Abstract
Background
There is limited data from India regarding medical management of congenital hyperinsulinism (CHI).
Objective
To study the molecular diagnosis, medical management and outcomes of children with CHI.
Study design
Ambispective.
Participants
Children with CHI admitted in from December, 2011 till March, 2020 at a tertiary care referral hospital.
Outcomes
Clinical and genetic profile, treatment, and response
Results
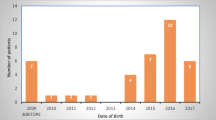
42 children with a median age of 3 days (range 1 day to 6 years) were enrolled, of which 23 (54.7%) were diazoxide-responsive. Mutations were identified in 28 out of 41 (68.2%) patients. The commonest gene affected was ABCC8 in 22 patients. The pathogenic variant c.331G>A in ABCC8 gene was identified in 6 unrelated cases from one community. Good response to daily octreotide was seen in 13 of the 19 (68.4%) diazoxide-unresponsive patients. Monthly long-acting octreotide was initiated and daily octreotide could be stopped or tapered in 9 patients. Sirolimus was tried with variable response in 6 patients but was discontinued in 5 due to adverse effects. Four patients had focal CHI, of which one underwent partial pancreatic resection. The disease severity reduced with age and neurodevelopment was good in the patients with identifiable genetic defects who were optimally managed.
Conclusions
Medical management of CHI is effective, if compliance can be ensured, with good quality of life and neurological outcomes.
Article PDF
Similar content being viewed by others
References
Kapoor RR, Flanagan SE, James C, et al. Hyperinsulinaemic hypoglycaemia. Arch Dis Child. 2009; 94:450–57.
Ludwig A, Ziegenhorn K, Empting S, et al. Glucose metabolism and neurological outcome in congenital hyperinsulinis. Semin Pediatr Surg. 2011;20:45–9.
Kostopoulou E, Shah P. Hyperinsulinaemic-hypogly caemiaan overview of a complex clinical condition. Eur J Pediatr. 2019;178:1151–160.
Yau D, Thomas W, Laver TW, et al. Using referral rates for genetic testing to determine the incidence of a rare disease: The minimal incidence of congenital hyperinsulinism in the UK is 1 in 28,389. PLoS One. 2020;15:e0228417.
Yorifuji T. Congenital hyperinsulinism: Current status and future perspectives. Ann Pediatr Endocrinol Metab. 2014;19: 57–68.
Stanley CA. Perspective on the genetics and diagnosis of congenital hyperinsulinism disorders. J Clin Endocrinol Metab. 2016;101:815–26.
Banerjee I, Salomon-Estebanez M, et al. Therapies and outcomes of congenital hyperinsulinism-induced hypogly-caemia. Diabet Med. 2019;36:9–21.
Arya VB, Senniappan S, Demirbilek H, et al. Pancreatic endocrine and exocrine function in children following near-total pancreatectomy for diffuse congenital hyperinsulinism. PLoS One. 2014;9:98054.
Jahnavi S, Poovazhagi V, Kanthimathi S, et al. Novel ABCC8 (SUR1) gene mutations in Asian Indian children with congenital hyperinsulinemic hypoglycemia. Ann Hum Genet. 2014;78:311–9.
Narayanan E, Kumutha J. Persistent hyperinsulinism—hyper-insulinemic hypoglycemia: follow up and review of 16 cases. Indian J of Appl Res. 2016;6:385–87.
Snider KE, Becker S, Boyajian L, et al. Genotype and phenotype correlations in 417 children with congenital hyperinsulinism. J Clin Endocrinol Metab. 2013;98:355–63.
Kapoor RR, Flanagan SE, Arya VB, et al. Clinical and molecular characterization of 300 patients with congenital hyper-insulinism. Eur J Endocrinol. 2013;168:557–64.
Gupta V, Khadgawat R, Ng HKT, et al. Population structure of Aggarwals of North India as Revealed by Molecular Markers. Genet Test Mol Biomarkers. 2010;14:781–85.
