Abstract
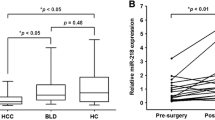
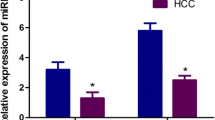
Previous studies have identified a number of microRNAs (miRNAs) that were aberrantly expressed in hepatocellular carcinoma (HCC) tissues. Nevertheless, their diagnostic and prognostic value in serum has not been fully evaluated. Herein, the levels of five serum miRNAs, namely, miR-182, miR-331-3p, miR-197, miR-492, and miR-581, were detected in 103 HCC patients, 95 benign liver diseases, and 40 healthy controls using real-time PCR technique. The results showed that, compared with benign liver diseases and healthy controls, the levels of serum miR-182 and miR-331-3p were significantly increased in HCC patients, both P < 0.001. Area under the receiver–operating characteristic (ROC) curves for serum miR-182 and miR-331-3p were 0.911 (95 % CI, 0.863–0.947) and 0.890 (95 % CI, 0.838–0.930), the sensitivity were 78.6 and 79.61 %, and the specificity were 91.58 and 86.32 %, respectively. Moreover, the combination of serum miR-182, miR-331-3p, and alpha-fetoprotein (AFP) can markedly increase the differential diagnostic value of benign and malignant liver diseases, especially better than serum AFP alone, P < 0.05. Serum miR-182 was positively correlated with serum AFP (P = 0.001), tumor size (P = 0.013), and TNM classification of malignant tumors (TNM) stage (P = 0.003); however, only TNM stage was demonstrated a significant correlation with serum miR-331-3p (P = 0.006). In addition, Kaplan–Meier survival curve, together with univariate and multivariate Cox proportional hazard analyses, further disclosed that serum miR-182 and miR-331-3p were associated with postoperative survival of HCC patients, and both of them were regarded to be independent prognostic factors for patients with HCC. Taken together, our present study indicates that serum miR-182 and miR-331-3p, upregulated in HCC, can provide positive diagnostic and prognostic values for HCC.





Similar content being viewed by others
References
Bruix J, Gores GJ, Mazzaferro V. Hepatocellular carcinoma: clinical frontiers and perspectives. Gut. 2014;63:844–55.
Xu WJ, Guo BL, Han YG, Shi L, Ma WS. Diagnostic value of alpha- fetoprotein-L3 and Golgi protein 73 in hepatocellular carcinomas with low AFP levels. Tumour Biol. 2014;35(12):12069–74.
Debruyne EN, Delanghe JR. Diagnosing and monitoring hepatocellular carcinoma with alpha-fetoprotein: new aspects and applications. Clin Chim Acta. 2008;395(1–2):19–26.
Park H, Park JY. Clinical significance of AFP and PIVKA-II responses for monitoring treatment outcomes and predicting prognosis in patients with hepatocellular carcinoma. Biomed Res Int. 2013;2013:310427.
Giannini EG, Sammito G, Farinati F, Ciccarese F, Pecorelli A, Rapaccini GL, et al. Determinants of alpha-fetoprotein levels in patients with hepatocellular carcinoma: implications for its clinical use. Cancer. 2014;120(14):2150–7.
Han LL, Lv Y, Guo H, Ruan ZP, Nan KJ. Implications of biomarkers in human hepatocellular carcinoma pathogenesis and therapy. World J Gastroenterol. 2014;20:10249–61.
Raza A, Sood GK. Hepatocellular carcinoma review: current treatment, and evidence-based medicine. World J Gastroenterol. 2014;20:4115–27.
Tutar L, Tutar E, Tutar Y. MicroRNAs and cancer; an overview. Curr Pharm Biotechnol. 2014;15:430–7.
Berger F, Reiser MF. Micro-RNAs as potential new molecular biomarkers in oncology: have they reached relevance for the clinical imaging sciences? Theranostics. 2013;3:943–52.
Li B, Huang P, Qiu J, Liao Y, Hong J, Yuan Y. MicroRNA-130a is down- regulated in hepatocellular carcinoma and associates with poor prognosis. Med Oncol. 2014;31:230.
Chen P, Zhao X, Ma L. Downregulation of microRNA-100 correlates with tumor progression and poor prognosis in hepatocellular carcinoma. Mol Cell Biochem. 2013;383:49–58.
Su ZX, Zhao J, Rong ZH, Geng WM, Wu YG, Qin CK. Upregulation of microRNA-25 associates with prognosis in hepatocellular carcinoma. Diagn Pathol. 2014;9:47.
