Abstract
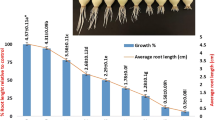
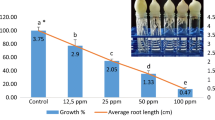
Pinoxaden is the one of the acetyl-CoA carboxylases (ACCase) inhibiting herbicides and used for controlling grass weeds. In this study, cyto-genotoxic effects of Pinoxaden on the Allium cepa roots were investigated using Allium ana-telophase and comet assays by determining the root growth, mitotic index (MI), mitotic phases, chromosomal aberrations (CAs) and DNA damage. Different concentrations of Pinoxaden from 0.5 to 100 mg/L were employed on root tips for 96 h to find the effective concentration that reduces root tip elongation by 50% in comparison with negative control (EC50). Pinoxaden concentrations of 1.25 mg/L (1/2xEC50), 2.5 mg/L (EC50) and 5 mg/L (2xEC50); methyl methane sulphonate (MMS, 10 mg/L) for positive control and distilled water for negative control were exposed to Allium bulbs for several time intervals (24, 48, 72 and 96 h). Pinoxaden showed cytotoxic effects by decreasing the root growth and MI. Pinoxaden induced CAs including disturbed ana-telophase, anaphase bridges, chromosome laggards, stickiness, polyploidy, micronucleus at 5 mg/L, c-metaphase and binuclear cells and also DNA damage compared with control group. The current study confirmed cyto-genotoxic effects of Pinoxaden. Further research is needed to clarify the cyto-genotoxic mechanisms of Pinoxaden at molecular level.



Similar content being viewed by others
References
Aksoy OD, Dane F, Sanal FE, Aktac T (2007) The effects of fusilade (fluazifop-p-butyl) on germination, mitotic frequency and α-amylase activity of lentil (Lens culinaris Medik.) seeds. Acta Physiol Plant 29(2):115–120. https://doi.org/10.1007/s11738-006-0014-1
Akyıl D, Özkara A, Erdoğmuş SF, Eren Y, Konuk M, Sağlam E (2015) Micronucleus assay in human lymphocytes after exposure to alloxydim sodium herbicide in vitro. Cytotechnology 67(6):1059–1066. https://doi.org/10.1007/s10616-014-9746-8
Ateeq B, Farah MA, Ali MN, Ahmad W (2002) Clastogenicity of pentachlorophenol, 2, 4D and butachlor evaluated by Allium root tip test. Mutat Res 514:105–113. https://doi.org/10.1016/S1383-5718(01)00327-8
Australian Pesticides and Veterinary Medicines Authority (APVMA) (2006) Axial 100EC selective herbicide
Barbério A, Voltolini JC, Mello MLS (2011) Standardization of bulb and root sample sizes for the Allium cepa test. Ecotoxicology 20(4):927–935. https://doi.org/10.1007/s10646-011-0602-8
Boumaza A, Lalaoui K, Khallef M, Sbayou H, Talbi H, Hilali A (2016) Assessment of cytotoxic and genotoxic effects of clodinafop-propargyl commercial formulation on Allium cepa L. J Mater Environm Sci 7(4):1245–1251 https://www.researchgate.net/publication/299395190
Clark RD (2018) Predicting mammalian metabolism and toxicity of pesticides in silico. Pest Manag Sci 74(9):1992–2003. https://doi.org/10.1002/ps.4935
Dutta J, Ahmad A, Singh J (2018) Study of industrial effluents induced genotoxicity on Allium cepa L. Caryologia 71(2):139–145. https://doi.org/10.1080/00087114.2018.1447631
El-Ghamery AA, El-Nahas AI, Mansour MM (2000) The action of atrazine herbicide as an indicator of cell division on chromosomes and nucleic acid content in root meristems of Allium cepa and Vicia faba. Cytologia 65:277–287. https://doi.org/10.1508/cytologia.65.277
Fenech M, Kirsch-Volders M, Natarajan AT, Surralles J, Crott JW, Parry J, Thomas P (2011) Molecular mechanisms of micronucleus, nucleoplasmic bridge and nuclear bud formation in mammalian and human cells. Mutagenesis 26(1):125–132. https://doi.org/10.1093/mutage/geq052
Fernandes TC, Mazzeo DEC, Marin-Morales MA (2007) Mechanism of micronuclei formation in polyploidizated cells of Allium cepa exposed to trifluralin herbicide. Pestic Biochem Physiol 88(3):252–259. https://doi.org/10.1016/j.pestbp.2006.12.003
Fiskesjö G (1985) The Allium test as a standard in environmental monitoring. Hereditas 102(1):99–112. https://doi.org/10.1111/j.1601-5223.1985.tb00471.x
Fusconi A, Repetto O, Bona E, Massa N, Gallo C, Dumas-Gaudot E, Berta G (2006) Effect of cadmium on meristem activity and nucleus ploidy in roots of Pisum sativum L. cv. Frisson seedlings. Environ Exp Bot 58:253–260. https://doi.org/10.1016/j.envexpbot.2005.09.008
