR, Vinayak V (2022) Latest trends and developments in microalgae as potential source for biofuels: the case of diatoms. Fuel 314:122738. https://doi.org/10.1016/j.fuel.2021.122738
Srimongkol P, Sangtanoo P, Songserm P, Watsuntorn W, Karnchanatat A (2022) Microalgae-based wastewater treatment for developing economic and environmental sustainability: current status and future prospects. Front Bioeng Biotechnol 10:904046. https://doi.org/10.3389/fbioe.2022.904046
Kashyap M, Chakraborty S, Kumari A, Rai A, Varjani S, Vinayak V (2023) Strategies and challenges to enhance commercial viability of algal biorefineries for biofuel production. Bioresour Technol 387:129551. https://doi.org/10.1016/j.biortech.2023.129551
Sutherland DL, Ralph PJ (2019) Microalgal bioremediation of emerging contaminants - opportunities and challenges. Water Res 164:114921. https://doi.org/10.1016/j.watres.2019.114921
Moldes D, Rojo EM, Bolado S, Garcia-Encina PA, Comesaña-Gándara B (2022) Biodegradable solvents: a promising tool to recover proteins from microalgae. Appl Sci 12(2391):1–18. https://doi.org/10.3390/app12052391
Waghmare AG, Salve MK, LeBlanc JG, Arya SS (2016) Concentration and characterization of microalgae proteins from Chlorella pyrenoidosa. Biores Bioproc 3(1). https://doi.org/10.1186/s40643-016-0094-8
Safi C, Frances C, Ursu AV, Laroche C, Pouzet C, Vaca-Garcia C, Pontalier P (2015) Understanding the effect of cell disruption methods on the diffusion of Chlorella vulgaris proteins and pigments in the aqueous phase. Algal Res 8:61–68. https://doi.org/10.1016/j.algal.2015.01.002
Phong WN, Le CF, Show PL, Lam HL, Ling TC (2016) Evaluation of different solvent types on the extraction of proteins from microalgae. Chem Eng Trans 52:1063–1068. https://doi.org/10.3303/CET1652178
Lam MK, Lee KT (2015) Bioethanol production from microalgae. In: Kim S-K (ed) Handbook of marine microalgae. Elsevier Inc., pp 197–208
Alhattab M, Kermanshahi-Pour A, Brooks MS (2018) Microalgae disruption techniques for product recovery: influence of cell wall composition. J Appl Phycol 31(1):61–88. https://doi.org/10.1007/s10811-018-1560-9
Bleakley S, Hayes M (2017) Algal proteins: extraction, application, and challenges concerning production. Foods 6(5). https://doi.org/10.3390/foods6050033
Espinosa-Ramirez J, Mondragon-Portocarrero AC, Rodriguez JA, Lorenzo JM, Santos EM (2023) Algae as a potential source of protein meat alternatives. Front Nutr 10:1254300. https://doi.org/10.3389/fnut.2023.1254300
de Souza Celente G, Sui Y, Acharya P (2023) Seaweed as an alternative protein source: prospective protein extraction technologies. Innov Food Sci Emerg Technol 86:103374. https://doi.org/10.1016/j.ifset.2023.103374
Parimi NS, Singh M, Kastner JR, Das KC, Forsberg LS, Azadi P (2015) Optimization of protein extraction from Spirulina platensis to generate a potential co-product and a biofuel feedstock with reduced nitrogen content. Front Energy Res 3. https://doi.org/10.3389/fenrg.2015.00030
D’Hondt E, Martín-Juárez J, Bolado S, Kasperoviciene J, Koreiviene J, Sulcius S, Elst K, Bastiaens L (2017) Cell disruption technologies. In: Cristina Gonzalez-Fernandez RM (ed) Microalgae-based biofuels and bioproducts. Elsevier Inc., pp 133–154
Ahirwar A, Meignen G, Khan MJ, Khan N, Rai A, Schoefs B, Marchand J, Varjani S, Vinayak V (2021) Nanotechnological approaches to disrupt the rigid cell walled microalgae grown in wastewater for value-added biocompounds: commercial applications, challenges, and breakthrough. Biomass Convers Biorefin 13(15):13309–13334. https://doi.org/10.1007/s13399-021-01965-1