Gu H, Guo X, Zou L, Zhu H, Zhang J. Upregulation of microRNA-372 associates with tumor progression and prognosis in hepatocellular carcinoma. Mol Cell Biochem. 2013;375:23–30.
Wang J, Zhang KY, Liu SM, Sen S. Tumor-associated circulating microRNAs as biomarkers of cancer. Molecules. 2014;19:1912–38.
Meng FL, Wang W, Jia WD. Diagnostic and prognostic significance of serum miR-24-3p in HBV-related hepatocellular carcinoma. Med Oncol. 2014;31:177.
Zhang ZQ, Meng H, Wang N, Liang LN, Liu LN, Lu SM, et al. Serum microRNA 143 and microRNA 215 as potential biomarkers for the diagnosis of chronic hepatitis and hepatocellular carcinoma. Diagn Pathol. 2014;9:135.
Koberle V, Kronenberger B, Pleli T, Trojan J, Imelmann E, Peveling-Oberhag J, et al. Serum microRNA-1 and microRNA-122 are prognostic markers in patients with hepatocellular carcinoma. Eur J Cancer. 2013;49:3442–9.
Zheng H, Liu JY, Song FJ, Chen KX. Advances in circulating microRNAs as diagnostic and prognostic markers for ovarian cancer. Cancer Biol Med. 2013;10:123–30.
Yang Y, Gu X, Zhou M, Xiang J, Chen Z. Serum microRNAs: a new diagnostic method for colorectal cancer. Biomed Rep. 2013;1:495–8.
Yang MH, Yu J, Jiang DM, Li WL, Wang S, Ding YQ. microRNA-182 targets special AT-rich sequence-binding protein 2 to promote colorectal cancer proliferation and metastasis. J Transl Med. 2014;12:109.
Lei R, Tang J, Zhuang X, Deng R, Li G, Yu J, et al. Suppression of MIM by microRNA-182 activates RhoA and promotes breast cancer metastasis. Oncogene. 2014;33:1287–96.
Zhu H, Fang J, Zhang J, Zhao Z, Liu L, Wang J, et al. miR-182 targets CHL1 and controls tumor growth and invasion in papillary thyroid carcinoma. Biochem Biophys Res Commun. 2014;450:857–62.
Wang J, Li J, Shen J, Wang C, Yang L, Zhang X. MicroRNA-182 downregulates metastasis suppressor 1 and contributes to metastasis of hepatocellular carcinoma. BMC Cancer. 2012;12:227.
Qin J, Luo M, Qian H, Chen W. Upregulated miR-182 increases drug resistance in cisplatin-treated HCC cell by regulating TP53INP1. Gene. 2014;538:342–7.
Epis MR, Giles KM, Kalinowski FC, Barker A, Cohen RJ, Leedman PJ. Regulation of expression of deoxyhypusine hydroxylase (DOHH), the enzyme that catalyzes the activation of eIF5A, by miR-331-3p and miR-642-5p in prostate cancer cells. J Biol Chem. 2012;287:35251–9.
Guo X, Guo L, Ji J, Zhang J, Zhang J, Chen X, et al. miRNA-331-3p directly targets E2F1 and induces growth arrest in human gastric cancer. Biochem Biophys Res Commun. 2010;398:1–6.
Epis MR, Giles KM, Candy PA, Webster RJ, Leedman PJ. miR-331-3p regulates expression of neuropilin-2 in glioblastoma. J Neurooncol. 2014;116:67–75.
Chang RM, Yang H, Fang F, Xu JF, Yang LY. MicroRNA-331-3p promotes proliferation and metastasis of hepatocellular carcinoma by targeting PH domain and leucine-rich repeat protein phosphatase. Hepatology. 2014;60:1251–63.
Acknowledgments
This study was supported by National Natural Science Youth Foundation of China (No. 81201351), the project of Jiangsu Provincial Commission of Health and Family Planning (No. H201453), the project of Health Bureau of Nantong city (No. WQ2014065), and the science and technology development project of Nantong City, China (Nos. HS2014061, HS2013054, and HS2011058).
Conflicts of interest
None
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, L., Chu, F., Cao, Y. et al. Serum miR-182 and miR-331-3p as diagnostic and prognostic markers in patients with hepatocellular carcinoma. Tumor Biol. 36, 7439–7447 (2015). https://doi.org/10.1007/s13277-015-3430-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13277-015-3430-2