Grant WF (1994) The present status of higher plant bioassays for the detection of environmental mutagens. Mutat Res 310(2):175–185. https://doi.org/10.1016/0027-5107(94)90112-0
Hwang JI, Lee SE, Kim JE (2017) Comparison of theoretical and experimental values for plant uptake of pesticide from soil. PLoS One 12(2):e0172254. https://doi.org/10.1371/journal.pone.0172254
He YT, Wang W, Shen W, Sun QY, Yin S (2019) Melatonin protects against fenoxaprop-ethyl exposure-induced meiotic defects in mouse oocytes. Toxicology 425:152241. https://doi.org/10.1016/j.tox.2019.152241
Hofer U, Muehlebach M, Hole S, Zoschke A (2006) Pinoxaden–for broad spectrum grass weed management in cereal crops. J Plant Dis Protect 20(5):989–995
Karadeniz A, Kaya B, Savaş B, Topcuoğlu F (2015) Effects of two herbicides, fluazyfop-p-butyl and fenoxaprop-p-ethyl, on genotoxicity in Drosophila smart assay and on proliferation and viability of hek293 cells from the perspective of carcinogenesis. Fresenius Environ Bull 24(6):2052–2054 https://www.researchgate.net/publication/305551613
Karaismailoglu MC (2015) Investigation of the potential toxic effects of prometryne herbicide on Allium cepa root tip cells with mitotic activity, chromosome aberration, micronucleus frequency, nuclear DNA amount and comet assay. Caryologia 68(4):323–329. https://doi.org/10.1080/00087114.2015.1109927
Lovleen AK, Rani A, Devi L, Angurana R, Amin DS (2017) Rерrotoxiсity profiling of bisрyribас sodium, pinoxaden and spinosad pesticides on Drosoрhilа mеlаnogаstеr. Toxicology International 24(3):233-239. https://doi.org/10.22506/ti/2017/v24/i3/166499
Kim JC (1986) A study of mode of action of fluazifop-butyl-II. Fluazifop-butyl effect on cell division, cell enlargement, and protein synthesis in oat (Avena sativa L.) roots. Kor J Weed Sci 6(2):168–173
Kocyigit A, Keles H, Selek S, Guzel S, Celik H, Erel O (2005) Increased DNA damage and oxidative stress in patients with cutaneous leishmaniasis. Mutat Res-Gen Tox En 585(1):71–78. https://doi.org/10.1016/j.mrgentox.2005.04.012
Kumari M, Mukherjee A, Chandrasekaran N (2009) Genotoxicity of silver nanoparticles in Allium cepa. Sci Total Environ 407(19):5243–5246. https://doi.org/10.1016/j.scitotenv.2009.06.024
Küçük D, Liman R (2018) Cytogenetic and genotoxic effects of 2-chlorophenol on Allium cepa L. root meristem cells. Envıron Sci Pollut R 25(36):36117–36123. https://doi.org/10.1007/s11356-018-3502-0
Lata H (2017) Impact of herbicides on biomolecular constituents of Eisenia fetida. J Entomol Scı Zool Stud 5(2):1375–1378
Leme DM, Marine-Morels MA (2009) Allium cepa test in environmental monitoring: a review on its applications. Mutat Res 682(1):71–81. https://doi.org/10.1016/j.mrrev.2009.06.002
Liman R, Ciğerci İH, Öztürk NS (2015) Determination of genotoxic effects of imazethapyr herbicide in Allium cepa root cells by mitotic activity, chromosome aberration, and comet assay. Pestic Biochem Physiol 118:38–42. https://doi.org/10.1016/j.pestbp.2014.11.007
Linda PC, Kim YS, Tong L (2010) Mechanism for the inhibition of the carboxyltransferase domain of acetyl-coenzyme A carboxylase by pinoxaden. Pr N Acad Sci 107(51):22072–22077. https://doi.org/10.1073/pnas.1012039107
Livanos P, Galatis B, Quader H, Apostolakos P (2012) Disturbance of reactive oxygen species homeostasis induces atypical tubulin polymer formation and affects mitosis in root-tip cells of Triticum turgidum and Arabidopsis thaliana. Cytoskeleton 69(1):1–21. https://doi.org/10.1002/cm.20538
Meng XL, Xu CB, Hui XJ (2010) Effects of two herbicides on DNA damage of bone marrow cells in mice. Chin J Pub He 6:80
Mesi A, Kopliku D (2013) Cytotoxic and genotoxic potency screening of two pesticides on Allium cepa L. Proc Tech 8(19):26. https://doi.org/10.1016/j.protcy.2013.11.005
Özkan S, Liman R (2019) Cytotoxicity and genotoxicity in Allium cepa L. root meristem cells exposed to the herbicide penoxsulam. Celal Bayar University J Sci 15(2):221–226. https://doi.org/10.18466/cbayarfbe.533466