Mendes-Pinto MM, Raposo MFJ, Bowen J, Young AJ, Morais R (2001) Evaluation of different cell disruption process on encysted cells of Haematococcus pluvialis. J App Phycol 13:19–24. https://doi.org/10.1023/A:1008183429747
Liu Y, Liu X, Cui Y, Yuan W (2022) Ultrasound for microalgal cell disruption and product extraction: a review. Ultrason Sonochem 87:106054. https://doi.org/10.1016/j.ultsonch.2022.106054
Safi C, Ursu AV, Laroche C, Zebib B, Merah O, Pontalier P, Vaca-Garcia C (2014) Aqueous extraction of proteins from microalgae: effect of different cell disruption methods. Algal Res 3:61–65. https://doi.org/10.1016/j.algal.2013.12.004
Kapoore RV, Butler TO, Pandhal J, Vaidyanathan S (2018) Microwave-assisted extraction for microalgae: from biofuels to biorefinery. Biology (Basel) 7(1). https://doi.org/10.3390/biology7010018
Bernaerts TMM, Gheysen L, Foubert I, Hendrickx ME, Van Loey AM (2019) Evaluating microalgal cell disruption upon ultra high pressure homogenization. Algal Res 42:101616. https://doi.org/10.1016/j.algal.2019.101616
Al-Zuhair S, Ashraf S, Hisaindee S, Darmaki NA, Battah S, Svistunenko D, Reeder B, Stanway G, Chaudhary A (2017) Enzymatic pre-treatment of microalgae cells for enhanced extraction of proteins. Eng Life Sci 17(2):175–185. https://doi.org/10.1002/elsc.201600127
Chiong T, Acquah C, Lau SY, Khor EH, Danquah MK (2016) Microalgal-based protein by-products: extraction, purification, and applications. In: Dhillon GS (ed) Protein Byproducts. Elsevier Inc., pp 213–234
Martins PL, Reis A, Duarte LC, Carvalheiro F (2022) Effective fractionation of microalgae biomass as an initial step for its utilization as a bioenergy feedstock. Energy Convers Manag: X 16:100317. https://doi.org/10.1016/j.ecmx.2022.100317
Buchanan M (2007) Solvent extractives of wood and pulp (proposed revision of T 204 cm-97). Tappi Press, Atlanta, GA
Sluiter A, Ruiz R, Scarlata C, Sluiter J, Templeton D (2008) Determination of extractives in biomass. (NREL/TP-510-42619). National Renewable Energy Laboratory, Golden, CO
Sluiter A, Hames B, Ruiz R, Scarlata C, Sluiter J, Templeton D (2008) Determination of ash in biomass. In: Laboratory analytical procedure (LAP). (NREL/TP-510-42622). National Renewable Energy Laboratory, Golden, CO
AOAC (1975) AOAC Official Methods of Analysis - "Total protein nitrogen: evaluation and comparison of four different methods". AOAC international, Washington D.C
Laurens LML (2015) Summative mass analysis of algal biomass – Integration of analytical procedures. (NREL/TP-5100-60943). National Renewable Energy Laboratory, Golden, CO
Wychen SV, Laurens LML (2015) Determination of total carbohydrates in algal biomass. (NREL/TP-5100-60957). National Renewable Energy Laboratory, Golden, CO
Branco PC, Dionísio AM, Torrado I, Carvalheiro F, Castilho PC, Duarte LC (2015) Autohydrolysis of Annona cherimola mill. seeds: optimization, modeling and products characterization. Biochem Eng J 104(12):2–9. https://doi.org/10.1016/j.bej.2015.06.006
Ursu AV, Marcati A, Sayd T, Sante-Lhoutellier V, Djelveh G, Michaud P (2014) Extraction, fractionation and functional properties of proteins from the microalgae Chlorella vulgaris. Bioresour Technol 157:134–139. https://doi.org/10.1016/j.biortech.2014.01.071
Barka A, Blecker C (2016) Microalgae as a potential source of single-cell proteins. A review. Biotechnol Agron Soc Environ 20(3):427–436. https://doi.org/10.25518/1780-4507.13132
Lardon L, Helias A, Sialve B, Steyer JP, Bernard O (2009) Life-cycle assessment of biodiesel production from microalgae. Environ Sci Technol 43(17):6475–6481. https://doi.org/10.1021/es900705j
Fajardo AR, Cerdán LE, Medina AR, Fernández FGA, Moreno PAG, Grima EM (2007) Lipid extraction from the microalga Phaeodactylum tricornutum. Eur J Lipid Sci Technol 109(2):120–126. https://doi.org/10.1002/ejlt.200600216