Mazor-Aronovitch K, Gillis D, Lobel D, et al. Long-term neurodevelopmental outcome in conservatively treated congenital hyperinsulinism. Eur J Endocrinol. 2007;157:491–97.
Treglia G, Mirk P, Giordano A, et al. Diagnostic performance of fluorine-18-dihydroxyphenylalanine positron emission tomography in diagnosing and localizing the focal form of congenital hyperinsulinism: A meta-analysis. Pediatr Radiol. 2012;42:1372–379.
Glaser B, Hirsch HJ, Landau H. Persistent hyperinsulinemic hypoglycemia of infancy: Long-term octreotide treatment without pancreatectomy. J Pediatr. 1993;123:644–50.
Astruc B, Marbach P, Bouterfa H, et al. Long-acting octreotide and prolonged-release lanreotide formulations have different pharmacokinetic profiles. J Clin Pharmacol. 2005;45:836–44.
Le Quan Sang KH, Arnoux JB, Mamoune A, et al. Successful treatment of congenital hyperinsulinism with long-acting release octreotide. Eur J Endocrinol. 2012;166:333–39.
Kühnen P, Marquard J, Ernert A, et al. Long-term lanreotide treatment in six patients with congenital hyperinsulinism. Horm Res Paediatr. 2012;78:106–112.
Herrera A, Vajravelu ME, Givler S, et al. Prevalence of adverse events in children with congenital hyperinsulinism treated with diazoxide. J Clin Endocrinol Metab. 2018;103: 4365–72.
Chen SC, Dastamani A, Pintus D, et al. Diazoxide-induced pulmonary hypertension in hyperinsulinaemic-hypoglycaemia: Recommendations from a multicentre study in the United Kingdom. Clin Endocrinol (Oxf). 2019;9:770–75.
Senniappan S, Brown RE, Hussain K. Sirolimus in severe hyperinsulinemic hypoglycemia. N Engl J Med. 2014;370: 2448–9.
Maria G, Antonia D, Michael A, et al. Sirolimus: efficacy and complications in children with hyperinsulinemic hypoglycemia: a 5-year follow-up study. J Endocr Soc. 2019;3:699–713.
Eljamel S, Griffiths A, Evans J, et al. The burden of congenital hyperinsulinism in the United Kingdom: a cost of illness study. Orphanet J Rare Dis. 2018;13:123.
Funding
Funding: SEF has a Sir Henry Dale Fellowship jointly funded by the Wellcome Trust and the Royal Society (Grant Number 105636/Z/14/Z). Costs associated with the genetic testing performed in Exeter were covered by funds donated by Congenital Hyper-insulinism International (a 501(c)3 organization). Costs of genetic testing at MDRF Chennai were covered by Indian Council for Medical Research (ICMR), New Delhi, India, through a project awarded to VR (no. 57/2/2014-NCD-II).
Author information
Authors and Affiliations
Contributions
Contributors: RS, KR: prepared the manuscript; RS, KR, AS, PMN, AK, ND, VJ: involved the diagnostic work up, clinical management of patients and data collection; SEF, JALH, VR, VM: performed the genetic studies; VJ: conceived the study, initiated the collaborations for genetic testing, critically reviewed the manuscript. All authors approved the final manuscript and are accountable for all aspects of the work.
Corresponding author
Ethics declarations
Ethics clearance: Ethics committee of AIIMS, New Delhi; No. IEC 109/5.2.21, RP-26/2021, dated February 05, 2021.
Competing interests: None stated.
Additional information
Note: Additional material related to this study is available with the online version at https://www.indianpediatrics.net
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sharma, R., Roy, K., Satapathy, A.K. et al. Molecular Characterization and Management of Congenital Hyperinsulinism: A Tertiary Centre Experience. Indian Pediatr 59, 105–109 (2022). https://doi.org/10.1007/s13312-022-2438-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-022-2438-0