Özkul M, Özel ÇA, Yüzbaşıoğlu D, Ünal F (2016) Does 2, 4-dichlorophenoxyacetic acid (2, 4-D) induce genotoxic effects in tissue cultured Allium roots? Cytotechnology 68(6):2395–2405. https://doi.org/10.1007/s10616-016-9956-3
Palsikowski PA, Roberto MM, Sommaggio LR, Souza PM, Morales AR, Marin-Morales MA (2018) Ecotoxicity evaluation of the biodegradable polymers PLA, PBAT and its blends using Allium cepa as test organism. J Polym Environ 26:938–945. https://doi.org/10.1007/s10924-017-0990-9
Radwan DEM (2012) Salicylic acid induced alleviation of oxidative stress caused by clethodim in maize (Zea mays L.) leaves. Pestic Biochem Physiol 102(2):182–188. https://doi.org/10.1016/j.pestbp.2012.01.002
Rajeshwari A, Suresh S, Chandrasekaran N, Mukherjee A (2016) Toxicity evaluation of gold nanoparticles using an Allium cepa bioassay. RSC Adv 6(29):24000–24009. https://doi.org/10.1039/c6ra04712b
Rosculete C, Bonciu E, Rosculete E, Olaru L (2019) Determination of the environmental pollution potential of some herbicides by the assessment of cytotoxic and genotoxic effects on Allium cepa. Int J Env Res Pub He 16(1):75. https://doi.org/10.3390/ijerph16010075
Saxena PN, Chauhan LKS, Gupta SK (2005) Cytogenetic effects of commercial formulation of cypermethrin in root meristem cells of Allium sativum: spectroscopic basis of chromosome damage. Toxicology 216(2–3):244–252. https://doi.org/10.1016/j.tox.2005.08.008
Sharma S, Vig AP (2012) Genotoxicity of atrazine, avenoxan, diuron and quizalofop-P-ethyl herbicides using the Allium cepa root chromosomal aberration assay. Terr Aquat Environ Toxicol 6(2):90–95
Silveira GL, Lima MGF, dos Reis GB, Palmieri MJ, Andrade-Vieria LF (2017) Toxic effects of environmental pollutants: comparative investigation using Allium cepa L. and Lactuca sativa L. Chemosphere 178:359–367. https://doi.org/10.1016/j.chemosphere.2017.03.048
Tice RR, Agurell E, Anderson D, Burlinson B, Hartmann A, Kobayashi H, Sasaki YF (2000) Single cell gel/comet assay: guidelines for in vitro and in vivo genetic toxicology testing. 35(3):206–221. https://doi.org/10.1002/(SICI)1098-2280(2000)35:3<206::AID-EM8>3.0.CO;2-J
Tkalec M, Malarić K, Pavlica M, Pevalek-Kozlina B, Vidaković-Cifrek Ž (2009) Effects of radiofrequency electromagnetic fields on seed germination and root meristematic cells of Allium cepa L. Mutat Res 672:76–81. https://doi.org/10.1016/j.mrgentox.2008.09.022
Ulus B, Çeliksöz M, Özhan G (2017) Cytotoxicity and genotoxicity of fenoxaprop-p-ethyl and fluzifob-p-butyl herbicides. Istanbul J Pharm 47(1):5–8. https://doi.org/10.5152/IstanbulJPharm.2017.002
Yin XH, Zhu GN, Zhuang HS, Wu HM, Li SN (2008) Effects of clodinafop-propargyl on DNA damage of hemocytes in silkworm. J Zhejiang Univ Agric Life Sci 34(4):361–366
Yildiz M, Arikan ES (2008) Genotoxicity testing of quizalofop-P-ethyl herbicide using the Allium cepa anaphase–telophase chromosome aberration assay. Caryologia 61(1):45–52. https://doi.org/10.1080/00087114.2008.10589608
Yüzbaşioğlu D, Ünal F, Sancak C (2009) Genotoxic effects of herbicide Illoxan (diclofop-methyl) on Allium cepa L. Turk J Biol 33(4):283–290. https://doi.org/10.3906/biy-0807-23
Webster PL, Macleod RD (1996) The root apical meristem and its margin. In: Waishel Y, Eshel A, Kafkafi U (eds) Plant roots. The hidden half, second edn. Marcel Dekker, New York, pp 51–76
Wenxiang XIAO, Jizhi MENG, Zuobin YANG, Mengdao LIU, Tingchu MA, Liang D, Xiwu GAO (2015) Efficacy assessment of 50 g/l pinoxaden EC for the control of Phalaris arundinacea in barley field. Agric Sci Technol 16(10):2314–2317
Author information
Authors and Affiliations
Contributions
G.A. and R.L. contributed to the design and implementation of the research, to the analysis of the results and to the writing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by: Izabela Pawłowicz
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Aydın, G., Liman, R. Cyto-genotoxic effects of Pinoxaden on Allium cepa L. roots. J Appl Genetics 61, 349–357 (2020). https://doi.org/10.1007/s13353-020-00560-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13353-020-00560-w