Yang F, Cheng C, Long L, Hu Q, Jia Q, Wu H, Xiang W (2015) Extracting lipids from several species of wet microalgae using ethanol at room temperature. Energy Fuels 29(4):2380–2386. https://doi.org/10.1021/ef5023576
Sriariyanun M, Show PL, Chew KW, Khoo KS, Tawai A, Cheng Y, Bhattacharyya D, Rodiahwati W, Panakkal EJ (2022) Effective solvents for proteins recovery from microalgae. E3S Web of Conferences 355:02009. https://doi.org/10.1051/e3sconf/202235502009
Nikolaidis A, Andreadis M, Moschakis T (2017) Effect of heat, pH, ultrasonication and ethanol on the denaturation of whey protein isolate using a newly developed approach in the analysis of difference-UV spectra. Food Chem 232:425–433. https://doi.org/10.1016/j.foodchem.2017.04.022
Noreen A, Mahmood S, Aziz I, Takriff MS (2021) Microalgae as potential protein sources: evidence from protein extraction and amino acid profiling of Chlorella vulgaris and Scenedesmus SP. Pak J Agric Res 53(3):821–829. https://doi.org/10.21162/PAKJAS/21.511
Pereira H (2019) Biotechnological applications of a promising marine chlorophyte (Tetraselmis sp. CTP4): a biorefinery approach. PhD thesis, Universidade do Algarve, Portugal
Taucher J, Baer S, Schwerna P, Hofmann D, Hümmer M, Buchholz R, Becker A (2016) Cell disruption and pressurized liquid extraction of carotenoids from microalgae. Microalgae J Thermodyn Catal 7(158):1–7. https://doi.org/10.4172/2158-7544.1000158
Azencott HR, Peter GF, Prausnitz MR (2007) Influence of the cell wall on intracellular delivery to algal cells by electroporation and sonication. Ultrasound Med Biol 33(11):1805–1817. https://doi.org/10.1016/j.ultrasmedbio.2007.05.008
Mercer P, Armenta RE (2011) Developments in oil extraction from microalgae. Eur J Lipid Sci Technol 113(5):539–547. https://doi.org/10.1002/ejlt.201000455
Soto-Sierra L, Stoykova P, Nikolov ZL (2018) Extraction and fractionation of microalgae-based protein products. Algal Res 36:175–192. https://doi.org/10.1016/j.algal.2018.10.023
Rashidi B, Trindade LM (2018) Detailed biochemical and morphologic characteristics of the green microalga Neochloris oleoabundans cell wall. Algal Res 35:152–159. https://doi.org/10.1016/j.algal.2018.08.033
Sierra LS, Dixon CK, Wilken LR (2017) Enzymatic cell disruption of the microalgae Chlamydomonas reinhardtii for lipid and protein extraction. Algal Res 25:149–159. https://doi.org/10.1016/j.algal.2017.04.004
Sari YW, Sanders JPM, Bruins ME (2016) Techno-economical evaluation of protein extraction for microalgae biorefinery. IOP Conf Series: Earth Environ Sci 31:012034. https://doi.org/10.1088/1755-1315/31/1/012034
Gifuni I, Lavenant L, Pruvost J, Masse A (2020) Recovery of microalgal protein by three-steps membrane filtration: advancements and feasibility. Algal Res 51:102082. https://doi.org/10.1016/j.algal.2020.102082
Duong-Ly KC, Gabelli SB (2014) Salting out of proteins using ammonium sulfate precipitation. Methods Enzymol 541:85–94. https://doi.org/10.1016/B978-0-12-420119-4.00007-0
Vilg JV, Undeland I (2017) pH-driven solubilization and isoelectric precipitation of proteins from the brown seaweed Saccharina latissima-effects of osmotic shock, water volume and temperature. J Appl Phycol 29(1):585–593. https://doi.org/10.1007/s10811-016-0957-6
Chen Y, Stevens MA, Zhu Y, Holmes J, Xu H (2013) Understanding of alkaline pretreatment parameters for corn stover enzymatic saccharification. Biotechnol Biofuels 6(8):2–11. https://doi.org/10.1186/1754-6834-6-8
Mahdy A, Mendez L, Ballesteros M, González-Fernández C (2014) Autohydrolysis and alkaline pretreatment effect on Chlorella vulgaris and Scenedesmus sp. methane production. Energy 78:48–52. https://doi.org/10.1016/j.energy.2014.05.052
Teh YY, Lee KT, Chen WH, Lin SC, Sheen HK, Tan IS (2017) Dilute sulfuric acid hydrolysis of red macroalgae Eucheuma denticulatum with microwave-assisted heating for biochar production and sugar recovery. Bioresour Technol 246:20–27. https://doi.org/10.1016/j.biortech.2017.07.101
Ben Yahmed N, Berrejeb N, Jmel MA, Jazzar S, Marzouki MN, Smaali I (2018) Efficient biocatalytic conversion of stranded green macroalgal biomass using a specific cellulases-based cocktail. Waste Biomass Valor 11(1):211–222. https://doi.org/10.1007/s12649-018-0397-4
Poulhazan A, Dickwella Widanage MC, Muszynski A, Arnold AA, Warschawski DE, Azadi P, Marcotte I, Wang T (2021) Identification and quantification of glycans in whole cells: architecture of microalgal polysaccharides described by solid-state nuclear magnetic resonance. J Am Chem Soc 143(46):19374–19388. https://doi.org/10.1021/jacs.1c07429
Flores EMM, Cravotto G, Bizzi CA, Santos D, Iop GD (2021) Ultrasound-assisted biomass valorization to industrial interesting products: state-of-the-art, perspectives and challenges. Ultrason Sonochem 72:105455. https://doi.org/10.1016/j.ultsonch.2020.105455
Chanda MJ, Merghoub N, El Arroussi H (2019) Microalgae polysaccharides: the new sustainable bioactive products for the development of plant bio-stimulants? World J Microbiol Biotechnol 35(11):177. https://doi.org/10.1007/s11274-019-2745-3
Karan H, de Boeck R, Roles J, Hankamer B, Ross IL (2019) Hydrothermal pre-treatment coupled with urea solubilisation enables efficient protein extraction from microalgae. Green Chem 21(23):6361–6371. https://doi.org/10.1039/c9gc03120k
Acknowledgements
The authors thank Graça Conceição, Natércia Sousa, Céu Penedo, and Belina Ribeiro, for their technical support.
Funding
Open access funding provided by FCT|FCCN (b-on). This work was partially supported by the project GREENBIOREFINERY (ERANETLAC/0001/2014-ELAC2014/BEE0357). Pedro L. Martins Ph.D. grant was supported by FCT (SFRH/BD/121704/2016). Centro de Estudos Florestais is a research unit funded by FCT under the strategic program PEst-OE/AGR/UI0239/2013). The work was partially carried out at the Biomass and Bioenergy Research Infrastructure (BBRI)-LISBOA-01-0145-FEDER-022059, which is supported by Operational Programme for Competitiveness and Internationalization (PORTUGAL2020), by Lisbon Portugal Regional Operational Programme (Lisboa 2020), and by North Portugal Regional Operational Programme (Norte 2020) under the Portugal 2020 Partnership Agreement, through the European Regional Development Fund (ERDF).
Author information
Authors and Affiliations
Contributions
Pedro L. Martins: formal analysis; investigation; methodology; writing—original draft; review and editing; Alberto Reis: funding acquisition; supervision, resources; review and editing; Luís C. Duarte: formal analysis; methodology; validation; visualization; review and editing; Helena Pereira: formal analysis; supervision; validation; visualization; review and editing; Florbela Carvalheiro: conceptualization; formal analysis; funding acquisition; methodology; supervision; validation; visualization; writing—original draft; review and editing.
Corresponding author
Ethics declarations
Ethical approval
This declaration is not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Martins, P.L., Duarte, L.C., Pereira, H. et al. Evaluation of different fractionation methods for the simultaneous protein and carbohydrate extraction from microalgae. Biomass Conv. Bioref. (2024). https://doi.org/10.1007/s13399-024-05279-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13399-024-05279-w